Application value of rapid on-site evaluation in sentinel lymph node biopsy of breast cancer
-
摘要:
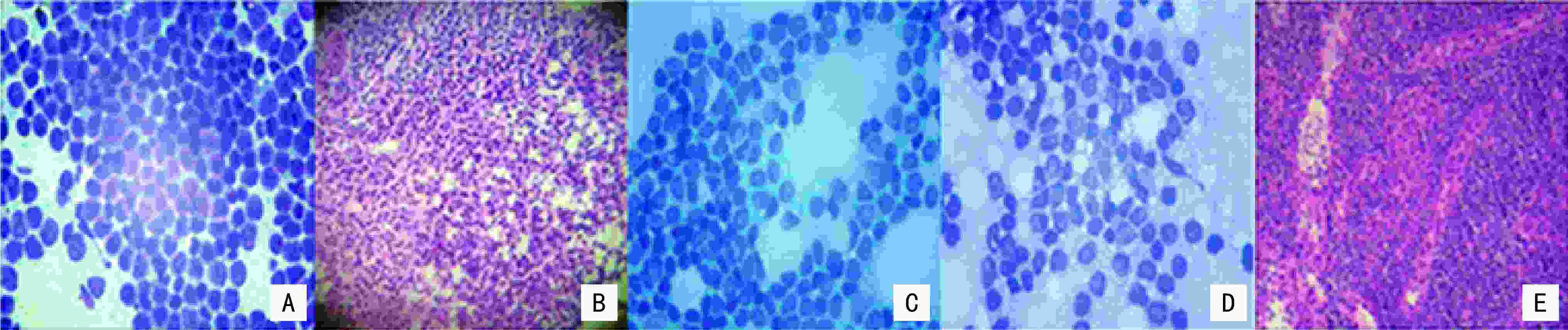
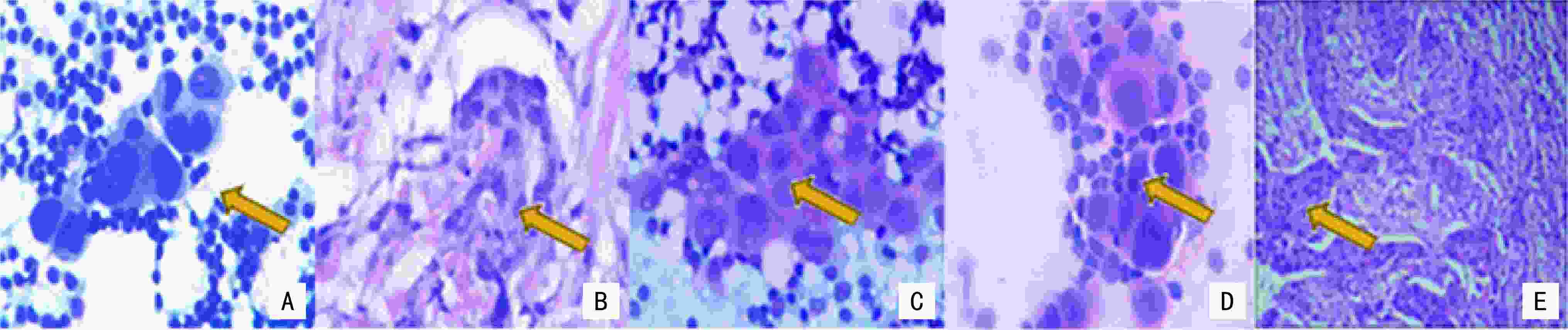
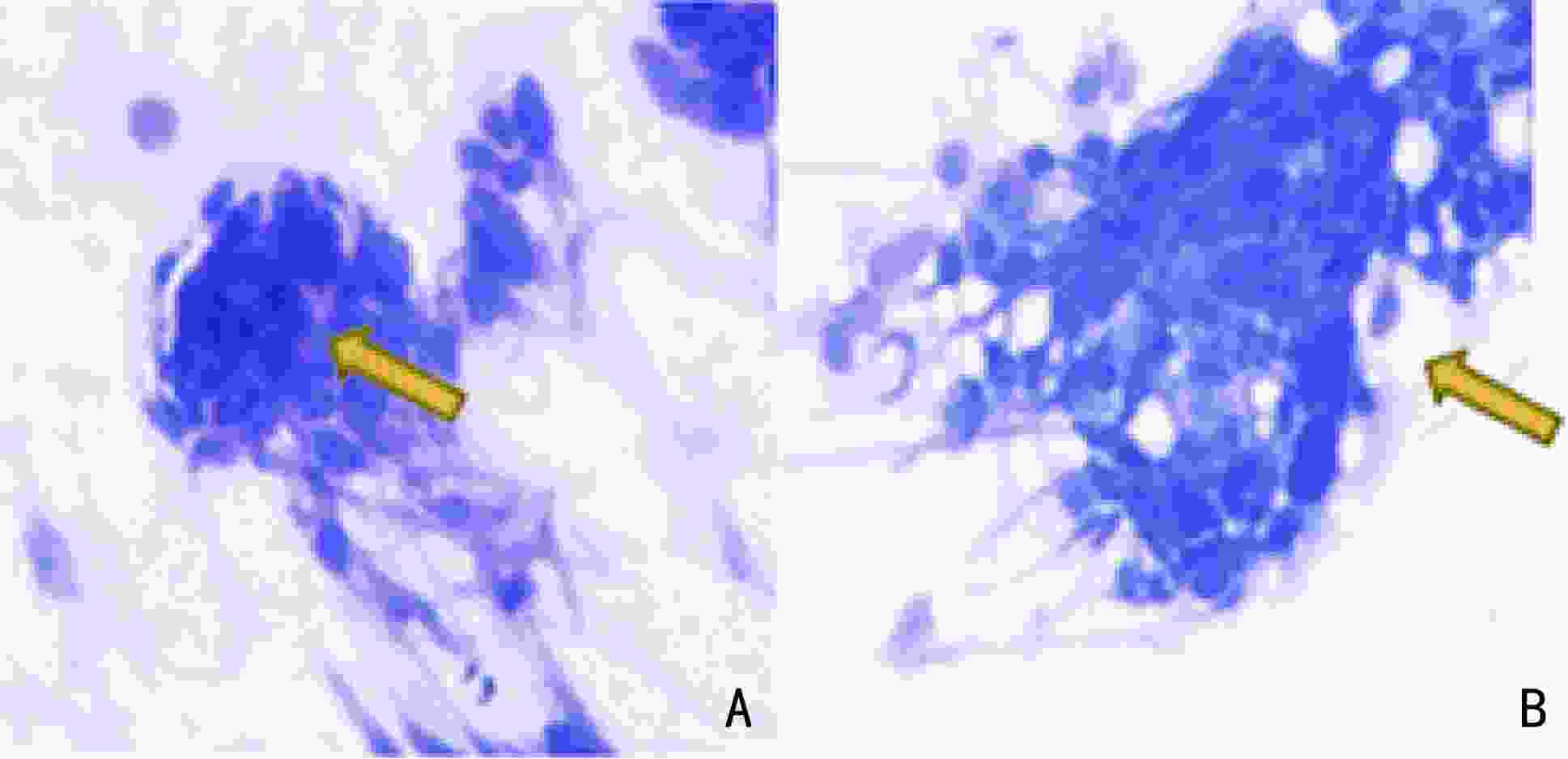
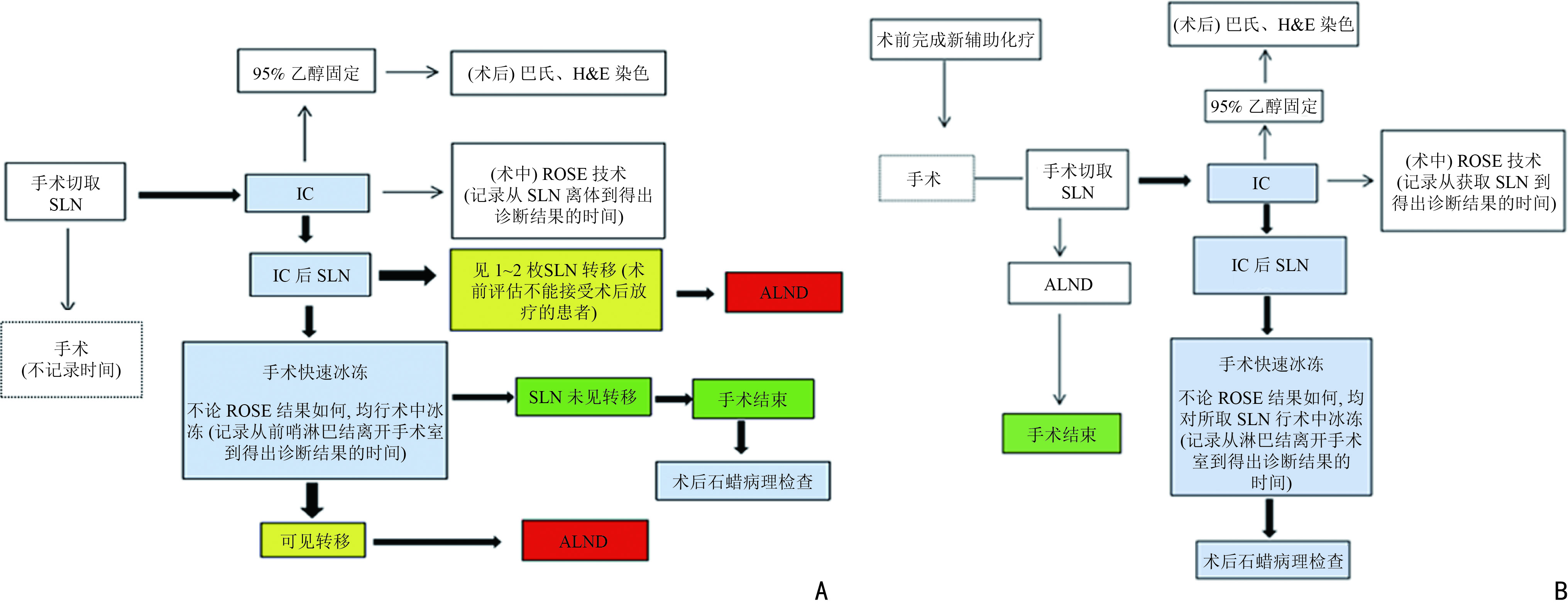
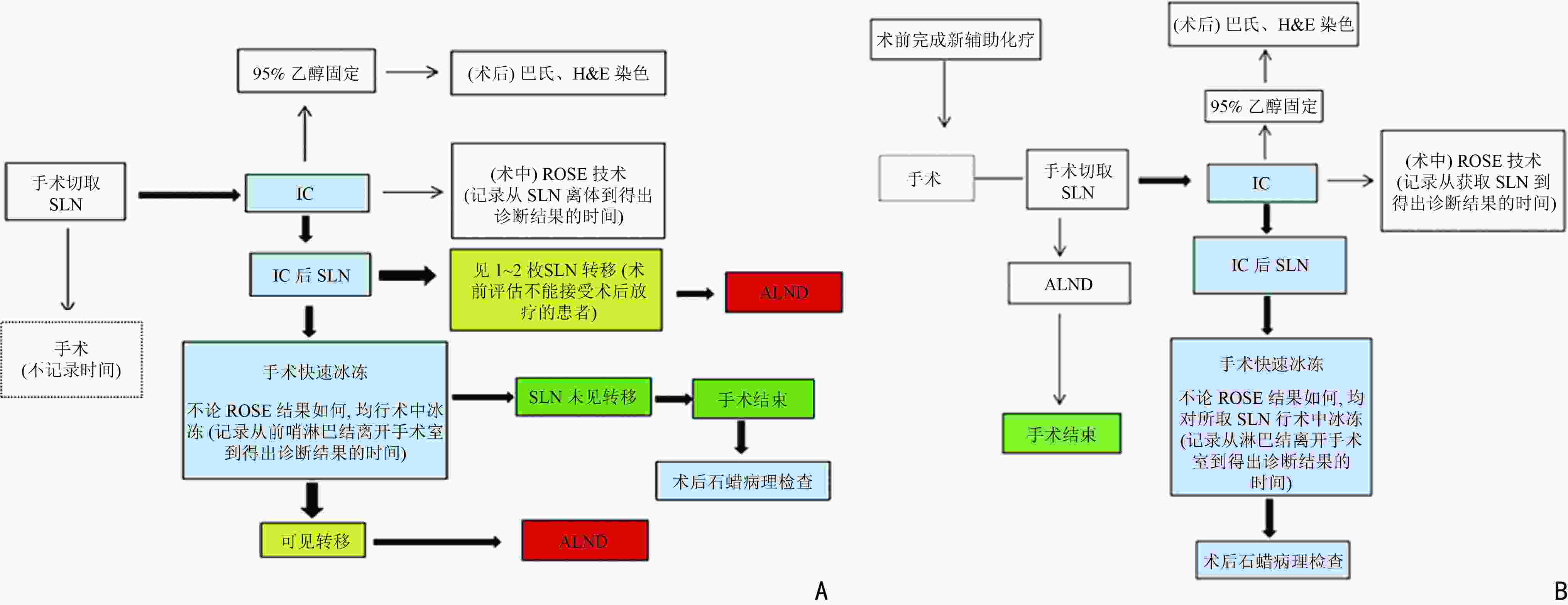
目的 探讨现场快速评估(rapid on-site evaluation,ROSE)技术在乳腺癌前哨淋巴结活检术(sentinel lymph node biopsy,SLNB)中的应用准确性及临床价值。 方法 纳入2019年12月至2021年12月于云南省肿瘤医院确诊的早期乳腺癌患者375例,分为行SLNB组195例(A组)、新辅助化疗后行SLNB组180例(B组),术中应用ROSE技术联合冰冻对A组444枚和B组479枚的前哨淋巴结进行快速诊断。以术后石蜡病理检查为金标准,同时再行细胞学巴氏染色及H&E染色对诊断结果进行比较,绘制各检测方法受试者工作特征( receiver operating characteristic,ROC) 曲线,计算ROC 曲线下面积( area under curve,AUC)并行McNemar 检验,评估ROSE技术的诊断效率、准确度、敏感度及特异度。 结果 ROSE技术平均诊断时间为6.53 min /例。A组和B组的ROSE技术的 AUC值分别为0.929和0.961、敏感度分别为82.35%和93.81%、特异度分别为97.56%和98.43%,A、B组与其他常规检测诊断方法比较,差异均无统计学意义(P=0.180、P=1.000)。 结论 ROSE技术联合冰冻用于乳腺癌SLNB能有效提高诊断效率,具有良好的临床应用及推广价值。 Abstract:Objective To investigate the accuracy and clinical value of rapid on-site evaluation (ROSE) in sentinel lymph node biopsy (SLNB) of breast cancer. Methods Three-hundred and seventy-five patients diagnosed with breast cancer at Yunnan Tumor Hospital from December 2019 to December 2021 were enrolled. One-hundred and ninety-five patients (group A) diagnosed with early breast cancer underwent SLNB alone, and 180 patients (group B) with locally advanced breast cancer underwent SLNB after neoadjuvant therapy. ROSE was used intraoperatively for rapid evaluation and diagnosis of 444 and 479 SLNs in group A and B, respectively. Paraffin was used as the gold standard to compare the diagnostic results. Meanwhile, intraoperative Papanikolaou (PAP) and hematoxylin and eosin (H & E) staining results between the two groups were compared to evaluate the diagnostic accuracy, sensitivity, and specificity of ROSE by McNemar test. The receiver operating characteristic (ROC) curve of each detection method was drawn, and the area under the ROC curve (AUC) was calculated. Results The average diagnosis time of ROSE was 6.53 min/case. The AUC of group A in ROSE was 0.929, with 82.35% sensitivity and 97.56% specificity. The AUC of group B in ROSE was 0.961, with 93.81% sensitivity and 98.43% specificity. The diagnostic results of ROSE showed no statistical differences on using other detection methods(P=0.180, P=1.000). Conclusions ROSE can improve the diagnostic efficiency while reducing the diagnostic cost of SLNB for breast cancer. Moreover, apart from having good clinical application and popularization value, it is expected to become a new method for intraoperative SLNB assessment. -
Key words:
- rapid on-site evaluation /
- breast cancer /
- sentinel lymph node biopsy
-
表 1 375例乳腺癌患者基本特征
病理因素 A组(n=195) B组(n=180) 肿瘤大小 Tis 25 (12.82) 0 (0) T1 112(57.44) 20 (11.11) T2 52 (26.67) 124(68.89) T3 6 (3.07) 36 (20.00) 病理类型 原位癌 25 (11.79) 0 (0) 非特殊浸润性癌 168(87.18) 166(92.22) 其他类型癌 2 (1.03) 14 (7.78) HER-2状态 扩增 24 (12.31) 34(18.89) 未扩增 171(87.69) 146(81.11) 激素受体状态 ER阳性及PR阳性 85 (43.59) 76(42.22) ER阳性及PR阴性 36 (18.46) 48(26.67) ER阴性及PR阳性 15 (7.69) 37(20.56) ER阴性及PR阴性 59 (30.26) 19(10.55) Ki-67增值指数(%) ≤20 76(38.97) 21(11.67) >20 119(61.03) 159(88.33) ()内单位为% 表 2 各诊断技术可见转移SLN数目(枚)
诊断方法 A组 B组 ROSE 30 91 细胞学巴氏染色 28 79 H&E染色 30 82 术中冰冻 29 84 术后石蜡病理检查 34 97 表 3 各诊断技术Z检验及McNemar检验比较
统计学方法A组 B组 ROSE 细胞学巴氏染色 H&E染色 术中冰冻 ROSE 细胞学巴氏染色 H&E染色 术中冰冻 AUC 0.929 0.903 0.923 0.971 0.961 0.894 0.902 0.909 敏感度(%) 82.35 88.29 85.29 88.24 93.81 81.44 84.60 86.67 特异度(%) 97.56 98.29 99.27 97.07 98.43 93.72 93.72 95.13 McNemar 检验 P=0.180 P=1.000 P=0.077 P=0.727 P=1.000 P=0.185 P=0.099 P=0.383 -
[1] DeSantis CE, Ma J, Gaudet MM, et al. Breast cancer statistics, 2019[J]. CA Cancer J Clin, 2019, 69(6):438-451. doi: 10.3322/caac.21583 [2] Rahool R, Haider G, Hayat M, et al. Factors associated with treatment delay in breast cancer: a prospective study[J]. Cureus, 2021, 13(2):e13242. [3] Zhang JL, Wang T, Yan CJ, et al. Clinical practice status of sentinel lymph node biopsy for early-stage breast cancer patients in China: a multicenter study[J]. Clin Epidemiol, 2020, 12:917-924. doi: 10.2147/CLEP.S264349 [4] Aldoheyan T, Klein J. Quality assurance review: intra-operative evaluation of sentinel lymph nodes in breast cancer[J]. Cancer Med, 2021, 10(20):7213-7221. doi: 10.1002/cam4.4264 [5] 中国抗癌协会乳腺癌专业委员会.中国抗癌协会乳腺癌诊治指南与规范(2021年版)[J].中国癌症杂志,2021,31(10):954-1040. doi: 10.19401/j.cnki.1007-3639.2021.10.013 [6] Jain D, Allen TC, Aisner DL, et al. Rapid on-site evaluation of endobronchial ultrasound-guided transbronchial needle aspirations for the diagnosis of lung cancer: a perspective from members of the pulmonary pathology society[J]. Arch Pathol Lab Med, 2018, 142(2):253-262. doi: 10.5858/arpa.2017-0114-SA [7] Huang ZQ, Zhuang DC, Feng AR, et al. Real-time and accuracy of rapid on-site cytological evaluation of lung cancer[J]. Transl Cancer Res, 2021, 10(1):479-486. doi: 10.21037/tcr-20-3294 [8] Boughey JC, Suman VJ, Mittendorf EA, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial[J]. JAMA, 2013, 310(14):1455-1461. doi: 10.1001/jama.2013.278932 [9] 郭宝良,李挺,刘荫华,等.早期乳腺癌染料法前哨淋巴结活检专家共识及技术操作指南(2018版)[J].中国实用外科杂志,2018,38(8):855-858. doi: 10.19538/j.cjps.issn1005-2208.2018.08.04 [10] Wang HS, Ren T, Wang X, et al. Rapid on-site evaluation of touch imprints of biopsies improves the diagnostic yield of transbronchial biopsy for endoscopically nonvisible malignancy: a retrospective study[J]. Jpn J Clin Oncol, 2021, 51(4):622-629. doi: 10.1093/jjco/hyaa245 [11] Petropoulou T, Kapoula A, Mastoraki A, et al. Imprint cytology versus frozen section analysis for intraoperative assessment of sentinel lymph node in breast cancer[J]. Breast Cancer (Dove Med Press), 2017, 9:325-330. [12] Mohammadnia Avval M, Hosseinzadeh M, Farahi Z, et al. Comparing scraping cytology with touch imprint cytology and frozen section analysis in the intraoperative diagnosis of sentinel lymph node metastasis in breast cancer[J]. Diagn Cytopathol, 2021, 49(4):475-479. doi: 10.1002/dc.24695 [13] Classe JM, Loaec C, Gimbergue P, et al. Sentinel lymph node biopsy without axillary lymphadenectomy after neoadjuvant chemotherapy is accurate and safe for selected patients: the GANEA 2 study[J]. Breast Cancer Res Treat, 2019, 173(2):343-352. doi: 10.1007/s10549-018-5004-7 -



 下载:
下载: