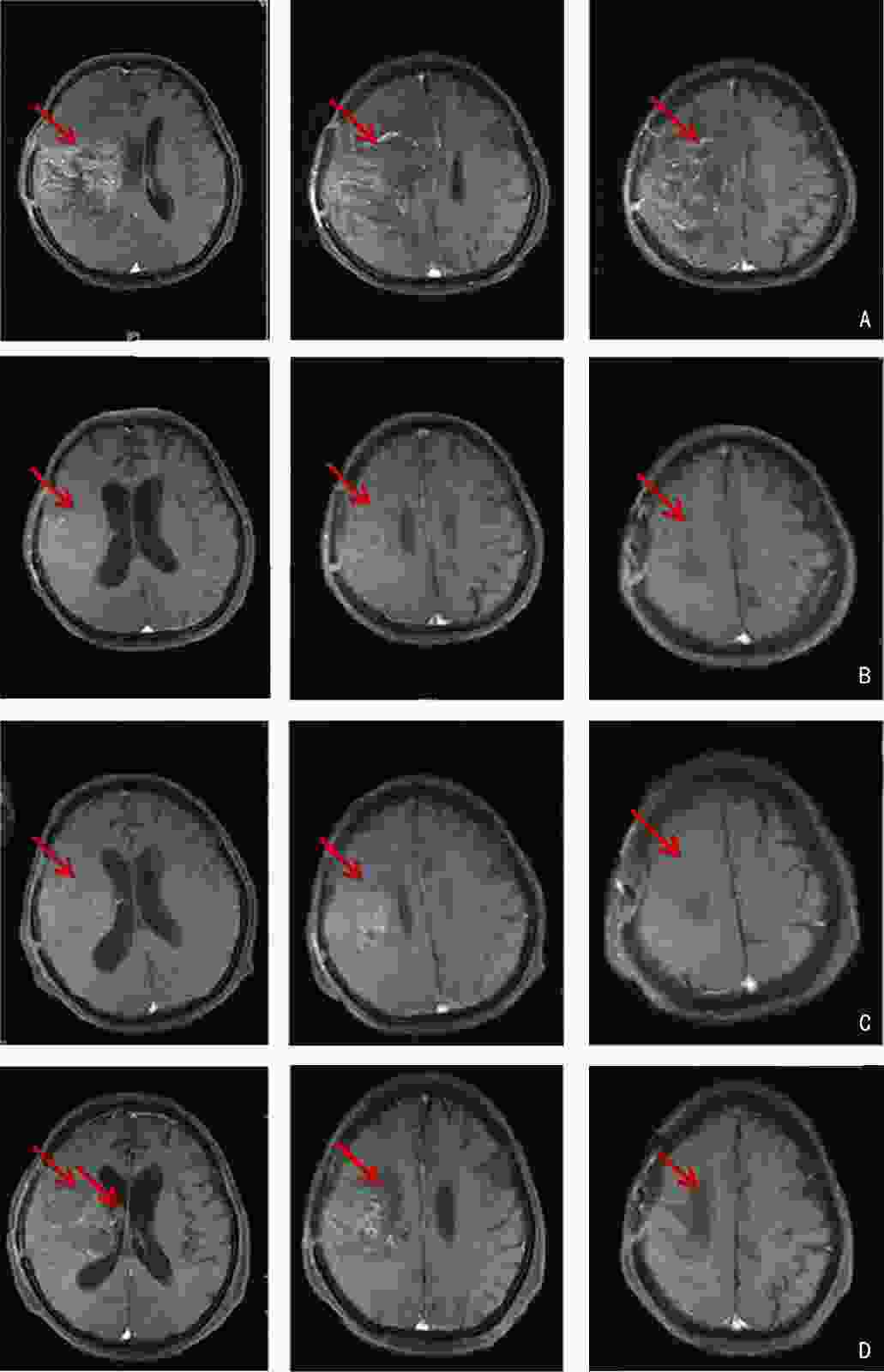
-
-
[1] Clarke J, Butowski N, Chang S. Recent advances in therapy for glioblastoma[J]. Arch Neurol, 2010, 67:279-283. [2] Reardon DA, Turner S, Peters KB, et al. A review of VEGF/VEGFR targeted therapeutics for recurrent glioblastoma[J]. J Natl Compr Canc Netw, 2011, 9(4):414-427. doi: 10.6004/jnccn.2011.0038 [3] Hutterer M, Nowosielski M, Haybaeck F, et al. A single-arm phase Ⅱ Austrian/German multicenter trial on continuous daily sunitinib inprimary glioblastoma at first recurrence (SURGE 01-07)[J]. Neuro Oncol, 2014, 16(1):92-102. doi: 10.1093/neuonc/not161 [4] Pan E, Yu D, Yue B, et al. A prospective phase Ⅱ single-institution trialof sunitinib for recurrent malignant glioma[J]. J Neurooncol, 2012, 110(1):111-118. doi: 10.1007/s11060-012-0943-z [5] Lee EQ, Kuhn J, Lamborn KR, et al. Phase Ⅰ/Ⅱ study of sorafenib incombination with temsirolimus for recurrentglioblastoma or gliosarcoma: North American Brain Tumor Consortium study 05-02[J]. Neuro Oncol, 2012, 14(12):1511-1518. doi: 10.1093/neuonc/nos264 [6] Shen G, Zheng F, Ren D, et al. Anlotinib: a novel multi-targeting tyrosinekinase inhibitor in clinical development[J]. J Hematol Oncol, 2018, 11(1):120. doi: 10.1186/s13045-018-0664-7 [7] Syed YY. Anlotinib: first global approval[J]. Drugs, 2018, 78(10):1057-1062. doi: 10.1007/s40265-018-0939-x [8] Lin B, Song X, Yang D, et al. CTNI-43. Anlotinib inhibits angiogenesis via suppressing the activation of VEGFR2 PDGFR and FGFR1[J]. Gene, 2018, 654:77-86. doi: 10.1016/j.gene.2018.02.026 [9] Lai SZ, Chen YY. Anlotinib combined with STUPP regimen in treating patients with newly diagnosed glioblastoma multiforme: a phase Ⅱ pilot study[J]. Neuro Oncol, 2020, 22(2):52. -



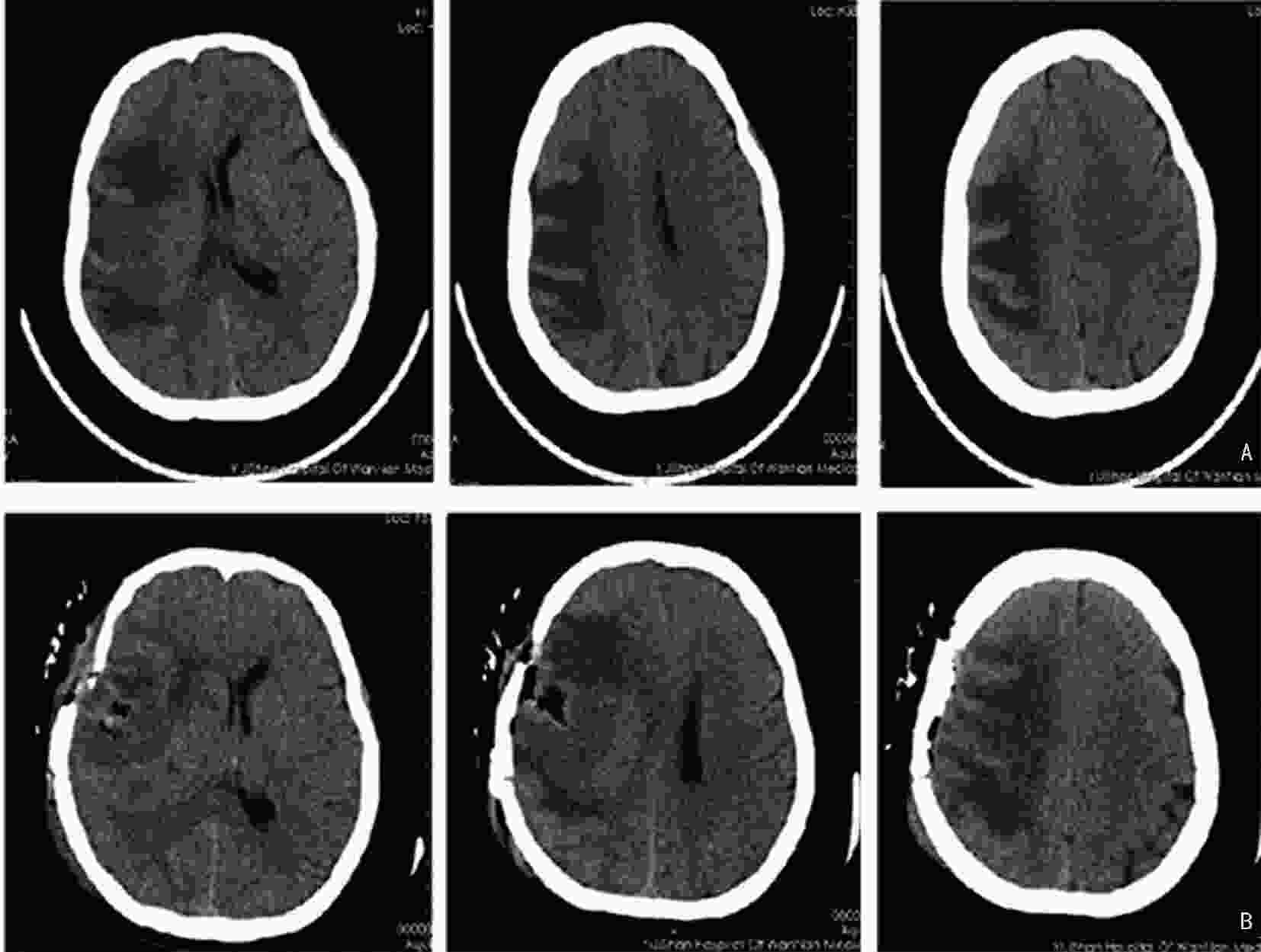
 下载:
下载: