-
摘要:
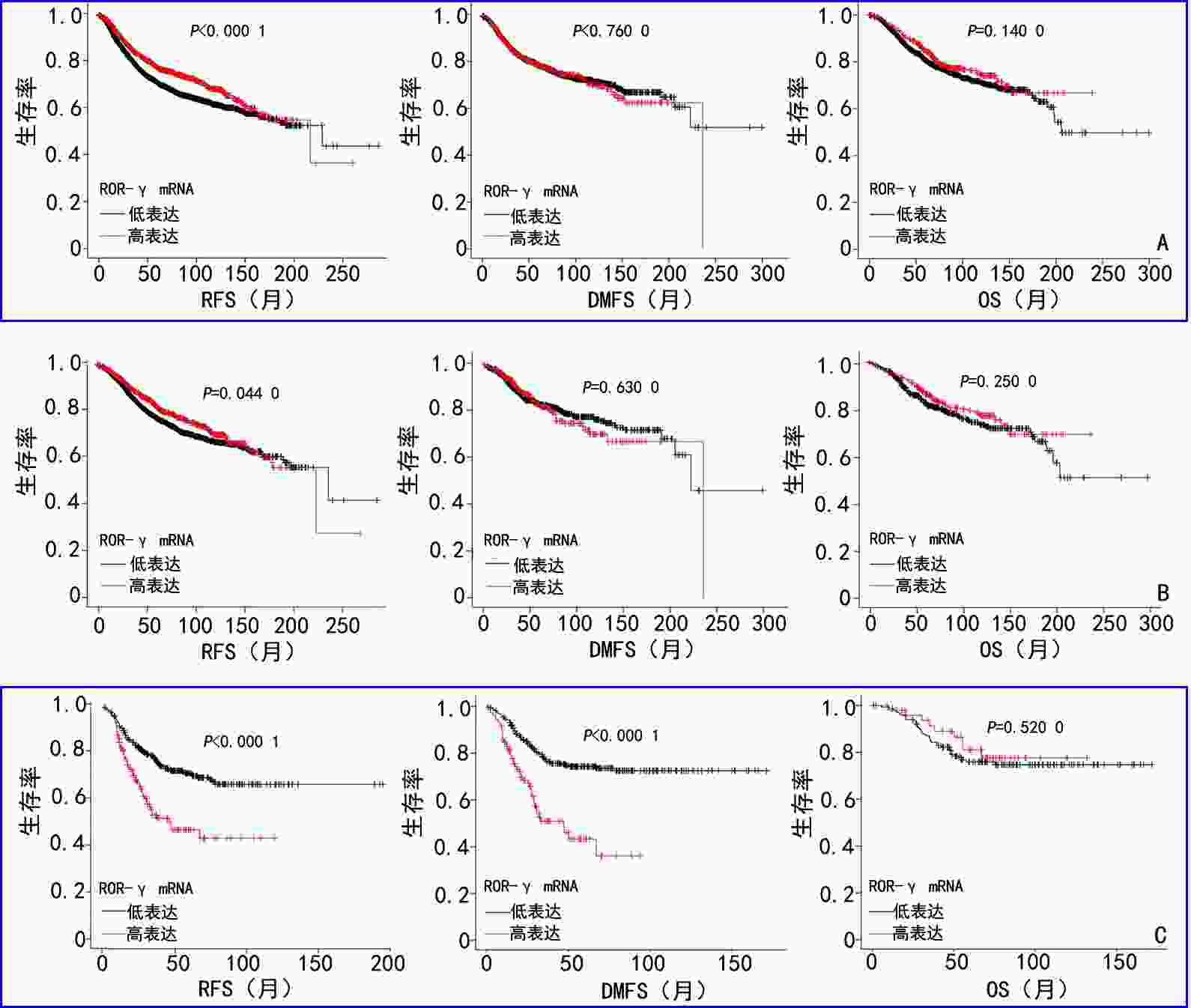
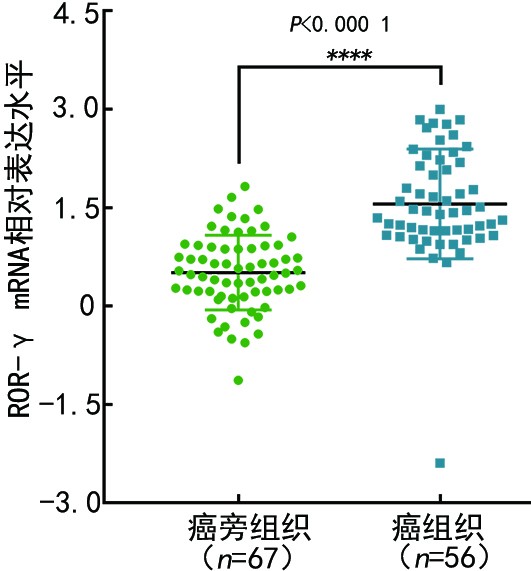
目的 探讨视黄酸受体相关孤儿受体-γ(retinoic acid receptor-related orphan receptor-γ,ROR-γ)在三阴性乳腺癌(triple-negative breast cancer,TNBC)中的表达及临床意义。 方法 采用Oncomine网站数据库分析乳腺浸润性癌与癌旁组织中ROR-γ mRNA的表达差异,通过Kaplan-Meier Plotter网站评估ROR-γ mRNA在乳腺癌不同分子亚型中的表达预后意义。收集2011年1月至2014年12月收治于天津医科大学肿瘤医院的269例TNBC患者的组织标本和临床病理资料,采用免疫组织化学染色法(immunohistochemistry,IHC)检测TNBC组织中的ROR-γ蛋白表达,并探讨其与患者临床病理特征、预后的关系。 结果 Oncomine和Kaplan-Meier Plotter网站分析结果显示,乳腺浸润性癌组织中的ROR-γ mRNA表达显著高于癌旁组织(P<0.000 1)。相较于其他分子亚型,ROR-γ在TNBC中的预后意义更为显著,且与不良预后相关。IHC检测结果显示,ROR-γ蛋白在TNBC组织中的表达高于癌旁组织,ROR-γ在TNBC中的高表达率为18.2%(49/269)。ROR-γ高表达的患者具有较高的发病年龄(P=0.001)、已绝经状态(P<0.001)、高肿瘤Ki-67指数(P=0.001)和淋巴结转移(P=0.003)。ROR-γ高表达是TNBC患者无病生存期(P=0.031)和总生存期(P=0.029)的独立影响因素。 结论 ROR-γ在TNBC组织中高表达,并与肿瘤的增殖活跃、淋巴结转移和患者不良预后相关,或可作为潜在的预后标志物,为TNBC的诊疗提供新的参考。 Abstract:Objective To investigate the expression and clinical significance of retinoic acid receptor-related orphan receptor-γ (ROR-γ) in triple-negative breast cancer (TNBC). Methods Differences in mRNA expression of ROR-γ between invasive breast cancer and adjacent tissues were analyzed via databases on the Oncomine website. The prognostic significance of ROR-γ in different molecular subtypes of breast cancer was evaluated using the Kaplan-Meier Plotter website. The tissue specimens and clinicopathological data of 269 TNBC patients admitted to Tianjin Medical University Cancer Institute and Hospital from January 2011 to December 2014 were collected. Immunohistochemistry (IHC) was performed to investigate the expression of ROR-γ protein in TNBC patients and validate the relationship between ROR-γ expression and the clinical-pathological features and prognosis of the patients. Results Data analysis results on the Oncomine and Kaplan-Meier Plotter websites showed that the expression of ROR-γ mRNA in invasive breast carcinoma tissues was significantly higher than that in adjacent tissues (P<0.0001). Compared with other molecular subtypes, ROR-γ shows a more significant prognostic value in TNBC, related to poor prognoses. IHC results showed that the expression of ROR-γ protein in TNBC tissue was higher than that in adjacent tissues; the high expression rate of ROR-γ in TNBC tissue was 18.2% (49/269). Patients with high ROR-γ expression tended to have an older onset age (P=0.001), a postmenopausal state (P<0.001), a high tumor Ki-67 index (P=0.001), and lymph node metastasis (P=0.003). High ROR-γ expression is an independent influencing factor for the disease-free survival (P=0.031) and overall survival (P=0.029) of TNBC patients. Conclusions ROR-γ is highly expressed in TNBC tissues and is associated with high tumor proliferation, lymph node metastasis, and poor prognosis. Furthermore, ROR-γ may serve as a potential prognostic marker, providing a new reference for the diagnosis and treatment of TNBC. -
表 1 TNBC组织中的ROR-γ蛋白表达与患者临床病理特征的关系
参数 例数(n=269) ROR-γ x2 P 低/无表达(n=220) 高表达(n=49) 年龄(岁) 10.440 0.001 ≤50 116 105(47.7) 11(22.4) >50 153 115(52.3) 38(77.6) 绝经状态 12.604 <0.001 未绝经 104 96(43.6) 8(16.3) 已绝经 165 124(56.4) 41(83.7) 乳腺癌家族史 − 0.090 无 246 198(90.0) 48(98.0) 有 23 22(10.0) 1(2.0) Ki-67(%) 11.536 0.001 <20 16 8(3.6) 8(16.3) ≥20 253 212(96.4) 41(83.7) p53 1.226 0.268 阴性 129 102(46.4) 27(55.1) 阳性 140 118(53.6) 22(44.9) 组织学类型 − 0.584 非特殊类型浸润性癌 253 207(94.1) 46(93.9) 其他特殊类型乳腺癌 16 13(5.9) 3(6.1) 组织学分级(级) 1.955 0.162 G1~G2 124 97(44.1) 27(55.1) G3 145 123(55.9) 22(44.9) 肿瘤T分期 0.292 0.864 T1 95 78(35.5) 17(34.7) T2 159 129(58.6) 30(61.2) T3 15 13(5.9) 2(4.1) 淋巴结状态 9.012 0.003 阴性 166 145(65.9) 21(42.9) 阳性 103 75(34.1) 28(57.1) ()内单位为% 表 2 影响TNBC患者DFS和OS的Cox比例风险回归模型多因素分析
参数 HR 95%CI P DFS 肿瘤T分期(T1 vs. T2 vs. T3) 2.240 1.279~3.923 0.005 淋巴结状态(阴性 vs. 阳性) 2.738 1.435~5.225 0.002 ROR-γ(低/无表达 vs. 高表达) 2.024 1.065~3.845 0.031 OS 肿瘤T分期(T1 vs. T2 vs. T3) 2.426 1.199~4.908 0.014 淋巴结状态(阴性 vs. 阳性) 5.324 1.948~14.548 0.001 ROR-γ(低/无表达 vs. 高表达) 2.403 1.092~5.286 0.029 HR:风险比;CI:置信区间 -
[1] Zhao S, Zuo WJ, Shao ZM, et al. Molecular subtypes and precision treatment of triple-negative breast cancer[J]. Ann Transl Med, 2020, 8(7):499. doi: 10.21037/atm.2020.03.194 [2] Korde LA, Somerfield MR, Carey LA, et al. Neoadjuvant chemotherapy, endocrine therapy, and targeted therapy for breast cancer: ASCO guideline[J]. J Clin Oncol, 2021, 39(13):1485-1505. doi: 10.1200/JCO.20.03399 [3] Garrido-castro AC, Lin NU, Polyak K. Insights into molecular classifications of triple-negative breast cancer: improving patient selection for treatment[J]. Cancer Discov, 2019, 9(2):176-198. doi: 10.1158/2159-8290.CD-18-1177 [4] Kojetin DJ, Burris TP. REV-ERB and ROR nuclear receptors as drug targets[J]. Nat Rev Drug Discov, 2014, 13(3):197-216. doi: 10.1038/nrd4100 [5] Fan J, Lv Z, Yang G, et al. Retinoic acid receptor-related orphan receptors: critical roles in tumorigenesis[J]. Front Immunol, 2018, 9:1187. doi: 10.3389/fimmu.2018.01187 [6] Wang J, Zou JX, Xue X, et al. ROR-gamma drives androgen receptor expression and represents a therapeutic target in castration-resistant prostate cancer[J]. Nat Med, 2016, 22(5):488-496. doi: 10.1038/nm.4070 [7] Huang Y, Liang H, He C, et al. Hepatitis B virus X protein-induced RORgamma expression to promote the migration and proliferation of hepatocellular carcinoma[J]. Biomed Res Int, 2019, 2019:5407126. [8] Gu F, Liu Y, Liu Y, et al. Distinct functions and prognostic values of RORs in gastric cancer[J]. Open Med (Wars), 2020, 15(1):424-434. doi: 10.1515/med-2020-0406 [9] Brozyna AA, Jozwicki W, Skobowiat C, et al. RORalpha and RORgamma expression inversely correlates with human melanoma progression[J]. Oncotarget, 2016, 7(39):63261-63282. doi: 10.18632/oncotarget.11211 [10] Cao D, Qi Z, Pang Y, et al. Retinoic acid-related orphan receptor C regulates proliferation, glycolysis, and chemoresistance via the PD-L1/ITGB6/STAT3 signaling axis in bladder cancer[J]. Cancer Res, 2019, 79(10):2604-2618. doi: 10.1158/0008-5472.CAN-18-3842 [11] Cai D, Wang J, Gao B, et al. RORgamma is a targetable master regulator of cholesterol biosynthesis in a cancer subtype[J]. Nat Commun, 2019, 10(1):4621. doi: 10.1038/s41467-019-12529-3 [12] Oh TG, Wang SM, Acharya BR, et al. The nuclear receptor, RORgamma, regulates pathways necessary for breast cancer metastasis[J]. EBio Medicine, 2016, 6:59-72. [13] Oh TG, Bailey P, Dray E, et al. PRMT2 and RORgamma expression are associated with breast cancer survival outcomes[J]. Mol Endocrinol, 2014, 28(7):1166-1185. doi: 10.1210/me.2013-1403 [14] Cai D, Zhang X, Chen HW. A master regulator of cholesterol biosynthesis constitutes a therapeutic liability of triple negative breast cancer[J]. Mol Cell Oncol, 2020, 7(2):1701362. doi: 10.1080/23723556.2019.1701362 -




 下载:
下载: