Application of a tumor electric field treatment system in patients with recurrent glioblastoma: results from a prospective, single-center, single-arm pilot study
-
摘要:
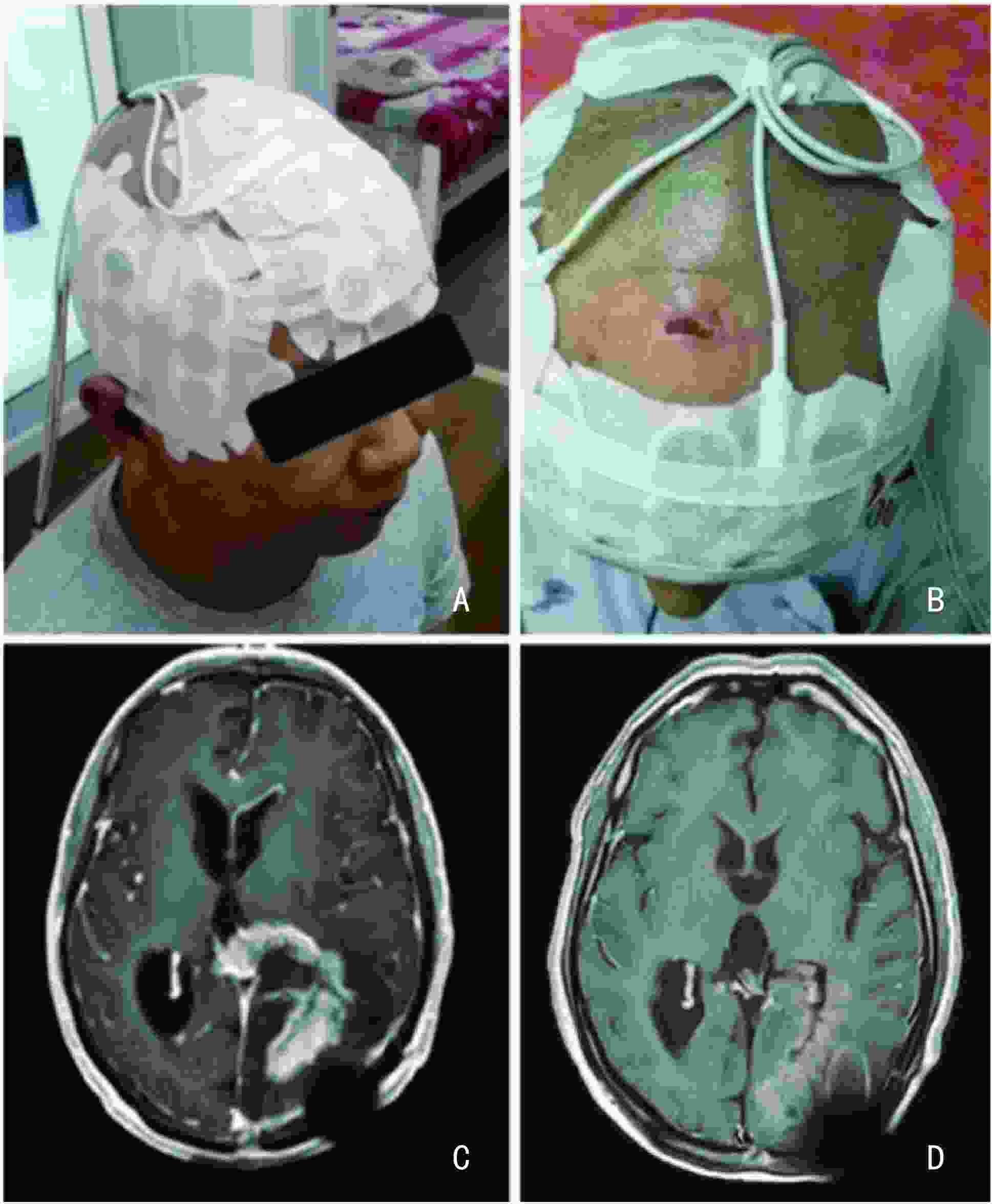
目的 评价肿瘤电场治疗仪用于治疗复发胶质母细胞瘤患者的初步安全性和有效性。 方法 选取2020年4月至2020年8月期间中南大学湘雅医院和解放军总医院6例受试者,均为男性,中位年龄53(48~68)岁。入组复发胶质母细胞瘤受试者并收集临床资料,通过观察受试者治疗期间不良事件的发生率、肿瘤进展时间(time to progression,TTP)和总生存时间(overall survival,OS)评估治疗的安全性和有效性。 结果 入组6例受试者中位随访时间10.9(8.4~22.7)个月,所有受试者发生肿瘤进展,5例随访至死亡,截至随访截止1例生存。中位肿瘤进展时间(median time to progression,mTTP)为4.7个月(95%CI:3.62~5.78),中位无进展生存时间(median progression-free survival,mPFS)为4.7个月(95%CI:3.62~5.78),中位总生存时间(median overall survival,mOS)为10.9个月(95%CI:8.86~12.94)。客观缓解率(CR+PR)33.3%;疾病控制率(SD+PR+CR)83.3%。治疗相关的不良事件主要为头皮皮疹,发生率为66.7%,经调整电极片黏贴位置、保持创口干燥、涂抹类固醇类软膏或抗生素软膏后均可有效缓解。 结论 肿瘤电场治疗仪用于治疗复发胶质母细胞瘤的受试者在电场治疗过程中呈现良好的初步安全性和有效性。 Abstract:Objective To evaluate preliminary safety and effectiveness of a tumor electric field treatment system in patients with recurrent glioblastoma. Methods In total, six patients were enrolled from April 2020 to August 2020 in Xiangya Hospital and Chinese PLA General Hospital. The median age was 53 (48-68) years. Patients with recurrent glioblastoma were enrolled, and their clinical data were collected. The safety and effectiveness of the treatment system were evaluated by assessing the incidence of adverse events, time to progression (TTP), and overall survival (OS). Results In total, the median follow-up time was 10.9 (8.4-22.7) months. Progressive disease was observed in all patients, five patients were followed up to death, and one patient is still alive. The median time to tumor progression (mTTP) was 4.7 months (95% CI: 3.62-5.78), median progression-free survival (mPFS) was 4.7 months (95% CI: 3.62-5.78), and median OS was 10.9 months (95% CI: 8.86-12.94). The objective response rate (CR+PR) was 33.3%; the disease control rate (SD+PR+CR) was 83.3%. The most common treatment-related adverse event was scalp rash, with an incidence rate of 66.7%, which could be effectively relieved by adjusting the sticking position of the transducer arrays, keeping the wound dry, and applying steroid or antibiotic ointment. Conclusions This study showed the preliminary safety and effectiveness of tumor electric field therapy in patients with recurrent glioblastoma. -
Key words:
- glioblastoma /
- tumor electric field treatment /
- safety /
- prognosis
-
表 1 6例复发GBM患者临床特征
项目 受试者编号 1 2 3 4 5 6 年龄(岁) 51 49 55 56 68 48 MGMT启动子 甲基化 甲基化 甲基化 甲基化 甲基化 非甲基化 IDH Mut WT WT WT WT WT TERT Mut Mut Mut Mut Mut Mut 本次复发时间 2020年4月 2020年5月 2020年4月 2020年6月 2020年6月 2020年5月 复发次数(次) 2 1 1 2 1 1 肿瘤部位 右颞、胼胝体 右额、顶 左顶 右丘脑 双额、胼胝体 左颞 再手术和病理 非手术 非手术 手术/GBM 非手术 非手术 手术/GBM 开始治疗时间 2020年4月 2020年5月 2020年5月 2020年6月 2020年7月 2020年8月 最佳反应 SD PD PR SD PR SD 最近TTP评估 2021年11月 2020年7月 2020年9月 2020年10月 2021年1月 2020年12月 是否进展 否 是 是 是 是 是 TTP(PFS,月) >21.2 2.1 4.0 4.7 6.6 4.9 随访截止时间(最近OS) 2022年1月 2021年3月 2021年1月 2021年8月 2021年6月 2021年5月 死亡 否 是 是 是 是 是 OS(月) 22.7 10.9 8.4 16.0 12.4 10.7 表 2 不良事件发生情况
项目 例数(%) 血液学检查 血小板减少 2(33.3) 血胆红素增高 2(33.3) 血清同型半胱氨酸增高 1(16.7) 天冬氨酸氨基转移酶增高 1(16.7) γ-谷氨酰转移酶增高 1(16.7) 高胆固醇血症 2(33.3) 尿酮体升高 1(16.7) 活化部分凝血活酶时间延长 1(16.7) 凝血酶原时间延长 1(16.7) INR增高 1(16.7) 操作并发症 疲劳 1(16.7) 跌倒 1(16.7) 头皮皮疹(医疗器械局部反应) 4(66.7) 电刺激感 1(16.7) 胃肠道疾病 恶心 1(16.7) 呕吐 1(16.7) 代谢与营养 高血糖症 3(50.0) 低钠血症 2(33.3) 高钠血症 1(16.7) 低钾血症 1(16.7) 高甘油三酯血症 2(33.3) 神经系统疾病 头痛 1(16.7) 头晕 1(16.7) -
[1] Korshøj AR, Lukacova S, Mikic N, et al. Tumor treating fields in cancer treatment in Denmark[J]. Ugeskr Laeger, 2019, 181(20A):V07180481. [2] Korshoej AR, Sørensen JCH, von Oettingen G, et al. Optimization of tumor treating fields using singular value decomposition and minimization of field anisotropy[J]. Phys Med Biol, 2019, 64(4):04NT03. [3] Stupp R, Taillibert S, Kanner A, et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial[J]. JAMA, 2017, 318(23):2306-2316. doi: 10.1001/jama.2017.18718 [4] Ghiaseddin AP, Shin D, Melnick K, et al. Tumor treating fields in the management of patients with malignant gliomas[J]. Curr Treat Options Oncol, 2020, 21(9):76. [5] Arvind R, Chandana SR, Borad MJ, et al. Tumor-treating fields: a fourth modality in cancer treatment, new practice updates[J]. Crit Rev Oncol Hematol, 2021, 168:103535. [6] Mun EJ, Babiker HM, Weinberg U, et al. Tumor-treating fields: a fourth modality in cancer treatment[J]. Clin Cancer Res, 2018, 24(2):266-275. [7] Karanam NK, Ding LH, Aroumougame A, et al. Tumor treating fields cause replication stress and interfere with DNA replication fork maintenance: implications for cancer therapy[J]. Transl Res, 2020, 217:33-46. [8] Wang MJ, Zhang CC, Wang X, et al. Tumor-treating fields (TTFields)-based cocktail therapy: a novel blueprint for glioblastoma treatment[J]. Am J Cancer Res, 2021, 11(4):1069-1086. [9] Miranda PC, Mekonnen A, Salvador R, et al. Predicting the electric field distribution in the brain for the treatment of glioblastoma[J]. Phys Med Biol, 2014, 59(15):4137-4147. [10] Wenger C, Salvador R, Basser PJ, et al. The electric field distribution in the brain during TTFields therapy and its dependence on tissue dielectric properties and anatomy: a computational study[J]. Phys Med Biol, 2015, 60(18):7339-7357. [11] Jenkins EPW, Finch A, Gerigk M, et al. Electrotherapies for glioblastoma[J]. Adv Sci (Weinh), 2021, 8(18):e2100978. [12] Luo CK, Xu SC, Dai G, et al. Tumor treating fields for high-grade gliomas[J]. Biomedecine Pharmacother, 2020, 127:110193. [13] Gera N, Yang A, Holtzman TS, et al. Tumor treating fields perturb the localization of septins and cause aberrant mitotic exit[J]. PLoS One, 2015, 10(5):e0125269. [14] Giladi M, Schneiderman RS, Porat Y, et al. Mitotic disruption and reduced clonogenicity of pancreatic cancer cells in vitro and in vivo by tumor treating fields[J]. Pancreatology, 2014, 14(1):54-63. doi: 10.1016/j.pan.2013.11.009 [15] Stupp R, Wong ET, Kanner AA, et al. NovoTTF-100A versus physician's choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality[J]. Eur J Cancer, 2012, 48(14):192-202. -



 下载:
下载:


