Effect of intraperitoneal perfusion in patients with advanced gastric cancer peritoneal metastasis
-
摘要:
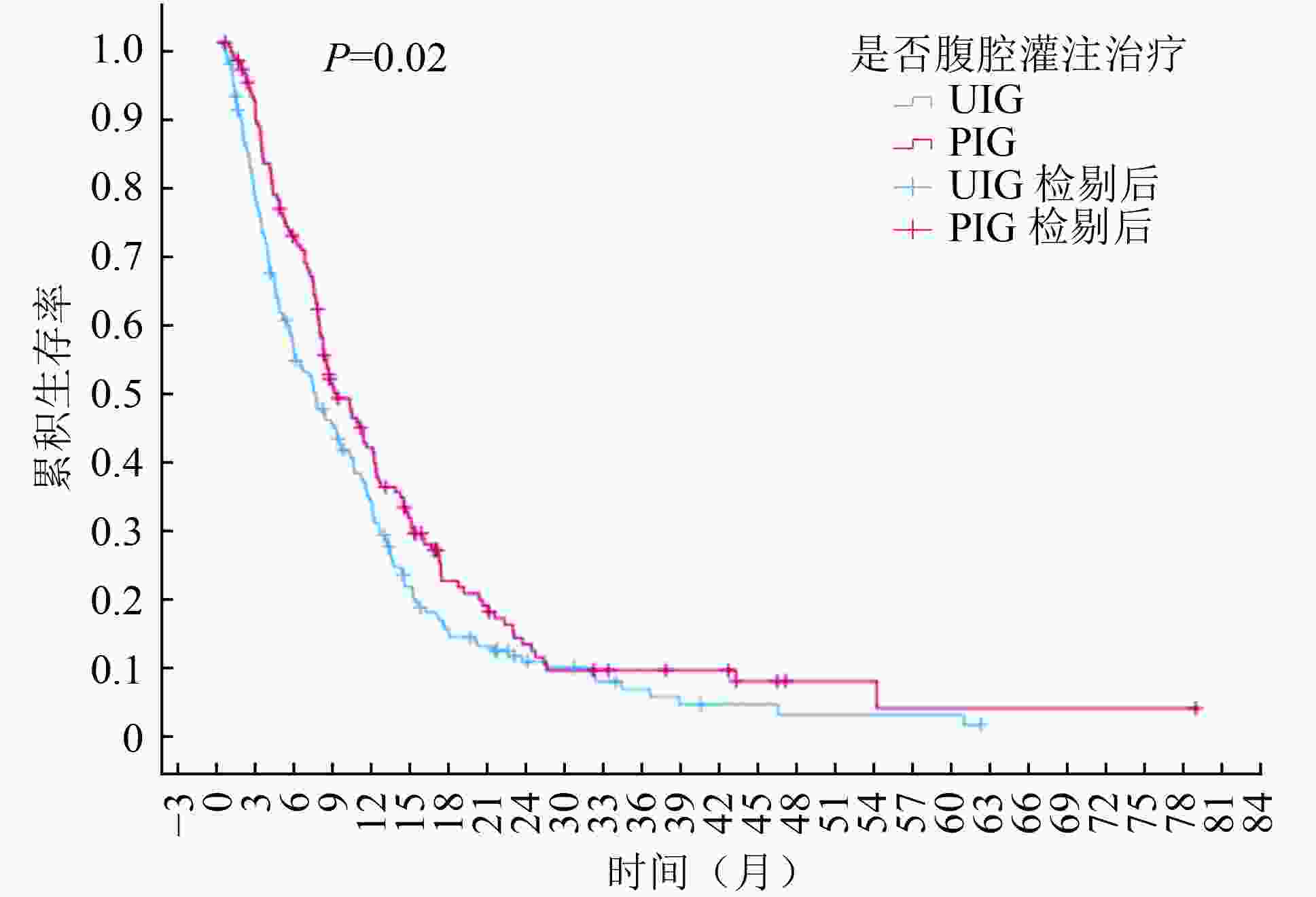
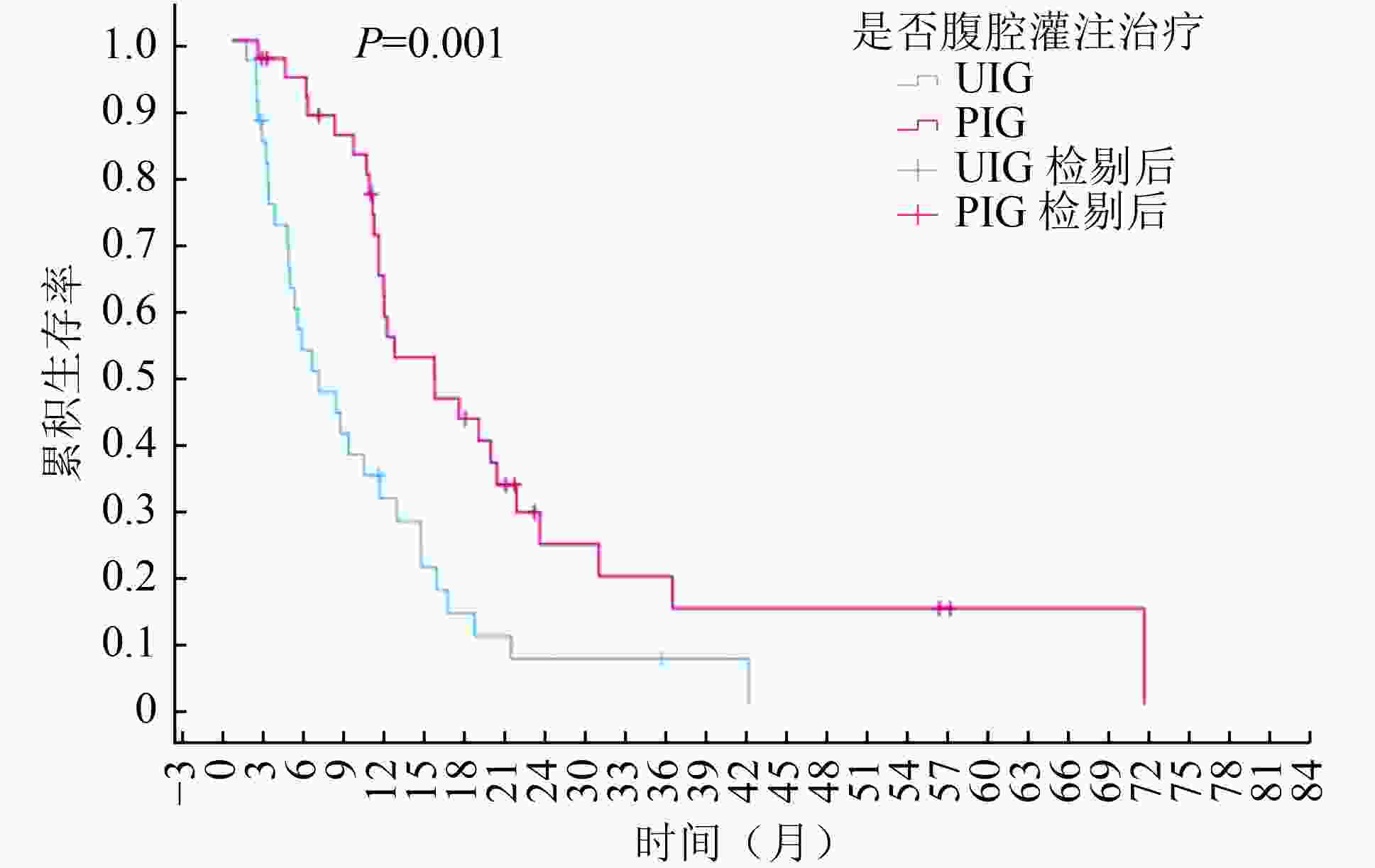
目的 探讨腹腔灌注治疗对胃癌腹膜转移患者生存的影响。 方法 回顾性分析2014年1月至2020年12月哈尔滨医科大学附属肿瘤医院收治的353例晚期胃癌合并腹膜转移患者临床资料。根据是否腹腔灌注给药分为灌注治疗组和未灌注治疗组,并对患者进行生存随访。Kaplan-Meier 法绘制生存曲线,Cox风险比例模型分析影响患者预后的因素,根据病灶部位、分化程度等进行亚组分析。 结果 灌注治疗组和未灌注治疗组中位总生存(overall survival,OS)期为9.50个月(95%CI:7.39~11.61)和7.83个月(95%CI:5.74~9.92,P=0.022)。体质量指数(BMI)>25及伴有腹水的胃癌腹膜转移患者更易从腹腔灌注中获益。多因素分析显示:腹腔积液(HR=1.52,95%CI:1.18~1.94)、全身系统治疗(HR=0.43,95%CI:0.33~0.56)、手术(HR=0.52,95%CI:0.39~0.70)、腹腔灌注治疗(HR=0.65,95%CI:0.52~0.82)是影响患者生存的因素。 结论 胃癌伴有腹膜转移患者可进行腹腔灌注治疗,与未灌注治疗患者相比,其生存期明显延长。Cox多因素分析显示无腹水、接受手术治疗、接受全身系统治疗和腹腔灌注治疗的患者预后更佳。分层分析显示BMI>25及伴有腹水的胃癌腹膜转移患者更易从腹腔灌注治疗中获益。 Abstract:Objective To investigate the effect of intraperitoneal perfusion on the survival of patients with gastric cancer peritoneal metastasis. Methods The clinical data of 353 patients with advanced gastric cancer and peritoneal metastasis admitted to the Cancer Hospital Affiliated to Harbin Medical University from January 2014 to December 2020 were analyzed retrospectively. The patients were assigned into intraperitoneal perfusion group and non-intraperitoneal perfusion group, based on the application of the intraperitoneal perfusion. The patients underwent regular follow-up. The survival curve was constructed via the Kaplan-Meier method. The Cox risk ratio model analysis was used to determine the effect on patient prognosis, and the sub-group analysis was performed based on the lesion's location and degree of differentiation. Results The median overall survival time of intraperitoneal perfusion group and non-intraperitoneal perfusion group was 9.50 months (95% confidence interval [CI]:7.39-11.61) and 7.83 months (95%CI:5.74-9.92), P= 0.022. Patients with a BMI>25 and abdominal metastasis with ascites benefitted significantly from intraperitoneal perfusion. Based on the multi-factor analysis, abdominal effusion (hazard ratio [HR]=1.52, 95%CI: 1.18-1.94), systemic treatment (HR=0.43, 95%CI: 0.33-0.56), surgical treatment (HR=0.52, 95%CI: 0.39-0.70), and intraperitoneal perfusion treatment (HR=0.65, 95%CI:0.52-0.82) significantly affected the survival of patients. Conclusions Patients with gastric cancer and peritoneal metastasis were effectively treated with intraperitoneal perfusion. Compared with non-intraperitoneal perfusion group, patients treated via intraperitoneal perfusion had a significantly prolonged survival period. Cox multi-factor analysis showed that patients with ascites, surgical treatment, systemic treatment, and intraperitoneal perfusion had more favorable outcomes. The layout analysis showed that patients with gastric cancer and peritoneal metastasis benefitted from intraperitoneal perfusion. -
表 1 患者一般情况
变量 PIG组(n=158) UIG组(n=195) P 性别 0.620 男 103(65.2) 132(67.7) 女 55(34.8) 63(32.3) BMI 0.505 <18.5 23(14.6) 33(16.9) 18.5~25.0 102(64.6) 125(64.1) >25 33(20.8) 37(19.0) 年龄(岁) 0.141 <60 95(60.1) 102(52.3) ≥60 63(39.9) 93(47.7) 腹水 0.014 无或少量 54(34.2) 92(47.2) 中量或大量 104(65.8) 103(52.8) 其他转移 0.533 无 101(63.9) 109(55.9) 有 57(36.1) 86(44.1) 病理类型 0.509 情况不明 31(19.6) 40(20.5) 黏液腺癌 52(32.9) 74(38.0) 单纯腺癌 75(47.5) 81(41.5) 病灶部位 0.542 全胃 6(3.8) 7(3.6) 胃底贲门 19(12.0) 27(13.9) 胃体 62(39.3) 88(45.1) 胃窦 71(44.9) 73(37.4) 全身治疗 0.550 是 103(65.2) 133(68.2) 否 55(34.8) 62(31.8) 手术治疗 0.412 是 30(19.0) 44(22.6) 否 128(81.0) 151(77.4) 放疗 0.109 是 3(1.9) 10(5.1) 否 155(98.1) 185(94.9) ( )内单位为% 表 2 患者全身治疗方案
一线方案 PIG组 UIG组 例数(例) 平均治疗周期数(个) 有后线治疗例数(例) 例数(例) 平均治疗周期数(个) 有后线治疗例数(例) 紫杉醇/多西他赛+替吉奥/卡培他滨 21 4.57 13 29 4.00 15 紫杉醇/多西他赛+SOX 2 4.50 2 4 3.75 1 替吉奥 15 1.67 3 17 1.50 5 XELOX/SOX 65 4.06 20 80 3.70 28 多西他赛+顺铂/洛铂 0 0 0 3 2.00 1 总计 103 38 133 50 表 3 灌注治疗的方案
灌注治疗方案 例数(例) 周期数(个) 紫杉醇热灌注 88 1.00 紫杉醇+洛铂 2 1.00 顺铂+氟尿嘧啶 5 2.20 顺铂 13 2.62 洛铂 11 1.18 氟尿嘧啶 31 1.00 贝伐+紫杉醇 2 3.00 贝伐 2 1.50 氟尿嘧啶+洛铂 4 1.75 表 4 患者生存的单因素分析
变量 例数(%) 中位OS(月) 95%CI P 性别 0.890 男 235(66.6) 8.77 7.07~10.47 女 118(33.4) 8.70 7.22~10.12 BMI 0.941 <18.5 56(15.9) 8.77 4.06~13.48 18.5~25.0 227(64.3) 8.57 6.76~10.38 >25.0 70(19.8) 8.63 7.72~9.54 年龄(岁) 0.454 <60 197(55.8) 8.87 7.14~10.60 ≥60 156(44.2) 8.30 6.92~9.68 腹水 0.375 无 146(41.4) 9.10 6.97~11.23 有 207(58.6) 8.57 7.31~9.83 其他转移 0.625 无 210(59.5) 8.57 7.50~9.64 有 143(40.5) 9.40 7.14~11.66 病理类型 0.232 未知 71(20.1) 9.90 7.58~12.22 黏液腺癌 126(35.7) 8.00 6.26~9.74 单纯腺癌 156(44.2) 9.07 7.07~11.07 病灶部位 0.153 全胃 13(3.7) 9.50 3.65~15.35 胃底贲门 46(13.0) 12.00 7.31~16.69 胃体 150(42.5) 7.80 6.51~9.09 胃窦 144(40.8) 9.70 7.52~11.88 全身治疗 <0.001 是 236(66.9) 11.50 9.83~13.17 否 117(33.1) 5.40 4.00~6.74 手术 <0.001 是 74(21.0) 15.13 11.42~18.85 否 279(79.0) 8.00 7.11~8.89 放疗 0.184 是 13(3.7) 12.70 9.85~15.55 否 340(96.3) 8.57 7.63~9.51 腹腔灌注 0.022 是 158(44.8) 9.50 7.39~11.61 否 195(55.2) 7.83 5.74~9.92 -
[1] Chen WQ, Zheng RS, Baade PD, et al. Cancer statistics in China, 2015[J]. CA A Cancer J Clin, 2016, 66(2):115-132. doi: 10.3322/caac.21338 [2] Thomassen I, van Gestel YR, van Ramshorst B, et al. Peritoneal carcinomatosis of gastric origin: a population-based study on incidence, survival and risk factors[J]. Int J Cancer, 2014, 134(3):622-628. doi: 10.1002/ijc.28373 [3] Wang FH, Shen L, Li J, et al. The chinese society of clinical oncology (CSCO): clinical guidelines for the diagnosis and treatment of gastric cancer[J]. Cancer Commun, 2019, 39(1):10. doi: 10.1186/s40880-019-0349-9 [4] Chan DY, Syn NL, Yap R, et al. Conversion surgery post-intraperitoneal paclitaxel and systemic chemotherapy for gastric cancer carcinomatosis peritonei. are we ready[J]? J Gastrointest Surg, 2017, 21(3):425-433. [5] Yamaguchi H, Kitayama J, Ishigami H, et al. A phase 2 trial of intravenous and intraperitoneal paclitaxel combined with S-1 for treatment of gastric cancer with macroscopic peritoneal metastasis[J]. Cancer, 2013, 119(18):3354-3358. doi: 10.1002/cncr.28204 [6] Ishigami H, Fujiwara Y, Fukushima R, et al. Phase Ⅲ trial comparing intraperitoneal and intravenous paclitaxel plus S-1 versus cisplatin plus S-1 in patients with gastric cancer with peritoneal metastasis: PHOENIX-GC trial[J]. J Clin Oncol, 2018, 36(19):1922-1929. doi: 10.1200/JCO.2018.77.8613 [7] Sarner M. Treatment of pancreatic exocrine deficiency[J]. World J Surg, 2003, 27(11):1192-1195. doi: 10.1007/s00268-003-7237-8 [8] de Bree E, Michelakis D, Stamatiou D, et al. Pharmacological principles of intraperitoneal and bidirectional chemotherapy[J]. Pleura Peritoneum, 2017, 2(2):47-62. doi: 10.1515/pp-2017-0010 [9] Kitayama J, Ishigami H, Yamaguchi H, et al. S-1 plus intravenous and intraperitoneal Paclitaxel for gastric cancer with peritoneal metastasis[J]. Gastrointest Cancer Res, 2012, 5(suppl_3): S10-S13. [10] Shitara K, Mizota A, Matsuo K, et al. Fluoropyrimidine plus cisplatin for patients with advanced or recurrent gastric cancer with peritoneal metastasis[J]. Gastric Cancer, 2013, 16(1):48-55. doi: 10.1007/s10120-012-0143-8 [11] de Bree E, Theodoropoulos PA, Rosing H, et al. Treatment of ovarian cancer using intraperitoneal chemotherapy with taxanes: from laboratory bench to bedside[J]. Cancer Treat Rev, 2006, 32(6):471-482. doi: 10.1016/j.ctrv.2006.07.006 [12] Kitayama J, Ishigami H, Yamaguchi H, et al. Intraperitoneal paclitaxel is useful as adjuvant chemotherapy for advanced gastric cancer with serosal exposure[J]. Case Rep Oncol, 2014, 7(1):58-64. doi: 10.1159/000358379 -




 下载:
下载: