Effects of LINC01234 on proliferation, invasion, and migration of acute myeloid leukemia cells by regulating IGF2BP1/c-Myc
-
摘要:
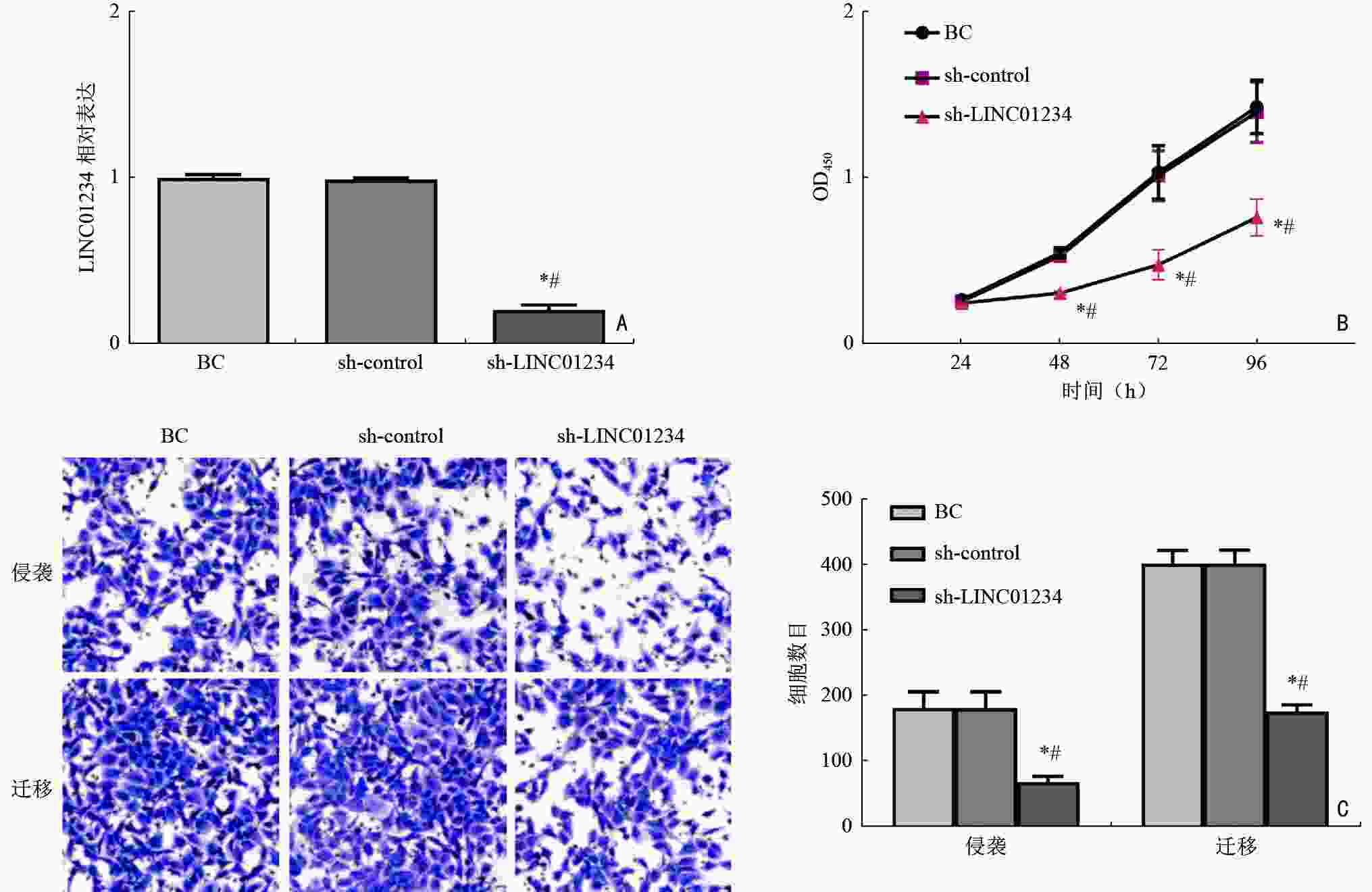
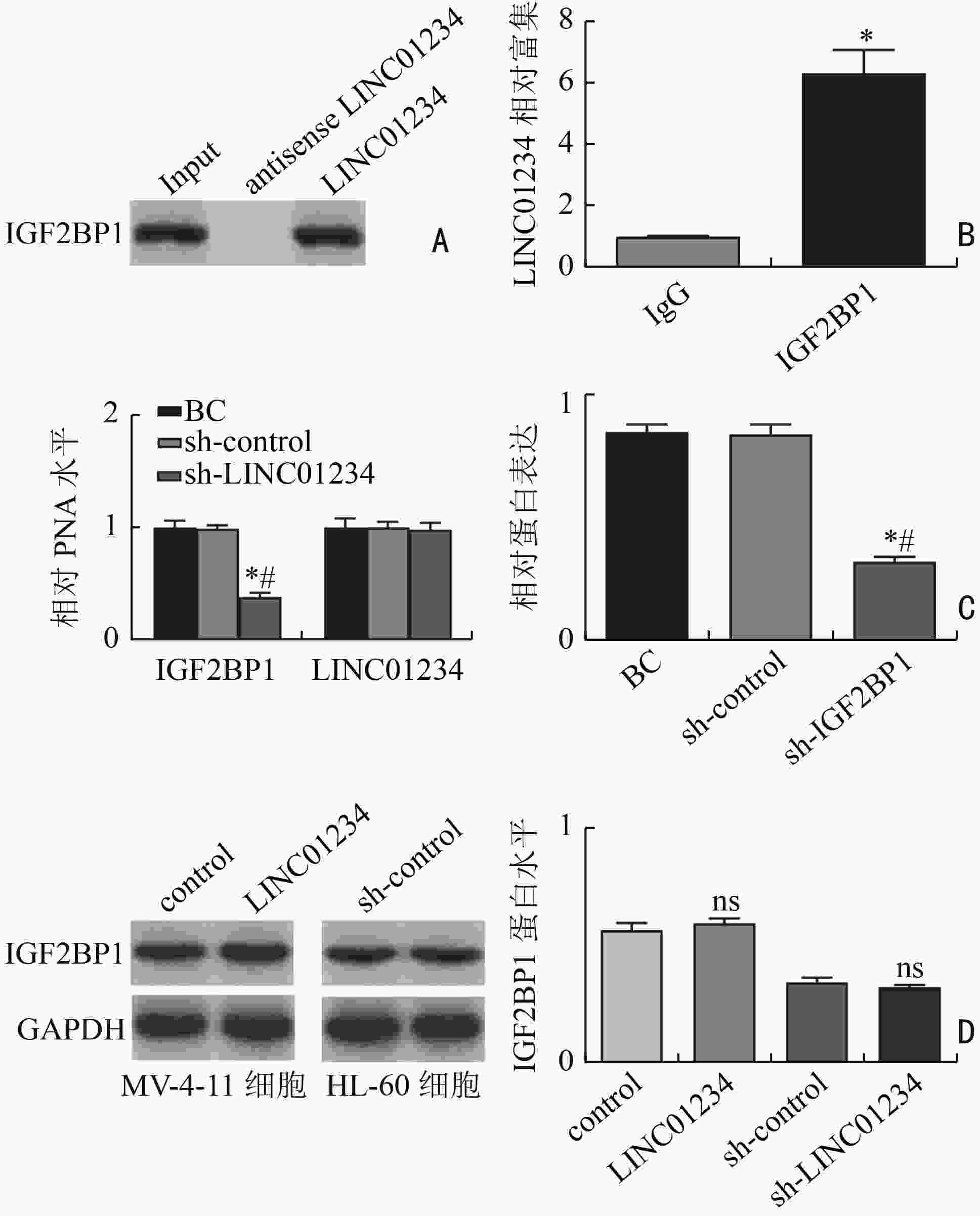
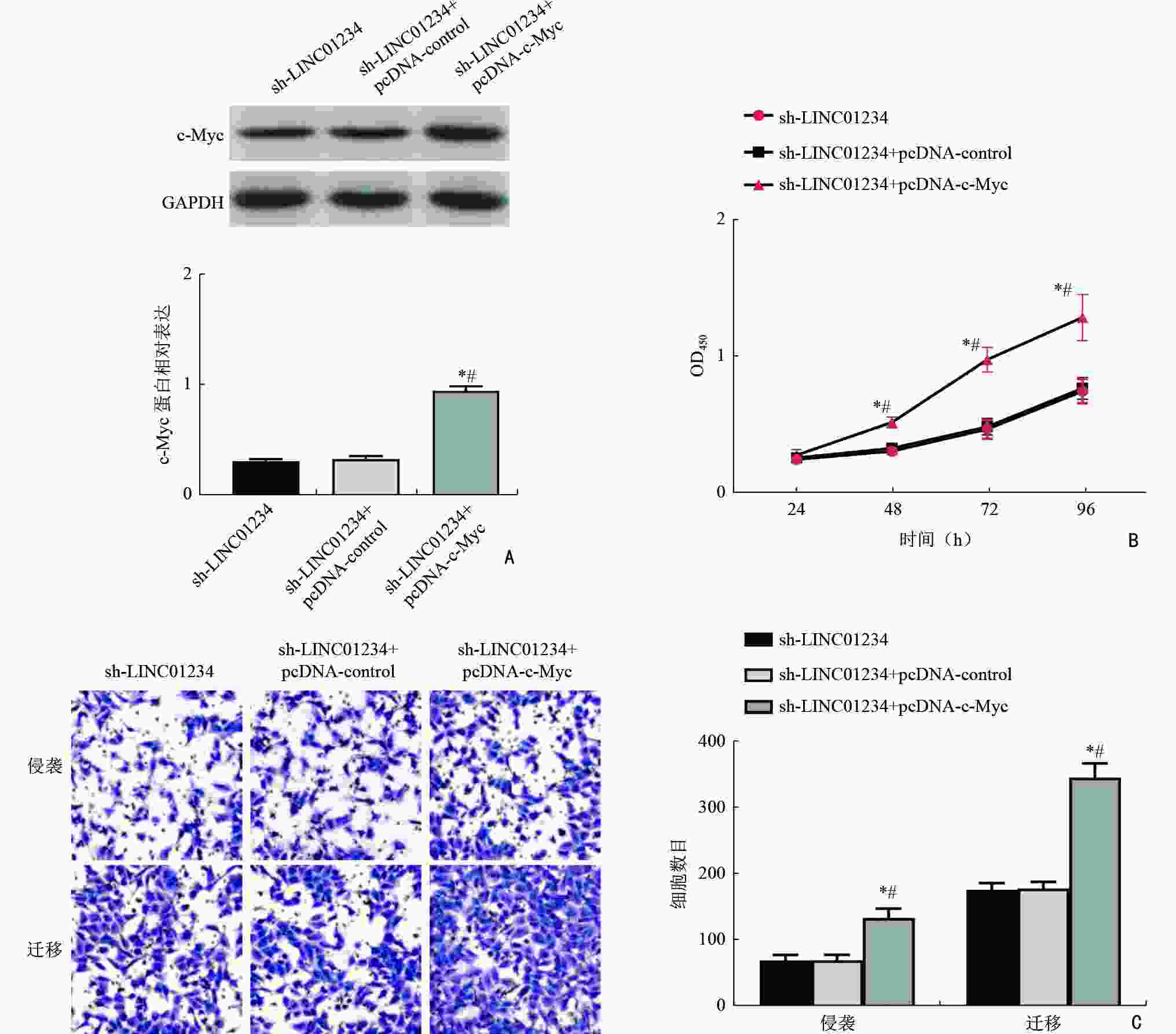
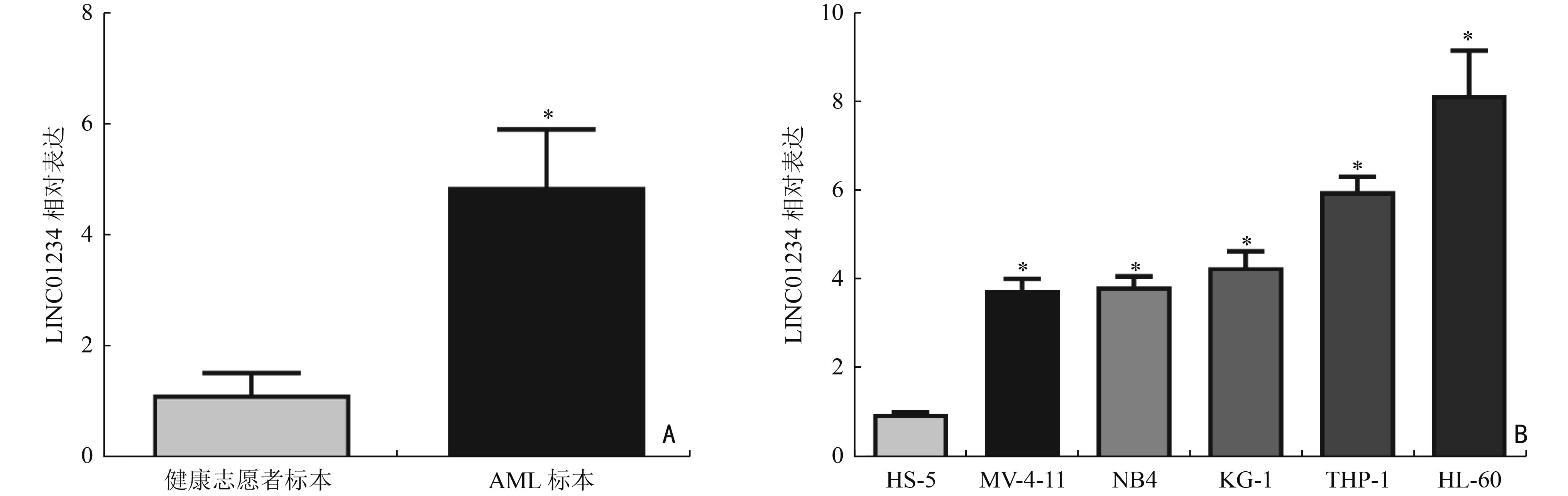
目的 探讨LINC01234通过调控胰岛素样生长因子2 mRNA结合蛋白1(insulin-like growth factor 2 mRNA binding protein 1,IGF2BP1)/c-Myc对急性髓系白血病(acute myeloid leukemia,AML)细胞增殖、侵袭、迁移的影响。 方法 分析2019年3月至2021年9月在湖北省恩施州中心医院确诊的31例AML患者和在本院体检的24例健康志愿者的外周血标本。通过实时定量PCR(real-time quantitative PCR,qRT-PCR)检测分析LINC01234在AML标本和细胞系(MV-4-11、NB4、KG-1、THP-1、HL-60)中的表达模式。HL-60细胞随机记为空白对照(blank control,BC)组、sh-control组、sh-LINC01234组、sh-LINC01234+pcDNA-control组、 sh-LINC01234+pcDNA-c-Myc组。qRT-PCR检测细胞中LINC01234、IGF2BP1和c-Myc的表达。细胞计数试剂盒-8实验、transwell迁移和侵袭实验用于细胞增殖能力、细胞侵袭和迁移功能的研究。通过RNA免疫沉淀和RNA pull-down实验证实LINC01234、IGF2BP1和c-Myc在AML细胞中的调节相关性。 结果 与健康志愿者标本和人骨髓基质细胞HS-5相比,LINC01234在AML标本和5种细胞系中表达均升高(P<0.05)。sh-LINC01234下调HL-60细胞中LINC01234水平后,OD值显著降低,侵袭、迁移细胞数目显著减少(P<0.05)。LINC01234结合IGF2BP1,促进IGF2BP1与c-Myc mRNA的相互作用,从而促进c-Myc mRNA的稳定性(P<0.05)。c-Myc过表达逆转了LINC01234沉默对HL-60细胞增殖、转移的阻碍作用(P<0.05)。 结论 LINC01234是一种新型AML相关的lncRNA,通过竞争性结合IGF2BP1促进c-Myc mRNA的稳定性,进而促进AML细胞增殖、侵袭和迁移。 Abstract:Objective To investigate the impact of LINC01234 on the proliferation, invasion, and migration of acute myeloid leukemia (AML) cells by regulating insulin-like growth factor 2 mRNA binding protein 1 (IGF2BP1)/c-Myc expression. Methods Peripheral blood samples were collected from 31 AML patients and 24 healthy volunteers who underwent physical examination at Enshi Center Hospital from March 2019 to September 2021. The expression pattern of LINC01234 in AML specimens and five cell lines, MV-4-11, NB4, KG-1, THP-1, and HL-60, was analyzed using real-time quantitative PCR (qRT-PCR). HL-60 cells were randomly selected for use in preparing the blank control (BC) group, sh-control group, sh-LINC01234 group, sh-LINC01234+pcDNA-control group, and sh-LINC01234 +pcDNA-c-Myc group. qRT-PCR was performed to measure the expression of LINC01234, IGF2BP1, and c-Myc in the cells. Cell counting kit-8 assay and transwell migration and invasion assay were performed to assess cell proliferation ability and cell migration and invasion, respectively. RNA immunoprecipitation and RNA pull-down experiments were performed to confirm the regulatory relevance of LINC01234, IGF2BP1, and c-Myc in AML cells. Results LINC01234 expression was higher in AML specimens and the five cell lines than in specimens from healthy volunteers and human bone marrow stromal cells HS-5 (P<0.05). After LINC01234 expression was downregulated by sh-LINC01234 in HL-60 cells, the OD value and the numbers of invasive and migrating cells were significantly reduced (P<0.05). LINC01234 bound to IGF2BP1 and promoted the interaction between IGF2BP1 and c-Myc mRNA, thereby improving the stability of c-Myc mRNA (P<0.05). Overexpression of c-Myc reversed the decrease in proliferation and metastasis of HL-60 cells caused by LINC01234 silencing (P<0.05). Conclusions LINC01234 is a novel AML-related lncRNA that promotes the stability of c-Myc mRNA by competitively binding to IGF2BP1, thereby promoting proliferation, invasion, and migration of AML cells. -
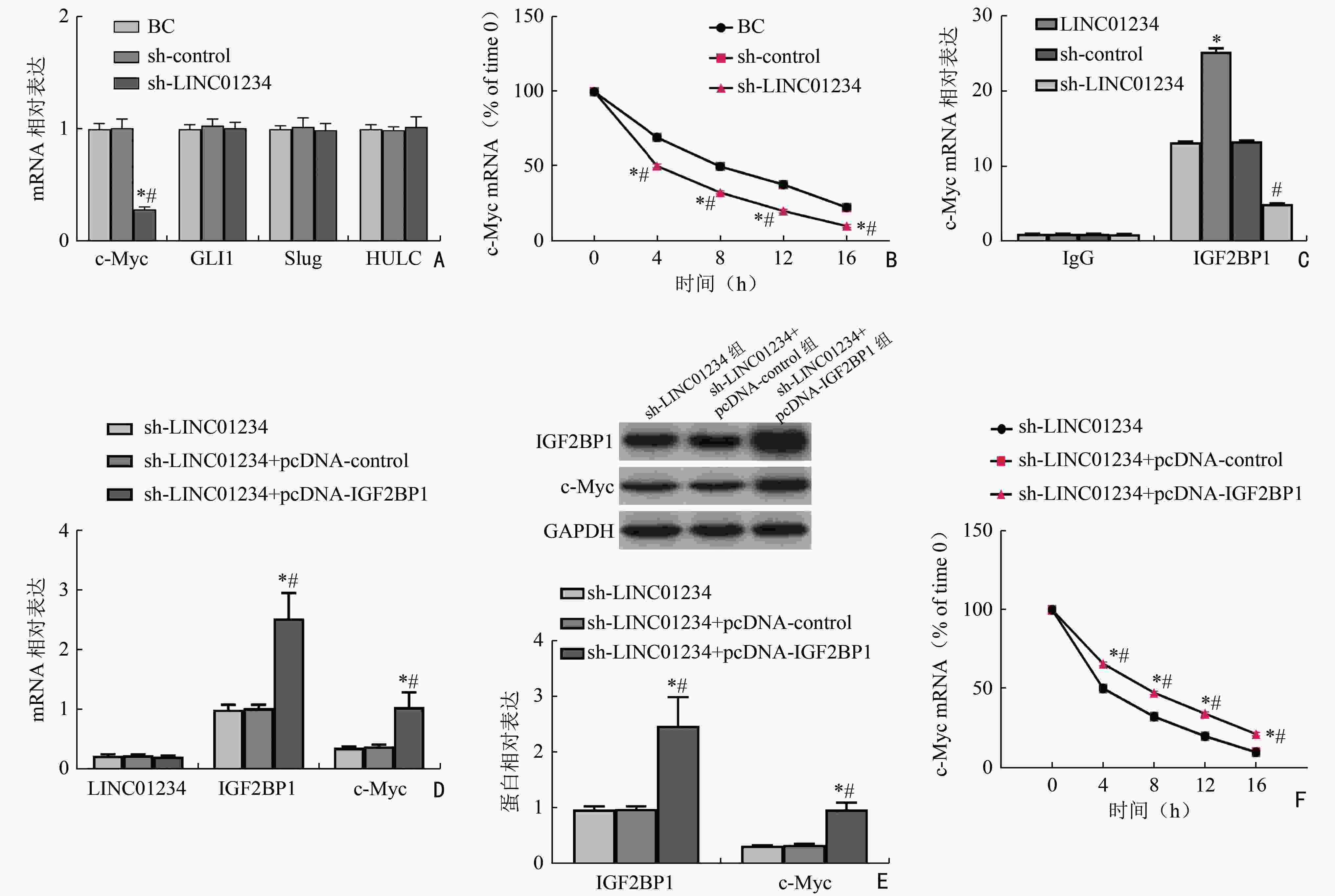
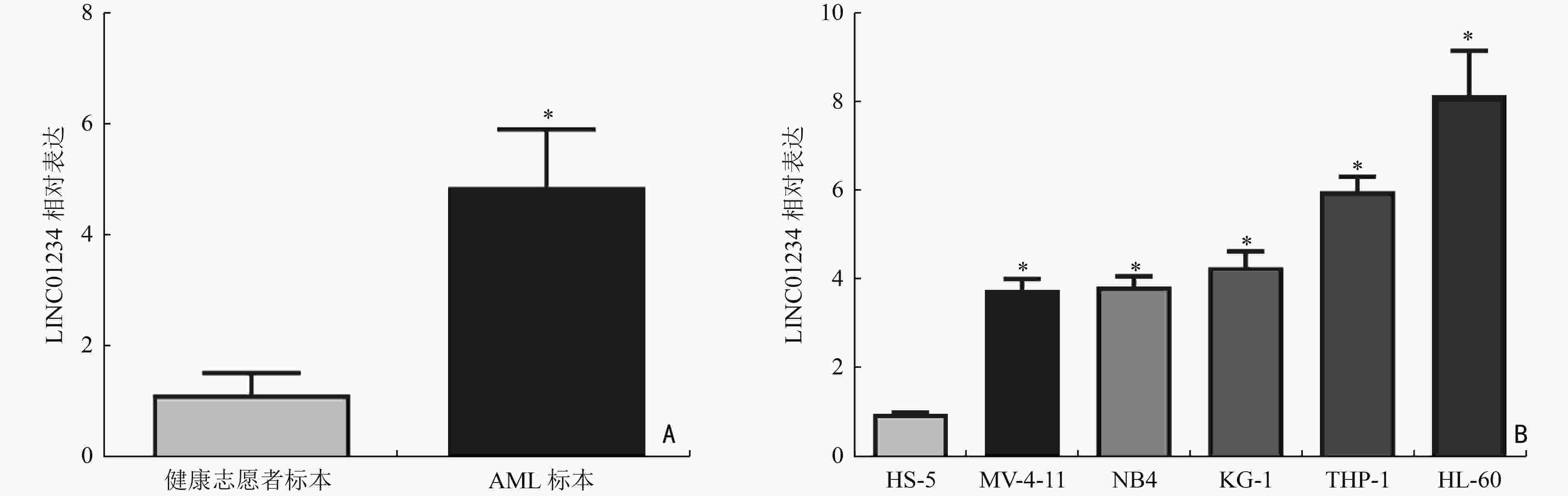
图 4 LINC01234通过竞争性结合IGF2BP1促进c-Myc mRNA的稳定性
A:LINC01234沉默对HL-60细胞中IGF2BP1的靶mRNA的影响;B:LINC01234沉默对放线菌素D处理后的HL-60细胞中c-Myc mRNA表达的影响;C:IGF2BP1抗体处理后的HL-60细胞中c-Myc mRNA表达的影响;D:各组细胞中LINC01234、IGF2BP1、c-Myc mRNA表达;E:各组细胞中IGF2BP1、c-Myc蛋白表达;F:各组细胞中c-Myc mRNA稳定性比较;*:与BC组或相比,P<0.05;与sh-control组相比,#:P<0.05;与control组相比,*:P<0.05;与sh-control组相比,#:P<0.05;与sh-LINC01234组相比,*:P<0.05;与sh-LINC01234+pcDNA-control组相比,#:P<0.05
-
[1] Bullinger L, Döhner K, Döhner H. Genomics of acute myeloid leukemia diagnosis and pathways[J]. J Clin Oncol, 2017, 35(9):934-946. doi: 10.1200/JCO.2016.71.2208 [2] Bose P, Vachhani P, Cortes JE. Treatment of relapsed/refractory acute myeloid leukemia[J]. Curr Treat Options Oncol, 2017, 18(3):17-47. [3] Döhner H, Weisdorf DJ, Bloomfield CD. Acute myeloid leukemia[J]. N Engl J Med, 2015, 373(12):1136-1152. doi: 10.1056/NEJMra1406184 [4] Huarte M. The emerging role of lncRNAs in cancer[J]. Nat Med, 2015, 21(11):1253-1261. doi: 10.1038/nm.3981 [5] Bhan A, Soleimani M, Mandal SS. Long noncoding RNA and cancer: a new paradigm[J]. Cancer Res, 2017, 77(15):3965-3981. doi: 10.1158/0008-5472.CAN-16-2634 [6] Li L, Wan D, Li L, et al. LncRNA RAET1K promotes the progression of acute myeloid leukemia by targeting miR-503-5p/INPP4B Axis[J]. Onco Targets Ther, 2021, 14(1):531-544. [7] Peng WX, Koirala P, Mo YY. LncRNA-mediated regulation of cell signaling in cancer[J]. Oncogene, 2017, 36(41):5661-5667. doi: 10.1038/onc.2017.184 [8] Fan CN, Ma L, Liu N. Systematic analysis of lncRNA-miRNA-mRNA competing endogenous RNA network identifies four-lncRNA signature as a prognostic biomarker for breast cancer[J]. J Transl Med, 2018, 16(1):264. doi: 10.1186/s12967-018-1640-2 [9] Wang Y, Zhou Q, Ma JJ. High expression of lnc-CRNDE presents as a biomarker for acute myeloid leukemia and promotes the malignant progression in acute myeloid leukemia cell line U937[J]. Eur Rev Med Pharmacol Sci, 2018, 22(3):763-770. [10] Bi M, Zheng L, Chen L, et al. ln RNA LINC01234 promotes triple-negative breast cancer progression through regulating the miR-429/SYNJ1 axis[J]. Am J Transl Res, 2021, 13(10):11399-11412. [11] Chen Y, Zhao H, Li H, et al. LINC01234/MicroRNA-31-5p/MAGEA3 axis mediates the proliferation and chemoresistance of hepatocellular carcinoma cells[J]. Mol Ther Nucleic Acids, 2020, 19(1):168-178. [12] Liao X, Zhan W, Zhang J, et al. Long noncoding RNA LINC01234 promoted cell proliferation and invasion via miR-1284/TRAF6 axis in colorectal cancer[J]. J Cell Biochem, 2020, 121(10):4295-4309. doi: 10.1002/jcb.29618 [13] Kadia TM, Ravandi F, O'Brien S, et al. Progress in acute myeloid leukemia[J]. Clin Lymph Myelo Leuk, 2015, 15(3):139-151. doi: 10.1016/j.clml.2014.08.006 [14] Kantarjian H. Acute myeloid leukemia-major progress over four decades and glimpses into the future[J]. Am J Hematol, 2016, 91(1):131-145. doi: 10.1002/ajh.24246 [15] Xiao F, Jia H, Wu D, et al. LINC01234 aggravates cell growth and migration of triple-negative breast cancer by activating the Wnt pathway[J]. Environ Toxicol, 2021, 36(10):1999-2012. doi: 10.1002/tox.23318 [16] Liu D, Jian X, Xu P, et al. Linc01234 promotes cell proliferation and metastasis in oral squamous cell carcinoma via miR-433/PAK4 axis[J]. BMC Cancer, 2020, 20(1):107-117. doi: 10.1186/s12885-020-6541-0 [17] Zhu P, He F, Hou Y, et al. A novel hypoxic long noncoding RNA KB-1980E6.3 maintains breast cancer stem cell stemness via interacting with IGF2BP1 to facilitate c-Myc mRNA stability[J]. Oncogene, 2021, 40(9):1609-1627. doi: 10.1038/s41388-020-01638-9 [18] He J, Zuo Q, Hu B, et al. A novel, liver-specific long noncoding RNA LINC01093 suppresses HCC progression by interaction with IGF2BP1 to facilitate decay of GLI1 mRNA[J]. Cancer Lett, 2019, 450(1):98-109. [19] Zirkel A, Lederer M, Stöhr N, et al. IGF2BP1 promotes mesenchymal cell properties and migration of tumor-derived cells by enhancing the expression of LEF1 and SNAI2 (SLUG)[J]. Nucleic Acids Res, 2013, 41(13):6618-6636. doi: 10.1093/nar/gkt410 [20] Hämmerle M, Gutschner T, Uckelmann H, et al. Posttranscriptional destabilization of the liver-specific long noncoding RNA HULC by the IGF2 mRNA-binding protein 1 (IGF2BP1)[J]. Hepatology, 2013, 58(5):1703-1712. doi: 10.1002/hep.26537 [21] Shang Y. LncRNA THOR acts as a retinoblastoma promoter through enhancing the combination of c-myc mRNA and IGF2BP1 protein[J].Biomed Pharmacother, 2018, 106(1):1243-1249. [22] McMahon SB. MYC and the control of apoptosis[J]. Cold Spring Harb Perspect Med, 2014, 4(7):a014407-a014416. doi: 10.1101/cshperspect.a014407 [23] Meristoudis C, Trangas T, Lambrianidou A, et al. Systematic analysis of the contribution of c-myc mRNA constituents upon cap and IRES mediated translation[J]. Biol Chem, 2015, 396(12):1301-1313. doi: 10.1515/hsz-2015-0190 [24] Lambrianidou A, Sereti E, Soupsana K, et al. mTORC2 deploys the mRNA binding protein IGF2BP1 to regulate c-MYC expression and promote cell survival[J]. Cell Signal, 2021, 80(1):109912-109925. [25] Chen Z, Chen X, Lei T, et al. Integrative analysis of NSCLC identifies LINC01234 as an oncogenic lncRNA that interacts with HNRNPA2B1 and regulates miR-106b biogenesis[J]. Mol Ther, 2020, 28(6):1479-1493. doi: 10.1016/j.ymthe.2020.03.010 -



 下载:
下载: