Clinical characteristics and prognosis of newly diagnosed diffuse large B-cell lymphoma with bulky mass
-
摘要:
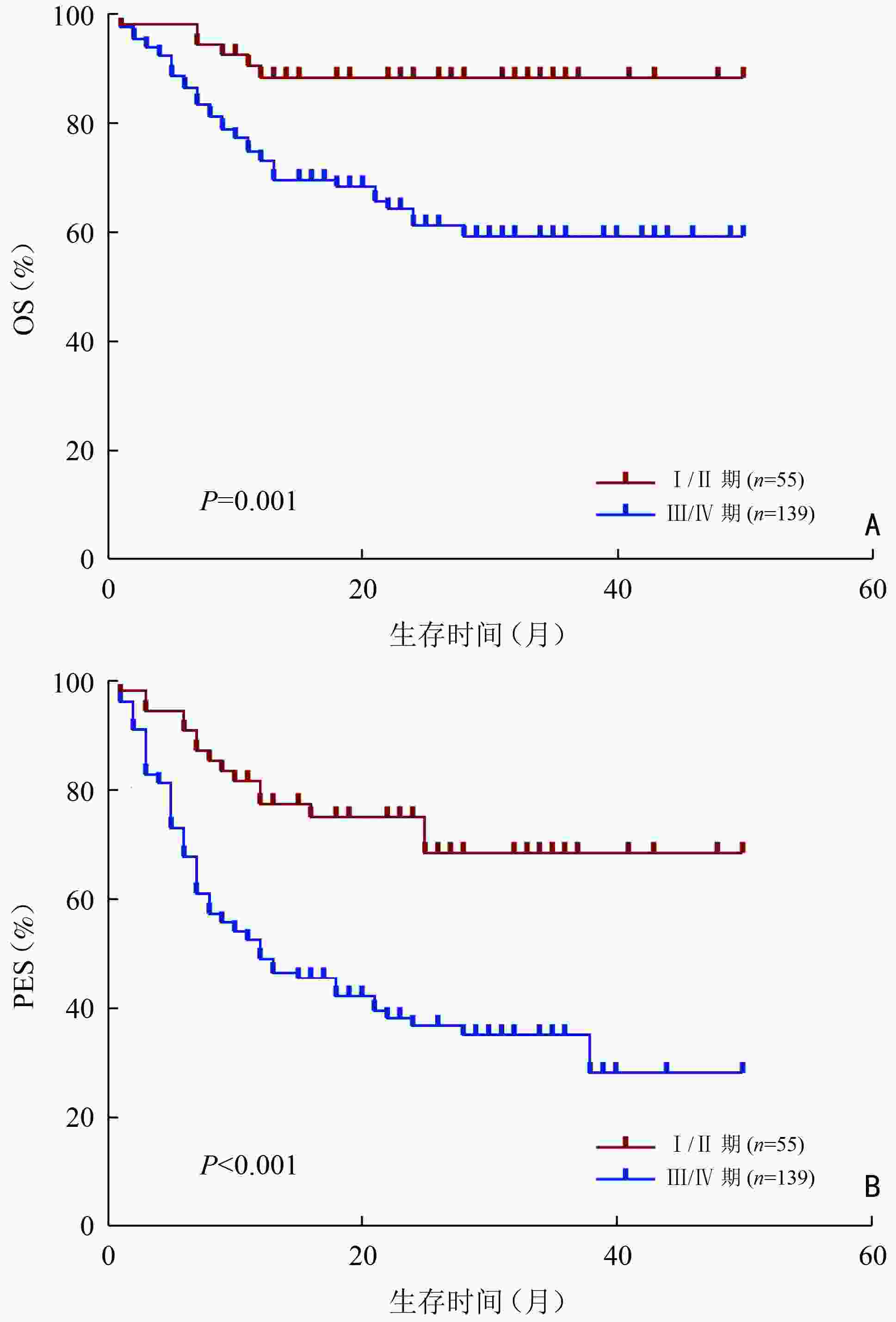
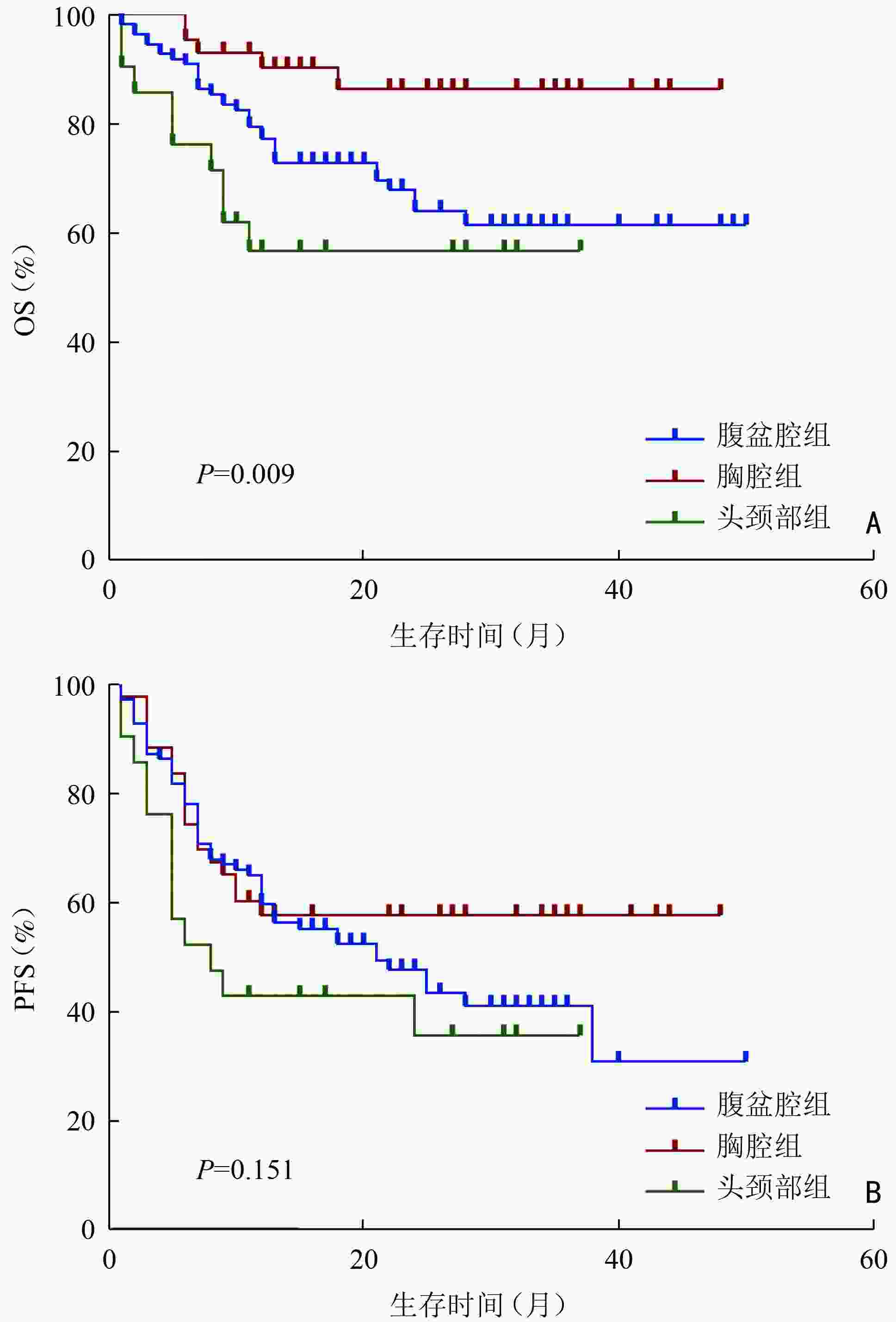
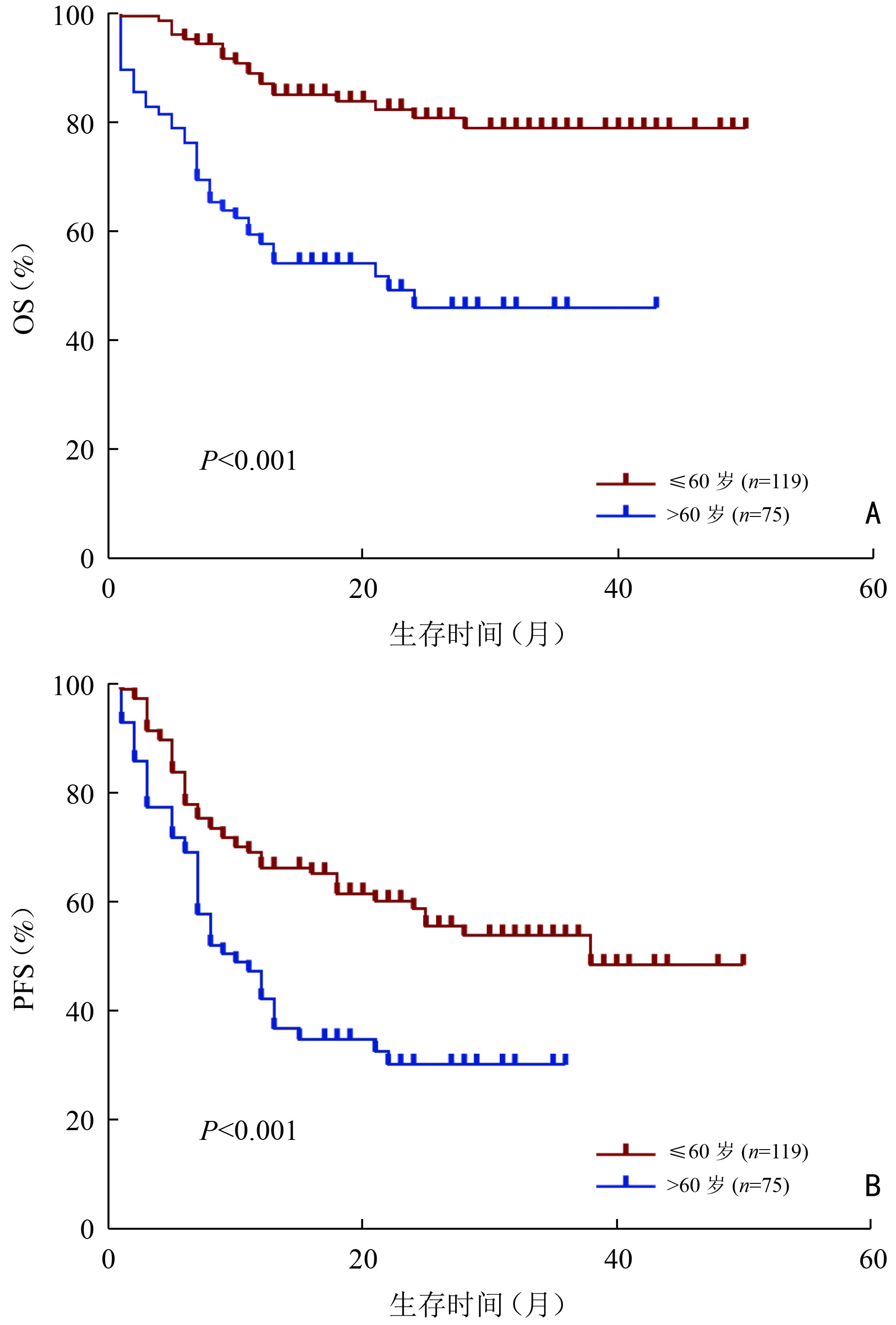
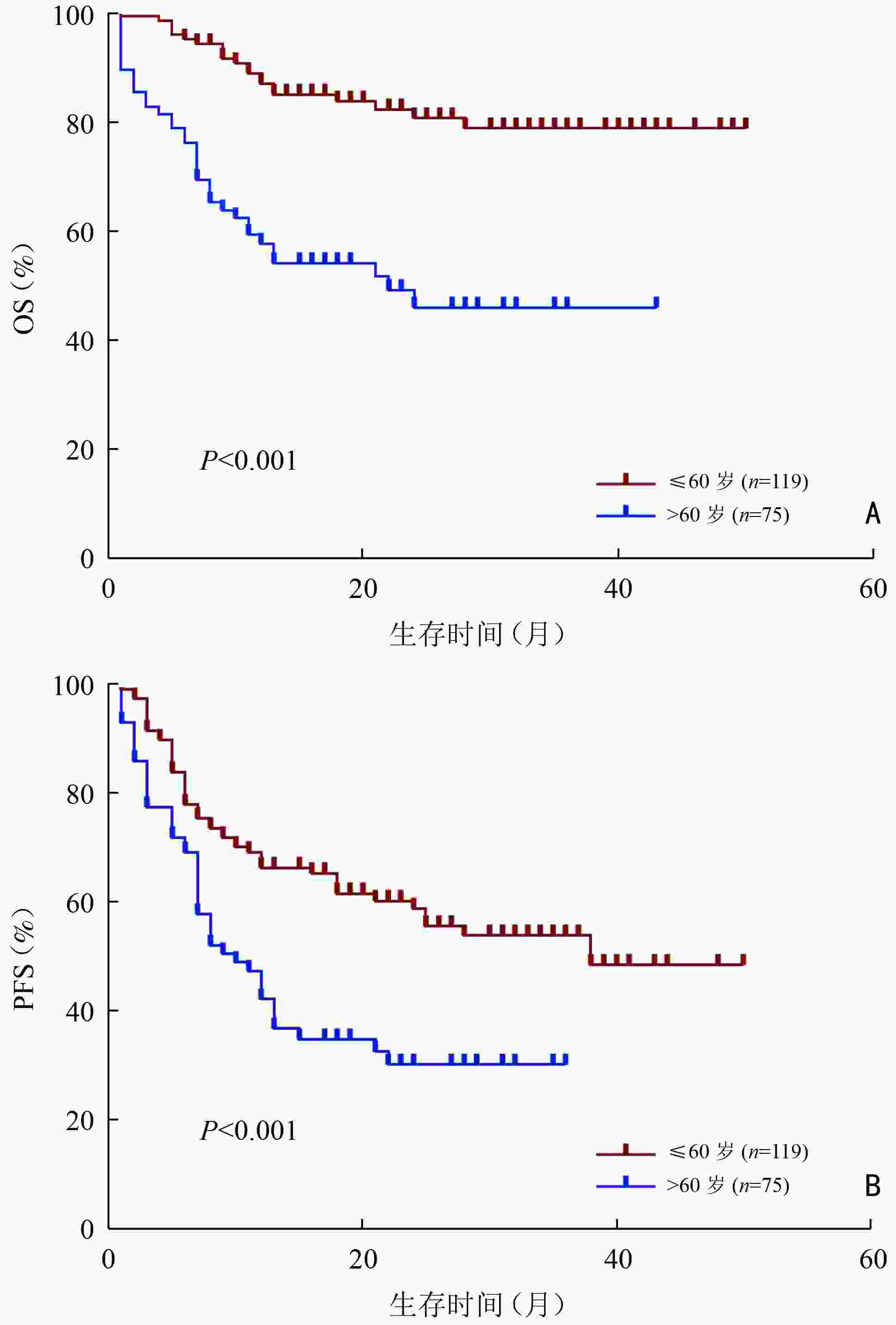
目的 分析初诊时伴有大肿块的弥漫性大B细胞淋巴瘤(diffuse large B-cell lymphoma,DLBCL)患者的临床特征与预后因素。 方法 回顾性分析2018年1月至2021年9月郑州大学第一附属医院收治的194例大肿块DLBCL患者的临床资料,其中大肿块定义为肿瘤最大直径(maximum tumor diameter,MTD)≥7.5 cm,根据大肿块所在部位将患者分为:腹盆腔组、胸腔组、头颈部组。 结果 194例大肿块DLBCL患者中位总生存(median overall survival,mOS)期为16(0~50)个月,患者1、3年总生存(overall survival,OS)率分别为75.6%、66.2%。1、3年无进展生存(progression-free survival,PFS)率分别为55.9%和44.1%。患者一线治疗后达到完全缓解(complete response, CR)、部分缓解(partial response,PR)、疾病稳定(stable disease,SD)、疾病进展(progressive disease,PD)分别为45例(23.2%)、78例(40.2%)、11例(5.7%)、60例(30.9%),一线治疗客观缓解率(overall response rate,ORR)为63.4%。单因素分析结果显示,年龄、Ann Arbor分期、β2-微球蛋白水平(β2-microglobulin,β2-MG)、有无中枢侵犯、是否放疗、大肿块所在部位是影响大肿块DLBCL患者OS的预后因素(P<0.05);Cox回归模型多因素分析结果显示,年龄>60岁、Ann Arbor分期Ⅲ/Ⅳ期、大肿块所在部位为腹盆腔组、头颈部组是大肿块DLBCL的独立不良预后因素(P<0.05)。腹盆腔组患者分期较晚、B症状多见、β2-微球蛋白水平多升高、C反应蛋白水平较高、Ki-67值较高;胸腔组年轻患者比例较高、临床分期较早、非GCB亚型更多见。腹盆腔组、胸腔组、头颈部组3组患者的3年OS率分别为60.4%、84.4%和54.2%,差异具有统计学意义(P<0.01)。 结论 大肿块DLBCL患者肿瘤负荷高,一线治疗缓解率低、疾病进展率高,年龄>60岁、临床分期晚、大肿块所在腹盆腔组和头颈部组的患者预后较差。 -
关键词:
- 弥漫性大B细胞淋巴瘤 /
- 大肿块 /
- 临床特征 /
- 预后分析 /
- 肿瘤部位
Abstract:Objective To analyze the clinical characteristics and prognostic factors of patients with diffuse large B-cell lymphoma (DLBCL) with bulky mass at initial diagnosis. Methods We retrospectively analyzed the clinical data of 194 patients with DLBCL with bulky mass admitted to The First Affiliated Hospital of Zhengzhou University from January 2018 to September 2021. Bulky mass was defined as a maximum tumor diameter (MTD) ≥7.5 cm. The patients were assigned into three groups, the abdominopelvic, thoracic cavity, and head and neck groups, according to the location of bulky mass. Results The median overall survival (OS) of 194 patients with DLBCL with bulky mass was 16 (range, 0–50) months, while the 1-year and 3-year OS rates were 75.6% and 66.2%, respectively. The 1-year and 3-year progression-free survival (PFS) rates were 55.9% and 44.1%, respectively. Following the first-line treatment, complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) were achieved in 45 (23.2%), 78(40.2%), 11 (5.7%) and 60 cases (30.9%), respectively. The objective response rate (ORR) of the first-line treatment was 63.4%. Univariate analysis revealed that the prognostic factors affecting OS included age, Ann Arbor stage, β2-microglobulin (β2-MG), central invasion, radiotherapy, and bulky mass location (P<0.05). Multivariate analysis using Cox regression model revealed that age >60 years, Ann Arbor stage Ⅲ/Ⅳ, bulky mass located in the abdominopelvic and head and neck regions were independent prognostic factors (P<0.05). Patients in the abdominopelvic group had a more advanced clinical stage, more symptoms of B, an increased β2-MG level, a higher C-reactive protein level, and a higher Ki-67 value, while the thoracic cavity group had a higher proportion of young patients, a less advanced clinical stage, and more non-GCB subtypes. The 3-year OS rates of patients in the abdominopelvic, thoracic cavity, and head and neck groups were 60.4%, 84.4% and 54.2%, respectively, with statistical differences (P<0.01). Conclusions Patients with DLBCL with bulky mass have a high tumor load, low remission rate, and high disease progression rate following the first-line treatment. Patients aged >60 years, those with an advanced clinical stage, and those with bulky mass in the abdominopelvic and head and neck regions have a poor prognosis. -
表 1 194例大肿块DLBCL患者总生存影响因素的单因素分析结果
因素 例数(例) 3年OS率(%) χ2 P 性别 0.349 0.555 男 112 67.90 女 82 63.90 年龄(岁) 26.129 <0.001 ≤60 119 78.70 >60 75 45.90 B症状 1.783 0.182 无 129 72.10 有 65 56.00 Ann Arbor分期(期) 12.035 0.001 Ⅰ/Ⅱ 55 88.40 Ⅲ/Ⅳ 139 57.20 LDH水平 0.171 0.679 正常 27 69.30 升高 163 67.60 β2-MG 11.838 0.001 正常 116 78.10 升高 74 50.20 CRP 2.079 0.149 正常 52 76.40 升高 87 63.40 COO分型(型) 0.445 0.505 GCB 50 58.60 non-GCB 141 68.70 Ki-67 0.014 0.906 <80 75 66.60 ≥80 116 66.64 中枢侵犯 6.215 0.013 有 7 28.60 无 184 69.00 骨髓侵犯 2.967 0.085 有 21 52.90 无 163 70.90 治疗方案 1.102 0.294 RCHOP 149 76.80 REPOCH 45 23.20 手术 0.028 0.886 是 6 3.10 否 188 96.90 放疗 4.495 0.034 是 26 13.40 否 168 86.60 合并症 3.063 0.080 有 53 27.30 无 141 72.70 大肿块直径(cm) 0.530 0.671 ≤10 87 67.90 >10 107 64.70 大肿块所在部位 9.496 0.009 腹盆腔组 112 60.40 胸腔组 44 84.80 头颈部组 22 54.20 其他 16 8.24 GCB:生发中心来源;non-GCB:非生发中心来源;LDH:乳酸脱氢酶;CRP:C反应蛋白 表 2 影响194例大肿块DLBCL患者总生存的多因素分析
因素 β χ2 HR 95%CI P 年龄 0.976 9.336 2.653 1.419~4.962 0.002 Ann Arbor分期 1.225 6.084 3.404 1.286~9.008 0.014 β2-MG水平 0.478 2.430 1.613 0.884~2.941 0.119 中枢侵犯 0.696 2.045 2.006 0.773~5.207 0.153 放疗 1.812 3.189 6.120 0.838~44.697 0.074 大肿块所在部位 腹盆腔组 −1.094 7.240 0.335 0.151~0.743 0.007 胸腔组 −1.282 5.038 0.278 0.091~0.850 0.025 头颈部组 — — 参照 参照 参照 表 3 不同部位分组大肿块DLBCL患者临床特征比较
项目 腹盆腔组 胸腔组 头颈部组 χ2 P (n=112例) (n=44例) (n=22例) 性别 3.693 0.158 男 63(56.3) 24(54.5) 15(68.2) 女 49(44.7) 20(45.5) 7(31.8) 年龄(岁) 17.069 <0.001 ≤60 61(54.5) 38(86.4) 12(54.5) >60 51(45.5) 6(13.6) 10(45.5) B症状 10.730 0.005 无 62(55.4) 34(77.3) 20(90.9) 有 50(44.6) 10(22.7) 2(9.1) Ann Arbor分期(期) 20.102 <0.001 Ⅰ/Ⅱ 21(18.8) 22(50.0) 9(40.9) Ⅲ/Ⅳ 91(81.2) 22(50.0) 13(59.1) LDH水平 0.249 0.883 正常 16(14.3) 6(13.6) 5(22.7) 升高 96(85.7) 36(81.8) 17(77.3) β2-MG水平 14.267 0.001 正常 57(50.9) 35(79.5) 17(77.3) 升高 53(47.3) 8(18.2) 5(22.7) CRP 4.375 0.011 正常 24(21.4) 13(29.5) 11(50) 升高 55(49.1) 18(40.9) 8(36.4) COO分型(型) 6.764 0.034 GCB 33(29.5) 5(11.4) 8(36.4) non-GCB 77(68.8) 39(88.6) 13(59.1) Ki-67 7.001 0.030 <80 36(32.1) 24(54.5) 8(36.7) ≥80 74(66.1) 19(43.2) 14(63.6) 中枢侵犯 0.885 0.642 有 5(4.5) 2(4.5) 0 无 106(94.6) 42(95.5) 21(95.5) 骨髓侵犯 2.795 0.247 有 13(11.6) 3(6.8) 2(9.1) 无 94(83.9) 39(88.6) 18(81.8) 大肿块直径(cm) 0.197 0.906 ≤10 53(47.3) 24(54.5) 14(63.6) >10 59(52.7) 20(45.5) 8(36.4) ( )内单位为% -
[1] Pfreundschuh M, Kuhnt E, Trümper L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group[J]. Lancet Oncol, 2011, 12(11):1013-1022. doi: 10.1016/S1470-2045(11)70235-2 [2] Takahashi H, Tomita N, Yokoyama M, et al. Prognostic impact of extranodal involvement in diffuse large B-cell lymphoma in the rituximab era[J]. Cancer, 2012, 118(17):4166-4172. doi: 10.1002/cncr.27381 [3] Ollila TA, Olszewski AJ. Extranodal diffuse large B cell lymphoma: molecular features, prognosis, and risk of central nervous system recurrence[J]. Curr Treat Options Oncol, 2018, 19(8):20-24. [4] Song MK, Chung JS, Sung-Yong O, et al. Clinical impact of bulky mass in the patient with primary extranodal diffuse large B cell lymphoma treated with R-CHOP therapy[J]. Ann Hematol, 2010, 89(10):985-991. doi: 10.1007/s00277-010-0964-7 [5] Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the lugano classification[J]. J Clin Oncol, 2014, 32(27):3059-3068. doi: 10.1200/JCO.2013.54.8800 [6] Oguchi M, Ikeda H, Isobe K, et al. Tumor bulk as a prognostic factor for the management of localized aggressive non-Hodgkin’s lymphoma: a survey of the Japan lymphoma radiation therapy group[J]. Int J Radiat Oncol Biol Phys, 2000, 48(1):161-168. doi: 10.1016/S0360-3016(00)00480-6 [7] López-Guillermo A, Colomo L, Jiménez M, et al. Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin[J]. J Clin Oncol, 2005, 23(12):2797-2804. doi: 10.1200/JCO.2005.07.155 [8] Coiffier B, Thieblemont C, Van Den Neste E, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the groupe d'Etudes des lymphomes de l'Adulte[J]. Blood, 2010, 116(12):2040-2045. doi: 10.1182/blood-2010-03-276246 [9] Blancher C, Moore JW, Talks KL, et al. Relationship of hypoxia-inducible factor (HIF)-1alpha and HIF-2alpha expression to vascular endothelial growth factor induction and hypoxia survival in human breast cancer cell lines[J]. Cancer Res, 2000, 60(24):7106-7113. [10] Kanemasa Y, Shimoyama T, Sasaki Y, et al. Beta-2 microglobulin as a significant prognostic factor and a new risk model for patients with diffuse large B-cell lymphoma[J]. Hematol Oncol, 2017, 35(4):440-446. [11] Benboubker L, Valat C, Linassier C, et al. A new serologic index for low-grade non-Hodgkin's lymphoma based on initial CA125 and LDH serum levels[J]. Ann Oncol, 2000, 11(11):1485-1491. doi: 10.1023/A:1026789232033 [12] Valat C, Linassier C. A new serologic index for low-grade non-Hodgkin's lymphoma based on initial CA125 and LDH serum levels[J]. Ann Oncol, 2000, 11(8):1485-1491. [13] Huang WC, Zhau HE, Chung LWK. Androgen receptor survival signaling is blocked by anti-beta2-microglobulin monoclonal antibody via a MAPK/lipogenic pathway in human prostate cancer cells[J]. J Biol Chem, 2010, 285(11):7947-7956. [14] Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced International Prognostic Index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era[J]. Blood, 2014, 123(6):837-842. doi: 10.1182/blood-2013-09-524108 [15] Castillo JJ, Winer ES, Olszewski AJ. Sites of extranodal involvement are prognostic in patients with diffuse large B-cell lymphoma in the rituximab era: an analysis of the Surveillance, Epidemiology and End Results database[J]. Am J Hematol, 2014, 89(3):310-314. doi: 10.1002/ajh.23638 [16] Habermann TM, Weller EA, Morrison VA, et al. Rituximab-CHOP versus CHOP alone or with maintenance rituximab in older patients with diffuse large B-cell lymphoma[J]. J Clin Oncol, 2006, 24(19):3121-3127. doi: 10.1200/JCO.2005.05.1003 [17] Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma[J]. N Engl J Med, 2002, 346(4):235-242. doi: 10.1056/NEJMoa011795 [18] Grillo-López AJ. Rituximab: an insider's historical perspective[J]. Semin Oncol, 2000, 27(6 suppl 12): 9-16. [19] Maloney DG, Smith B, Rose A. Rituximab: Mechanism of action and resistance[J]. Semin Oncol, 2002, 29(1):2-9. -



 下载:
下载: