Efficacy and safety of second-line treatment regimens for advanced gastric/esophagogastric junction adenocarcinoma: a retrospective study
-
摘要:
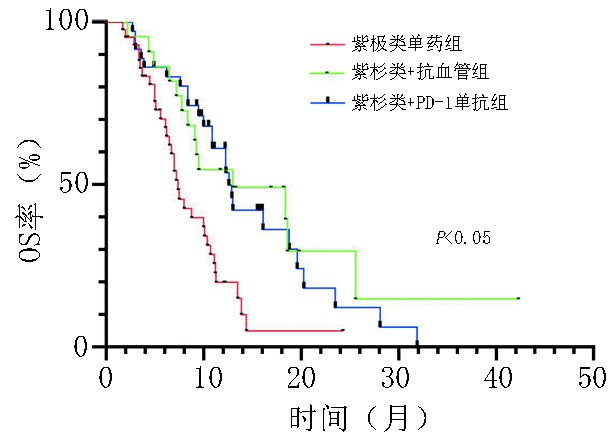
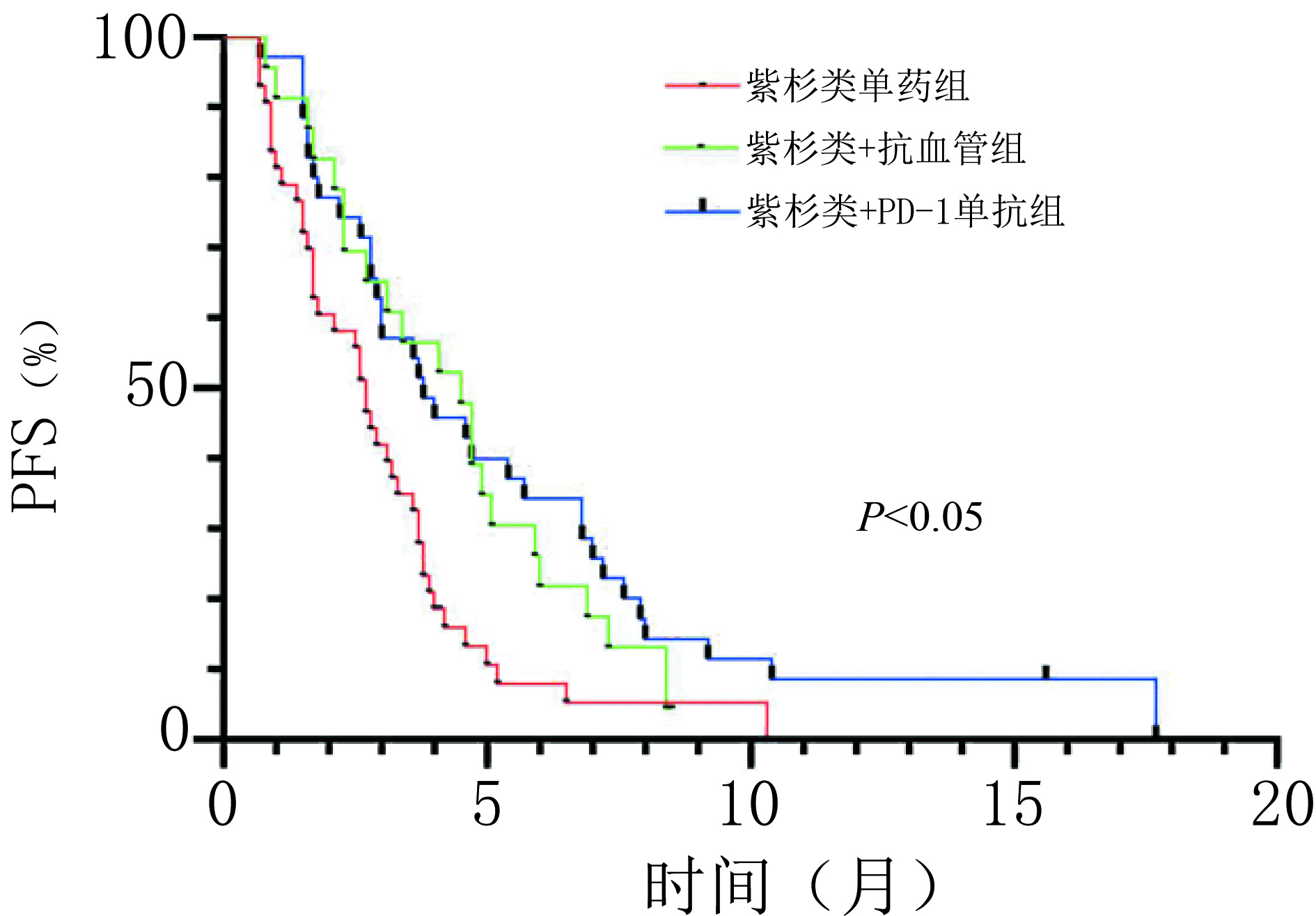
目的 探讨晚期胃/食管胃结合部(gastric/gastroesophageal junction,G/GEJ)腺癌适宜的二线治疗方案。 方法 回顾性分析2019年1月至2021年3月于河南省肿瘤医院以紫杉醇单药和紫杉醇联合抗血管或程序性细胞死亡受体-1(programmed cell death protein 1,PD-1)单抗作为二线治疗的晚期G/GEJ腺癌的临床资料。 结果 收集101例患者,中位随访时间为10.4个月,中位总生存期(overall survival,OS)为9.5个月。紫杉醇单药治疗组(43例)、紫杉类联合抗血管组(22例)和紫杉类联合免疫组(36例)客观缓解率(overall response rate,ORR)分别为9.3%、 27.3% 和30.6%,疾病控制率(disease control rate,DCR)分别为60.5%、86.4% 和80.6%,中位无进展生存期(progression-free survival,PFS)分别为2.7个月、4.3个月和3.9个月,中位OS分别为7.0个月、12.0个月和11.0个月,差异均具有统计学意义(均P<0.05)。三组不良反应均可控,无新型不良事件发生,联合抗血管组高血压发生率为40.9%(9/22),联合PD-1单抗组免疫相关不良反应发生率为19.4%(7/36),与其他两组相比差异均具有统计学意义(均P<0.05);多因素分析显示美国东部肿瘤协作组(ECOG)评分、腹膜转移是患者OS的独立影响因素(P<0.05)。 结论 紫杉类联合抗血管或PD-1单抗治疗可有效延长患者PFS及OS,临床疗效显著且安全性高,是晚期胃癌二线治疗可选择的方案。 Abstract:Objective To explore appropriate second-line treatment regimens for advanced gastric/gastroesophageal junction (G/GEJ) adenocarcinoma. Methods We retrospectively analyzed the clinical data of patients with advanced G/GEJ adenocarcinoma treated with paclitaxel monotherapy and paclitaxel in combination with anti-vascular or programmed cell death receptor 1 (PD-1) monoclonal antibody as second-line therapy at Henan Cancer Hospital from January 2019 to March 2021. Results The clinical data of 101 patients were collected with a median follow-up time of 10.4 months and a median overall survival (OS) of 9.5 months. In the paclitaxel monotherapy (43 cases), paclitaxel combined with anti-vascular (22 cases), and paclitaxel combined with immune (36 cases) groups, the objective response rates (ORR) were 9.3%, 27.3%, and 30.6%, respectively; the disease control rates (DCR) were 60.5%, 86.4%, and 80.6%, respectively; the median progression-free survival (PFS) was 2.7 months, 4.3 months, and 3.9 months, respectively; and the median OS was 7.0 months, 12.0 months, and 11.0 months, respectively, which were statistically significant (P<0.05). Adverse reactions in all three groups were controllable, and no novel adverse events occurred. The incidence of hypertension in the combined anti-vascular group was 40.9% (9/22) and that of immune-related adverse reactions in the combined PD-1 monoclonal antibody group was 19.4% (7/36), both of which were statistically significant compared with those in the other two groups (P < 0.05). A multifactorial analysis showed that the Eastern Cooperative Oncology Group (ECOG) score and peritoneal metastasis were independent influencing factors for patients’ OS (P< 0.05). Conclusions Paclitaxel combined with anti-vascular or PD-1 monoclonal antibody can effectively and safely prolong the PFS and OS of patients and is a second-line treatment option for advanced G/GEJ adenocarcinoma. -
Key words:
- gastric cancer /
- second-line therapy /
- targeted therapy /
- immunotherapy /
- efficacy
-
表 1 晚期G/GEJ腺癌的临床特征
基本特征 A组
(n=43)B组
(n=22)C组
(n=36)χ² P 性别 1.551 0.519 女 10 8 12 男 33 14 24 ECOG评分(分) 3.610 0.165 0~1 32 20 25 2 11 2 11 肝转移 0.872 0.633 是 16 8 10 否 27 14 26 腹膜转移 0.599 0.758 是 11 6 7 否 32 16 29 肿瘤位置 1.839 0.164 G 22 13 26 GEJ 21 9 10 分化程度* 0.223 1.000 中-低 7 3 5 低 25 10 21 PD-L1表达(CPS评分)* 3.592 0.487 <1 9 6 11 1~5 3 7 8 >5 3 2 8 MMR表达* 1.689 0.675 MSS/MSI-L 16 9 19 MSI-H 0 0 2 EBER表达* 1.701 0.426 阴性 11 6 14 阳性 0 1 2 MMR:错配修复;MSS:微卫星稳定;MSI-L:微卫星低度不稳定;MSI-H:微卫星高度不稳定;*:部分患者未行此项检查或病例资料残缺 表 2 三组化疗方案治疗晚期胃癌的临床疗效及PFS与OS对比
组别 A B C χ² P CR(n) 0 1 0 — — PR(n) 4 5 10 — — SD(n) 22 13 18 — — PD(n) 17 3 7 — — ORR(%) 9.3 27.3 30.6 6.399 0.037 DCR(%) 60.5 86.4 80.6 6.501 0.041 PFS(月) 2.7 4.3 3.9 14.139 0.001 OS(月) 7.0 12.0 11.0 16.788 <0.001 表 3 晚期G/GEJ腺癌中位OS单因素及多因素分析
临床特征 单因素分析 多因素分析 χ² P HR(95%CI) P 年龄 79.310 <0.010 — — 分化程度 3.676 0.045 — — ECOG评分 6.504 0.010 2.22(1.01~4.88) 0.048 腹膜转移 12.998 <0.010 3.97(1.76~9.94) 0.001 治疗方案 16.880 <0.010 — 0.004 A组 vs. B组 9.872 0.002 0.24(0.95~0.62) 0.003 A组 vs. C组 10.478 0.001 0.39(0.19~0.78) 0.007 表 4 不良反应发生情况
不良事件 ≥3级 χ² P A组 B组 C组 贫血 11(25.6) 1(4.5) 7(19.4) 1.140 0.581 白细胞减少 10(23.3) 1(4.5) 9(25.0) 0.043 1.000 中性粒细胞减少 9(20.9) 2(9.1) 8(22.2) 3.879 0.159 淋巴细胞减少 2(4.7) 1(4.5) 2(5.6) 2.960 0.224 肝功能损害 0 1(4.5) 0 1.325 0.518 消化道反应 1(2.3) 1(4.5) 1(2.8) 1.870 0.360 高血压 0 3(13.6) 0 17.430 <0.001 免疫相关不良事件 0 0 2(5.6) 10.950 0.001 ( )内单位为% -
[1] Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3):209-249. doi: 10.3322/caac.21660 [2] Ghosn M, Tabchi S, Kourie HR, et al. Metastatic gastric cancer treatment: second line and beyond[J]. World J Gastroenterol, 2016,22(11):3069-3077. [3] Wilke H, Muro K, van Cutsem E, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial[J]. Lancet Oncol, 2014, 15(11):1224-1235. [4] 秦叔逵,李进.阿帕替尼治疗胃癌的临床应用专家共识[J].临床肿瘤学杂志,2015,20(9):841-847. [5] Peng WR, Zhang FL, Wang ZS, et al. Large scale, multicenter, prospective study of apatinib in advanced gastric cancer: a real-world study from China[J]. Cancer Manag Res, 2020, 12:6977-6985. doi: 10.2147/CMAR.S249153 [6] Guo YS, Tang JH, Huang XE, et al. Efficacy and toxicity of apatinib combined with or without chemotherapy for patients with advanced or metastatic chemotherapy-refractory gastric adenocarcinoma: a prospective clinical study[J]. Medicine (Baltimore), 2019, 98(6):e13908. doi: 10.1097/MD.0000000000013908 [7] Nie CY, Lv HF, Liu YJ, et al. Clinical study of sintilimab as second-line or above therapy in patients with advanced or metastatic gastric cancer: a retrospective study[J]. Front Oncol, 2021, 11:741865. [8] Song Y, Li N, Li Q, et al. HX008, an anti-PD1 antibody, plus irinotecan as second-line treatment for advanced gastric or gastroesophageal junction cancer: a multicenter, single-arm phase II trial[J]. J Immunother Cancer, 2020, 8(2):e001279. doi: 10.1136/jitc-2020-001279 [9] Thallinger CMR, Raderer M, Hejna M. Esophageal cancer: a critical evaluation of systemic second-line therapy[J]. J Clin Oncol, 2011, 29(35):4709-4714. doi: 10.1200/JCO.2011.36.7599 [10] Ford HER, Marshall A, Bridgewater JA, et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): an open-label, phase 3 randomised controlled trial[J]. Lancet Oncol, 2014, 15(1):78-86. doi: 10.1016/S1470-2045(13)70549-7 [11] Hironaka S, Ueda S, Yasui H, et al. Randomized, open-label, phase III study comparing irinotecan with paclitaxel in patients with advanced gastric cancer without severe peritoneal metastasis after failure of prior combination chemotherapy using fluoropyrimidine plus platinum: WJOG 4007 trial[J]. J Clin Oncol, 2013, 31(35):4438-4444. doi: 10.1200/JCO.2012.48.5805 [12] Shitara K, Takashima A, Fujitani K, et al. Nab-paclitaxel versus solvent-based paclitaxel in patients with previously treated advanced gastric cancer(ABSOLUTE): an open-label, randomised, non-inferiority, phase 3 trial[J]. Lancet Gastroenterol Hepatol, 2017, 2(4):277-287. [13] 杨牡丹,高峻,刘晓玲,等.甲磺酸阿帕替尼联合多西他赛二线治疗晚期胃癌的临床研究[J].中华肿瘤杂志,2020,42(7):594-597. doi: 10.3760/cma.j.cn112152-20190121-00027 [14] Zhang Y, Xu JY, Wang Q, et al. Efficacy and safety of second-line therapy with apatinib combined with chemotherapy as second-line therapy in advanced gastric cancer: a single-arm, open-label, prospective, multicenter study[J]. Ann Transl Med, 2022, 10(11):641. [15] Shitara K, Özgüroğlu M, Bang YJ, et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial[J]. Lancet, 2018, 392(10142):123-133. doi: 10.1016/S0140-6736(18)31257-1 [16] Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial[J]. JAMA Oncol, 2018, 4(5):e180013. [17] Shitara K, Van Cutsem E, Bang YJ, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial[J]. JAMA Oncol, 2020, 6(10):1571-1580. -



 下载:
下载: