Glucose metabolism reprogramming in tumor-associated macrophages and targeted therapy
-
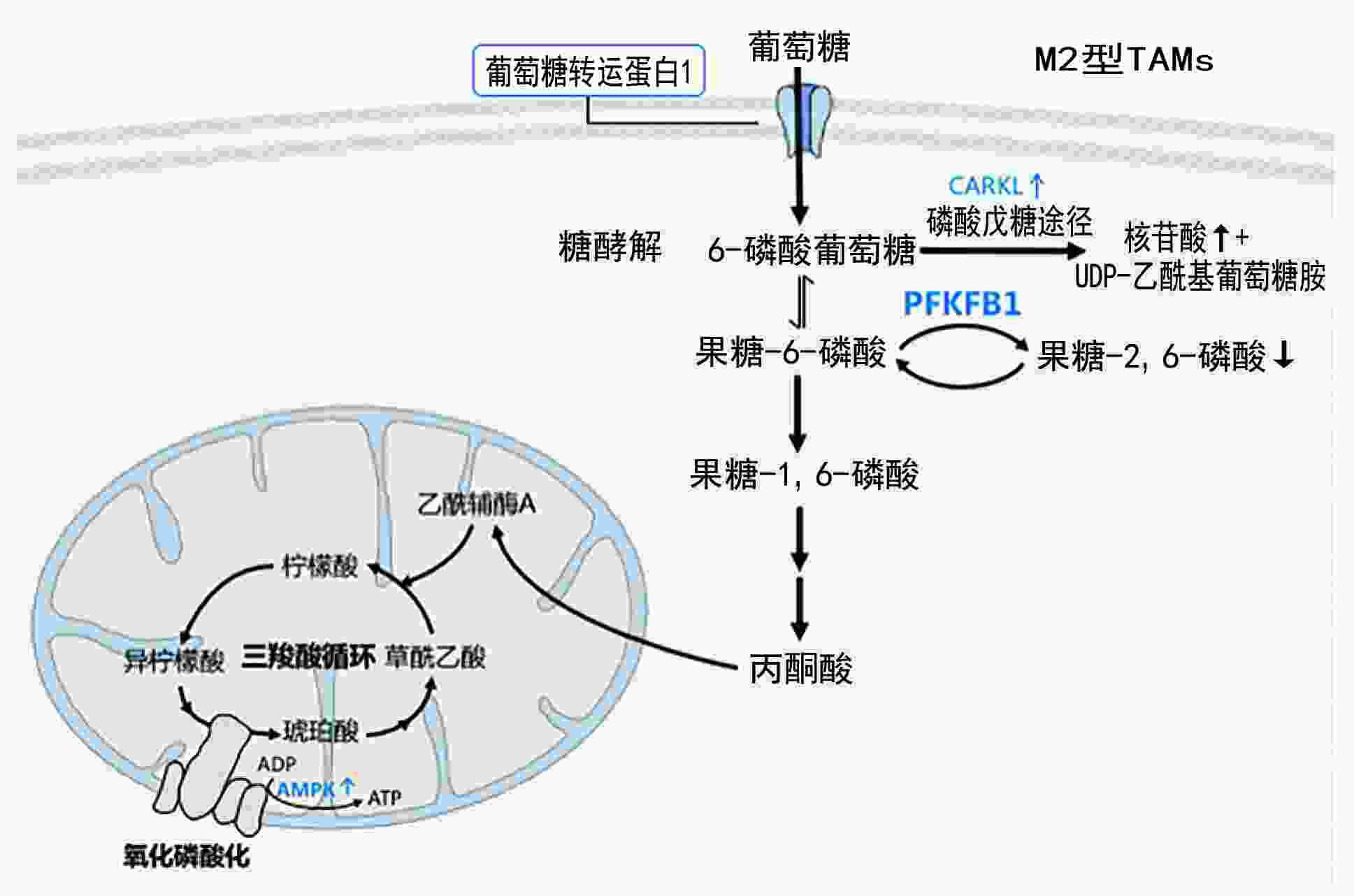
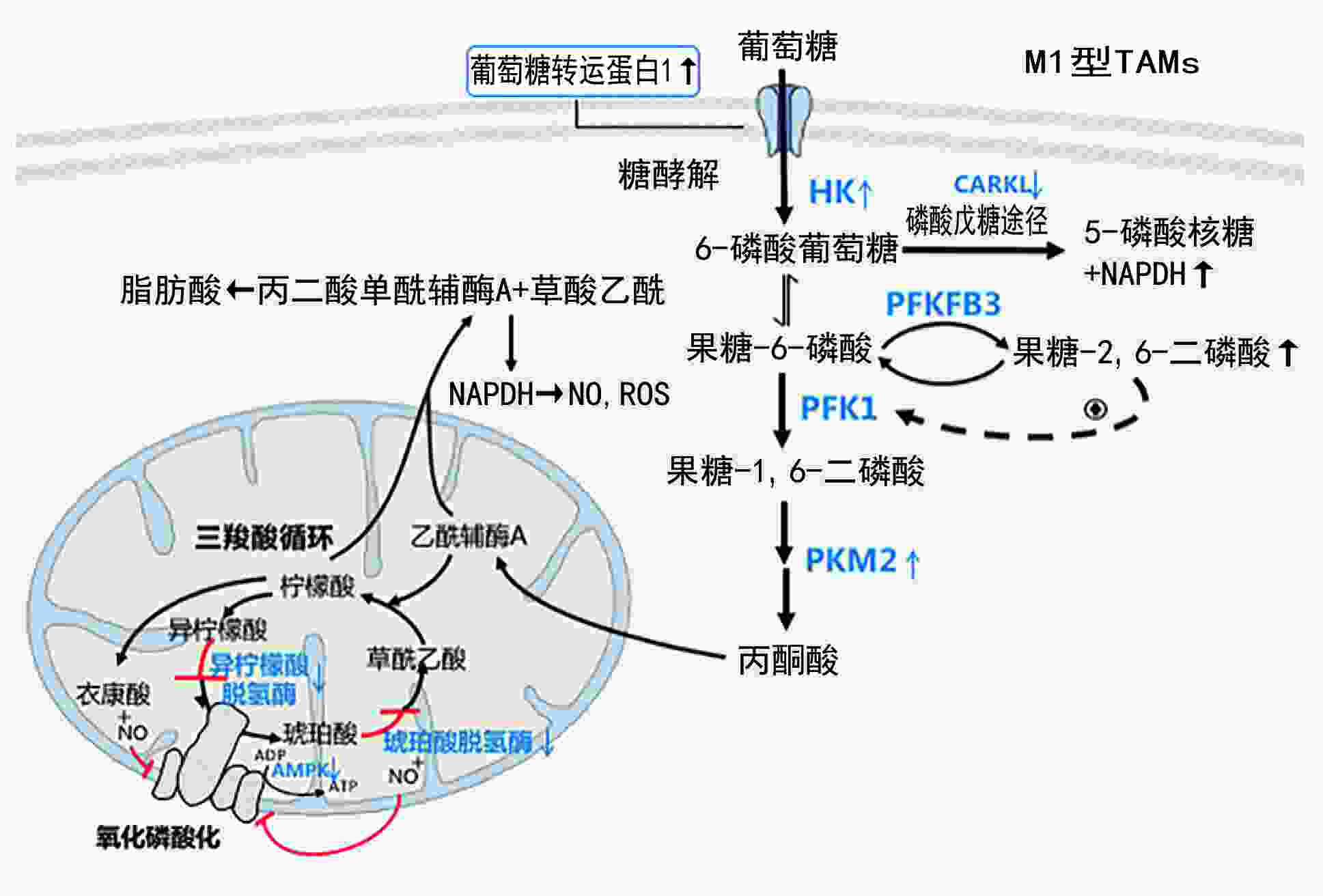
摘要: 肿瘤相关巨噬细胞(tumor-associated macrophages,TAMs)作为肿瘤微环境(tumor microenvironment,TME)中重要的免疫细胞,具有高度异质性及可塑性,在肿瘤细胞分泌的细胞因子刺激下,可发生表型、代谢及功能变化。TAMs代谢改变以糖代谢重编程为主,M1型TAMs有氧糖酵解、磷酸戊糖途径增强,三羧酸循环减弱,具有抗肿瘤功能;M2型TAMs具有完整的三羧酸循环,可促进肿瘤进展。而在TME作用下TAMs具有多种表现形式,其糖代谢重编程可影响肿瘤迁移、侵袭及血管生成,而具体作用机制尚不明确。本文旨在探讨TAMs糖代谢重编程作用机制及其与肿瘤免疫相关性,提示TME中TAMs糖代谢重编程对肿瘤发展和靶向治疗有重要意义,可为肿瘤治疗提供新思路。Abstract: Tumor-associated macrophages (TAMs) are highly heterogeneous and plastic, functioning as critical immune cells in the tumor microenvironment (TME). TAMs change their phenotype, metabolism, and function upon the stimulation of cytokines secreted by tumor cells. Different types of glucose metabolism mainly characterize TAMs metabolism. M1-type TAMs have enhanced aerobic glycolysis and pentose phosphate pathway activity, while triphosphate cycle activity is reduced. In contrast, M2-type TAMs have complete triphosphate cycle activity. The distribution of TAM types within the TME varies, and the reprogramming of glucose metabolism can affect tumor migration, invasion, and angiogenesis. However, the specific mechanisms of glucose metabolism reprogramming affecting the tumor environment remain unclear. This review summarizes the current understanding of glucose metabolism reprogramming mechanisms in TAMs and their correlation with the immune response in the TME. Understanding TAM glucose metabolism reprogramming in tumor development is essential for generating targeted therapies and providing new directions for tumor treatment.
-
表 1 靶向TAMs极化的临床试验药物
药物 靶点和机制 肿瘤类型 临床试验编号 分期 PLX3397 CSF1R抑制剂 实体瘤 NCT02452424 Ⅰ、Ⅱ IMC-CS4 CSF1R抑制剂 实体瘤 NCT01346358 Ⅰ FPA008 抗CSF1R抗体 晚期胰腺癌 NCT03336216 Ⅱ MLN1202 抗CCR2单克隆抗体 转移性癌 NCT01015560 Ⅱ CP-870, 893 激动性抗CD40抗体 实体瘤 NCT02157831 Ⅰ APX005M 激动性抗CD40抗体 消化系统肿瘤 NCT03165994 Ⅱ 2DG HK-2抑制剂 实体瘤 NCT00096707 Ⅰ -
[1] Arneth B. Tumor microenvironment[J]. Medicina (Kaunas), 2019, 56(1):15. doi: 10.3390/medicina56010015 [2] Chen D, Zhang X, Li Z, et al. Metabolic regulatory crosstalk between tumor microenvironment and tumor-associated macrophages[J]. Theranostics, 2021, 11(3):1016-1030. [3] Boutilier AJ, Elsawa SF. Macrophage polarization states in the tumor microenvironment[J]. Int J Mol Sci, 2021, 22(13):6995. doi: 10.3390/ijms22136995 [4] Wang J, Mi S, Ding M, et al. Metabolism and polarization regulation of macrophages in the tumor microenvironment[J]. Cancer Lett, 2022, 543:215766. doi: 10.1016/j.canlet.2022.215766 [5] Reinfeld BI, Madden MZ, Wolf MM, et al. Cell-programmed nutrient partitioning in the tumour microenvironment[J]. Nature, 2021, 593(7858):282-288. doi: 10.1038/s41586-021-03442-1 [6] Liu Y, Xu R, Gu H, et al. Metabolic reprogramming in macrophage responses[J]. Biomark Res, 2021, 9(1):1. doi: 10.1186/s40364-020-00251-y [7] Wu L, Zhang X, Zheng L, et al. RIPK3 orchestrates fatty acid metabolism in tumor-associated macrophages and hepatocarcinogenesis[J]. Cancer Immunol Res, 2020, 8(5):710-721. doi: 10.1158/2326-6066.CIR-19-0261 [8] M de-Brito N, Duncan-Moretti J, C da-Costa H, et al. Aerobic glycolysis is a metabolic requirement to maintain the M2-like polarization of tumor-associated macrophages[J]. Biochim Biophys Acta Mol Cell Res, 2020, 1867(2):118604. doi: 10.1016/j.bbamcr.2019.118604 [9] Moon JS, Hisata S, Park MA, et al. mTORC1-induced HK1-dependent glycolysis regulates NLRP3 inflammasome activation[J]. Cell Rep, 2015, 12(1):102-115. doi: 10.1016/j.celrep.2015.05.046 [10] El Sayed R, Haibe Y, Amhaz G, et al. Metabolic factors affecting tumor immunogenicity: what is happening at the cellular level[J]. Int J Mol Sci, 2021, 22(4):2142. doi: 10.3390/ijms22042142 [11] Yu Q, Wang Y, Dong L, et al. Regulations of glycolytic activities on macrophages functions in tumor and infectious inflammation[J]. Front Cell Infect Microbiol, 2020, 10:287. doi: 10.3389/fcimb.2020.00287 [12] Jiang H, Shi H, Sun M, et al. PFKFB3-Driven macrophage glycolytic metabolism is a crucial component of innate antiviral defense[J]. J Immunol, 2016, 197(7):2880-2890. doi: 10.4049/jimmunol.1600474 [13] Zhang Q, Wang J, Yadav DK, et al. Glucose metabolism: the metabolic signature of tumor associated macrophage[J]. Front Immunol, 2021, 12:702580. doi: 10.3389/fimmu.2021.702580 [14] Xie M, Yu Y, Kang R, et al. PKM2-dependent glycolysis promotes NLRP3 and AIM2 inflammasome activation[J]. Nat Commun, 2016, 7:13280. doi: 10.1038/ncomms13280 [15] Haschemi A, Kosma P, Gille L, et al. The sedoheptulose kinase CARKL directs macrophage polarization through control of glucose metabolism[J]. Cell Metab, 2012, 15(6):813-826. doi: 10.1016/j.cmet.2012.04.023 [16] Jha AK, Huang SC, Sergushichev A, et al. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization[J]. Immunity, 2015, 42(3):419-430. doi: 10.1016/j.immuni.2015.02.005 [17] Palmieri EM, Gonzalez-Cotto M, Baseler WA, et al. Nitric oxide orchestrates metabolic rewiring in M1 macrophages by targeting aconitase 2 and pyruvate dehydrogenase[J]. Nat Commun, 2020, 11(1):698. doi: 10.1038/s41467-020-14433-7 [18] Sag D, Carling D, Stout RD, et al. Adenosine 5'-monophosphate-activated protein kinase promotes macrophage polarization to an anti-inflammatory functional phenotype[J]. J Immunol, 2008, 181(12):8633-8641. [19] Rendra E, Riabov V, Mossel DM, et al. Reactive oxygen species (ROS) in macrophage activation and function in diabetes[J]. Immunobiology, 2019, 224(2):242-253. doi: 10.1016/j.imbio.2018.11.010 [20] Nielsen SR, Schmid MC. Macrophages as key drivers of cancerprogression and metastasis[J]. Mediators Inflamm, 2017, 2017:9624760. [21] Larionova I, Kazakova E, Patysheva M, et al. Transcriptional, epigenetic and metabolic programming of tumor-associated macrophages[J]. Cancers (Basel), 2020, 12(6):1411. doi: 10.3390/cancers12061411 [22] Wenes M, Shang M, Di Matteo M, et al. Macrophage metabolism controls tumor blood vessel morphogenesis and metastasis[J]. Cell Metab, 2016, 24(5):701-715. doi: 10.1016/j.cmet.2016.09.008 [23] Cassetta L, Pollard JW. Targeting macrophages: therapeutic approaches in cancer[J]. Nat Rev Drug Discov, 2018, 17(12):887-904. doi: 10.1038/nrd.2018.169 [24] Yang H, Zhang Q, Xu M, et al. CCL2-CCR2 axis recruits tumor associated macrophages to induce immune evasion through PD-1 signaling in esophageal carcinogenesis[J]. Mol Cancer, 2020, 19(1):41. [25] Xiao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer[J]. Pharmacol Ther, 2021, 221:107753. doi: 10.1016/j.pharmthera.2020.107753 [26] Li DK, Wang W. Characteristics and clinical trial results of agonistic anti-CD40 antibodies in the treatment of malignancies[J]. Oncol Lett, 2020, 20(5):176. [27] Chiang CF, Chao TT, Su YF, et al. Metformin-treated cancer cells modulate macrophage polarization through AMPK-NF-κB signaling[J]. Oncotarget, 2017, 8(13):20706-20718. doi: 10.18632/oncotarget.14982 [28] Jiang M, Li X, Zhang J, et al. Dual inhibition of endoplasmic reticulum stress and oxidation stress manipulates the polarization of macrophages under hypoxia to sensitize immunotherapy[J]. ACS Nano, 2021, 15(9):14522-14534. doi: 10.1021/acsnano.1c04068 [29] Wokoun U, Hellriegel M, Emons G, et al. Co-treatment of breast cancer cells with pharmacologic doses of 2-deoxy-D-glucose and metformin: Starving tumors[J]. Oncol Rep, 2017, 37(4):2418-2424. doi: 10.3892/or.2017.5491 [30] Raez LE, Papadopoulos K, Ricart AD, et al. A phase I dose-escalation trial of 2-deoxy-D-glucose alone or combined with docetaxel in patients with advanced solid tumors[J]. Cancer Chemother Pharmacol, 2013, 71(2):523-530. -



 下载:
下载: