Clinical Efficacy of Neo-Adjuvant Intra-Arterial Chemotherapy for Locally Advanced Cervical Carcinoma
-
摘要:
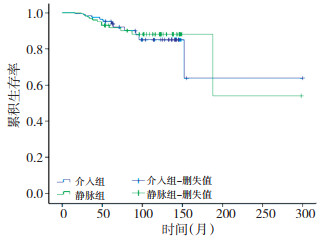
目的 探讨新辅助介入化疗在局部晚期宫颈癌临床治疗中的作用。 方法 对87例局部晚期宫颈癌患者进行回顾性分析, 根据新辅助化疗的不同途径分为新辅助介入化疗(IA-NAC)组42例和新辅助静脉化疗(IV-NAC)组45例。化疗方案为DDP(顺铂)60mg/m2+BLM(博来霉素)25 mg/m2+VCR(长春新碱)1 mg/m2+MMC(丝裂霉素)10mg/m2; 或DDP 70mg/m2+ADM(阿霉素)30 mg/m2。化疗后根据检查决定手术或放疗。评价两组的近期和远期疗效、手术切除率、病理学变化和不良反应。 结果 两组近期疗效均较好, 但无显著性差异; 3、5年生存率两组比较差异无统计学意义(P > 0.05);Ⅱb期患者的手术率IA-NAC组高于IV-NAC组, IA-NAC组平均生存时间较IV-NAC组长(P < 0.05)。 结论 新辅助动脉介入化疗的组织缓解率、平均生存时间较静脉化疗好, 但未能提高其远期生存率。 Abstract:Objective The aim of the current study is to determine the clinical efficacy of neo-adjuvant intra-arterial chemotherapy in locally advanced cervical carcinoma. Methods From January 1997 to December 2005, 87 previously untreated patients with stage IB2-IIB cervical cancers were randomly divided into two groups: IA-NAC and IV-NAC.The drugs used in the current study included cisplatin(60 mg/m2) with mitomycin(10 mg/m2), bleomycin(25 mg/m2), and vincristine(1 mg/m2) or cisplatin(70 mg/m2) with doxorubicin hydrochloride(30 mg/m2) for one course.The response was assessed using pelvic and histological examinations. Results Significant differences in the overall clinical response rate between the two groups were observed.The three-and five-year survival rates were not significantly different either.The operability ofⅡb stage patients was higher in IA-NAC group(P < 0.05) compared with theIV-NAC group, and no significant differences in the mean survival time was found between the two groups(P < 0.05). Conclusion Neo-adjuvant intra-arterial chemotherapy can increase the operability rate in patients with stage IIB cervical cancer and can raise the mean survival time.However, neo-adjuvant intra-arterial chemotherapy does not improve long-term survival rates compared with intravenous chemotherapy. -
表 1 IA-NAC组和IV-NAC组近期疗效对比分析 例
Table 1. Comparison of the overall clinical response rates between the IA-NAC and IV-NAC groups

表 2 IA-NAC组和IV-NAC组组织学对比评价 例
Table 2. Comparison of the histological results between the IA-NAC and IV-NAC groups

-
[1] Motoyama S, Hamana S, Ku Y, et al. Neoadjuvant high-dose intraarterial infusion chemotherapy under percutaneous pelvic perfusion wim extracorporeal chemofiltration in patients with stagesⅢ-Ⅳa cervical cancer[J]. Gynecol Oncol, 2004, 95(3): 576-582. doi: 10.1016/j.ygyno.2004.08.027 [2] Behtash N, Nazari Z, Ayatollahi H, et al. Neoadjuvant chemotherapy and radical surgery compared to radical surgery alone in bulky stage IB-nA cervical cancer[J]. Eur J Surg Oncol, 2006, 32(10), 1226-1230. doi: 10.1016/j.ejso.2006.07.012 [3] Kumar JV, Doval DC, Rao R, et al. A retrospective study of patients with locally advanced cancer of the cervix treated with neoadjuvant chemotherapy followed by radical surgery[J]. Int J Gynecol Cancer, 2009, 19(3): 417-422. doi: 10.1111/IGC.0b013e3181a1c6df [4] Gatcliffe TA, Tewari KS, Shah A, et al. A feasibility study of topotecan with standard-dose cisplatin and concurrent primary radiation therapy in locally advanced cervical cancer[J]. Gynecol Oncol, 2009, 112(1): 85-89. doi: 10.1016/j.ygyno.2008.09.029 [5] 魏丽惠, 孙秀丽. 动脉灌注化疗的应用及前景[J]. 中国妇产科临床杂志, 2003, 4(3): 163-164. https://www.cnki.com.cn/Article/CJFDTOTAL-FKLC200303000.htm [6] Wu Q, Zhang Y. Neoadjuvant intra-arterial infusion chemotherapy followed by surgery in patients with locally advanced cervical cancer[J]. Chin GermanJ Clin Oncol, 2009, 8(9): 537-540. doi: 10.1007/s10330-009-0108-5 [7] Terai Y, Kanemura M, Sasaki H, et al. Long-term follow-up of neoadjuvant intraarterial chemotherapy using an original four-lumen double-balloon(4L-DB) catheter for locally advanced uterine cervical cancer[J]. Int J Clin Oncol, 2009, 14(1): 56-62. doi: 10.1007/s10147-008-0801-3 [8] Eddy GL, Bundy BN, Creasman WT, et al. Treatment of("bulky") stage IB cervical cancer with or without neoadjuvant vincristine and cisplatin prior to radical hysterectomy and pelvic/para-aortic lymphadenectomy: a phase HI trial of the gynecologic oncology group[J]. Gynecol Oncol, 2007, 106(2): 362-369. doi: 10.1016/j.ygyno.2007.04.007 [9] Fanfani F, Fagotti A, Ferrandina G, et al. Neoadjuvant chemoradiation followed by radical hysterectomy in FIGO Stage 111K cervical cancer: feasibility, complications, and clinical outcome[J]. Int J Gynecol Cancer, 2009, 19(6): 1119-1124. doi: 10.1111/IGC.0b013e3181a8b08f [10] 陈慧君, 吴绪锋, 梁川, 等. 新辅助化疗在局部晚期宫颈癌中的疗效观察[J]. 中国肿瘤临床, 2007, 34(4): 46-48. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZL200704014.htm [11] 宋红林, 黄清华, 李力. 新辅助化疗在宫颈癌治疗中的疗效分析[J]. 中国肿瘤临床, 2010, 37(24): 1473-1477. doi: 10.3969/j.issn.1000-8179.2010.24.025 [12] Yamakawa Y, Fujimura M, Hidaka T, et al. Neoadjuvant intraarterial infusion chemotherapy in patients with stage IB2-ⅢB cervical cancer[J]. Gynecol Oncol, 2000, 77(2): 264-270. doi: 10.1006/gyno.2000.5730 -




 下载:
下载: