Relationship of Bcl-2 Expression with MMP2 and MMP9 Expression in Hepatocellular Carcinoma, and Their Correlation with Recurrence, Metastasis, and Prognosis
-
摘要:
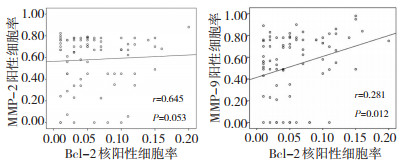
目的 探讨抗凋亡蛋白Bcl-2、基质金属蛋白酶-2(MMP-2)和MMP-9在肝细胞肝癌中表达的关联性, 及其与复发、转移和预后的关系。 方法 以石蜡包埋组织切片, 免疫组织化学SP法染色检测97例肝细胞肝癌, 分别在复发与转移组49例和非复发与转移组48例中分析Bc1-2核表达和MMP-2、MMP-9表达的情况。 结果 复发与转移组Bcl-2核表达高于无复发与转移组(x2=7.912, P=0.005), 复发转移组MMP-9表达高于无复发转移组(x2=6.545, P=0.011), Bcl-2核表达阳性细胞率与MMP-9阳性细胞率存在相关性, 而与MMP-2无关Bcl-2核表达组患者生存时间较短, 差异具有统计学意义。 结论 Bcl-2核表达阳性患者更易出现复发与转移, 且与MMP-9过度表达存在关联性, 表明Bcl-2入核可能对肿瘤转移的相关生物学功能具有调节作用, 可作为评价HCC复发与转移、不良生存预后的候选临床标志。 Abstract:Objective To determine the correlation of the expression of the anti-apoptotic protein Bcl-2 with the expression of matrix metalloproteinases MMP-2 and MMP-9 in human hepatocellular carcinoma, and their relationship with recurrence, metastasis, and prognosis. Methods Paraffin-embedded tissue sections of 97 hepatocellular carcinoma cases were subjected to SP immunohistochemical staining.The nuclear expression of Bcl-2 as well as the expression of MMP-2 and MMP-9 were detected.A total of 49 cases had recurrence and metastasis, and the remaining 48 did not. Results The nuclear expression of Bcl-2 was higher in cases with recurrence and/or metastasis(t = -2.488, P = 0.015).Similarly, MMP-2 and MMP-9 expression were higher in cases with recurrence and/or metastasis(Z = -2.623, P = 0.009;Z = -2.028, P = 0.043).The nuclear expression of Bcl-2 was correlated with the expression of MMP-9, but not with that of MMP-2.Patients with positive nuclear expression of Bcl-2 had shorter survival time, and the difference was significant. Conclusion The nuclear expression of Bcl-2 was significantly correlated with metastasis and MMP-9 expression. Bcl-2 relocation into the nucleus may regulate metastasis-related proteins, which can be used as a clinical marker for evaluating recurrence, metastasis, and prognosis. -
Key words:
- Bcl-2 /
- MMP-2 /
- MMP-9 /
- Recurrence and metastasis
-
表 1 肝癌Bcl-2、MMP-2和MMP-9阳性细胞率与复发转移的关系 例
Table 1. Relationships of the positive rates of Bcl-2, MMP-2, and MMP-9 with metastasis

-
[1] Fransvea E, Angelotti U, Antonaci S, et al. Blocking transforming growth factor—beta up—regulates E—cadherin and reduces migration and invasion of hepatocellular carcinoma cells[J]. Hepatology, 2008, 47(5): 1557-1566. doi: 10.1002/hep.22201 [2] Yang MH, Chen CL, Chau GY, et al. Comprehensive analysis of the independent effect of twist and snail in promoting metastasis of hepatocellular carcinoma[J]. Hepatology, 2009, 50(5): 1464-1474. doi: 10.1002/hep.23221 [3] Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis[J]. Cell, 2004, 118(3): 277-279. doi: 10.1016/j.cell.2004.07.011 [4] Yang J, Mani SA, Donaher JL, et al. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis[J]. Cell, 2004, 117(7): 927-939. doi: 10.1016/j.cell.2004.06.006 [5] Sun T, Sun BC, X Zhao L, et al. Promotion of tumor cell metastasis and vasculogenic mimicry by way of transcription coactivation by Bel-2 and Twistl: a study of hepatocellular carcinoma[J]. Hepatology, 2011, 54(5): 1690-1706. doi: 10.1002/hep.24543 [6] Thiery JP, Sleeman JP. Complex networks orchestrate epithelial -mesenchymal transitions[J]. Nat Rev Mol Cell Biol, 2006, 7(2): 131-142. doi: 10.1038/nrm1835 [7] Trisciuoglio D, Gabellini C, Desideri M, et al. Bel-2 regulates HIF—lalpha protein stabilization in hypoxic melanoma cells via the molecular chaperone HSP90[J]. PLoS One, 2010, 5(7): e11772. doi: 10.1371/journal.pone.0011772 [8] Zuo J, Ishikawa T, Boutros S, et al. Bel-2 overexpression induces a partial epithelial to mesenchymal transition and promotes squamous carcinoma cell invasion and metastasis[J], Mol Cancer Res, 2010, 8(2): 170-182. [9] Moreno-Bueno G, Portillo F, Cano A. Transcriptional regulation of cell polarity in EMT and cancer[J]. Oncogene, 2008, 27(55): 6958-6969. doi: 10.1038/onc.2008.346 [10] Sun T, Zhao N, Zhao XL, et al. Expression and functional significance of Twistl in hepatocellular carcinoma: its role in vasculogenic mimicry[J]. Hepatology, 2010, 51(2): 545-556. doi: 10.1002/hep.23311 [11] Saintigny Y, Dumay A, Lambert S, et al. A novel role for the Bel-2 protein family: specific suppression of the RAD51 recombination pathway[J]. EMBO J, 2001, 20(10): 2596-2607. doi: 10.1093/emboj/20.10.2596 [12] Reed JC. Bel—2—family proteins and hematologic malignancies: history and future prospects[J]. Blood, 2008, 111(7): 3322-3330. doi: 10.1182/blood-2007-09-078162 [13] Tang Y, Nakada MT, Kesavan P, et al. Extracellular matrix metalloproteinase inducer stimulates tumor angiogenesis by elevating vascular endothelial cell growth factor and matrix metalloproteinases[J]. Cancer Res, 2005, 65(8): 3193-3199. doi: 10.1158/0008-5472.CAN-04-3605 [14] Deryugina EI, QuigleyJP. Matrix metalloproteinases and tumor metastasis[J]. Cancer Metastasis Rev, 2006, 25(1): 9-34. doi: 10.1007/s10555-006-7886-9 [15] Donadio AC, Remedi MM, Susperreguy S, et al. Extracellular matrix metalloproteinase inducer(EMMPRIN) and matrix metalloproteinases (MMPs) as regulators of tumor—host interaction in a spontaneous metastasis model in rats[J]. Histochem Cell Biol, 2008, 130 (6): 1155-1164. doi: 10.1007/s00418-008-0496-6 [16] Orlichenko LS, Radisky DC. Matrix metalloproteinases stimulate epithelial—mesenchymal transition during tumor development[J]. Clin Exp Metastasis, 2008, 25(6): 593-600. doi: 10.1007/s10585-008-9143-9 [17] Zhao XL, Sun T, Che N, et al. Promotion of hepatocellular carcinoma metastasis tlrrough matrix metalloproteinase activation by epithelial—mesenchymal transition regulator Twistl[J]. J Cell Mol Med, 2011, 15(3): 691-700. doi: 10.1111/j.1582-4934.2010.01052.x -




 下载:
下载: