Effect of ERK and Akt Inhibition on Trichostatin A-induced Apoptosis of Ovarian Cancer Cells
-
摘要:
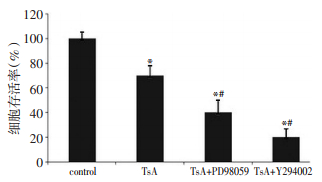
目的 探讨抑制胞外信号调节激酶(extracellular regulated protein kinases,ERK)和蛋白激酶B(Akt)对曲古柳菌素(TSA)诱导的卵巢癌细胞凋亡的影响。 方法 常规培养OVCAR-3卵巢癌细胞株,分为对照、TSA、TSA+PD98059和TSA+Y294002四组,其中TSA组为TSA 2.5 μM处理24 h,TSA+PD98059组和TSA+Y294002组采用5 μM PD98059或Y294002预处理1 h后,2.5 μM TSA处理24 h。MTT法分析各组细胞生存情况,Western blot法检测P53及细胞色素C蛋白的表达,分光光度法检测Caspase-9,Caspase-8和Caspase-3活性。 结果 MTT分析结果显示,与对照组比较,TSA处理后显著降低OVCAR-3细胞的存活率(P < 0.05);与TSA单独处理比较,采用5 μM PD98059或Y294002预处理可以进一步降低OVCAR-3细胞的存活率(P < 0.05)。Western bot分析结果显示TSA处理可以显著增加OVCAR-3细胞中P53及细胞色素C蛋白的表达,增加OVCAR-3细胞Caspase-9,Caspase-8和Caspase-3活性;对比TSA单独处理,TSA和Y294002联合应用可以增加OVCAR-3细胞中P53及细胞色素C蛋白的表达,提高Caspase-9,Caspase-8和Caspase-3的活性(P < 0.05),而TSA和PD98059联合应用无这种效果。 结论 TSA可以诱导卵巢癌OVCAR-3细胞凋亡,其机制可能与上调P53及细胞色素C蛋白表达,激活Caspase-9、Caspase-8、Caspase-3和线粒体凋亡途径有关。抑制Akt可以通过以上机制进一步促进TSA诱导的细胞凋亡。 Abstract:Objective To explore the effect of ERK and Akt inhibition on trichostatin A (TSA) -induced apoptosis of ovarian carcinoma cells. Methods OVCAR-3 cells were divided into four groups as follows: control group, TSA treatment group, TSA + PD98059 group, and TSA + Y294002. The cells were pretreated with PD98059 or Y294002 for 1 h, and then treated with TSA for 24 h. The cell viability was assayed via MTT. The apoptotic-related proteins were detected by Western blot. The activities of the caspases were determined using the caspase assay kits according to the manufacturer's instructions. Results Compared with that of the control groups, the cell viability decreased in the TSA treatment group. The pretreatment with PD98059 or Y294002 further decreased the cell viability. The result of the Western blot indicated that the TSA treatment increased the expression of cytochrome C and P53 and the expression of caspase-8, caspase-9, and caspase-3. The inhibition of Akt promoted the TSA-induced apoptosis and the activation of apoptosis-related proteins and caspases. However, the inhibition of ERK did not yield the same results. Conclusion TSA can induce the apoptosis of OVCAR-3 cells by increasing the protein expression of cytochrome C and P53 and promoting the activation of caspase-8, caspase-9, and caspase-3. Moreover, TSA enhanced the mitochondria-mediated apoptotic pathways. The inhibition of Akt may increase the apoptotic effect of TSA through the biological mechanisms mentioned above. -
Key words:
- ERK /
- Akt /
- Trichostatin A /
- Ovarian cancer /
- Apoptosis
-
图 2 Western blot检测各处理组凋亡相关蛋白的表达
A. Western blot检测卵巢癌OVCAR-3细胞P53和细胞色素C蛋白表达情况(1. 对照组;2. TSA组;3. TSA+PD98059;4. TSA+Y294002);B. 各处理组P53半定量分析结果(*:与control组比较,P<0.05;#:与TSA组比较,P<0.05);C. 各处理组细胞色素C半定量分析结果(*:与control组比较,P<0.05;#:与TSA组比较,P<0.05)
Figure 2. Expression of apoptosis-related protein in the four treatment groups by Western blot
-
[1] Platta CS, Greenblatt DY, Kunnimalaiyaan M, et al. The HDAC inhibitor trichostatin A inhibits growth of small cell lung cancer cells [J]. J Surg Res, 2007, 142(2): 219-226. doi: 10.1016/j.jss.2006.12.555 [2] Wu ZQ, Zhang R, Chao C, et al. Histone deacetylase inhibitor trichostatin A induced caspase-independent apoptosis in human gastric cancer cell[J]. Chin Med J(Engl), 2007, 120(23): 2112-2118. http://d.wanfangdata.com.cn/Periodical/zhcmj200723009 [3] Zhou C, Qiu L, Sun Y, et al. Inhibition of EGFR/PI3K/AKT cell survival pathway promotes TSA's effect on cell death and migration in human ovarian cancer cells[J]. Int J Oncol, 2006, 29(1): 269-278. http://www.spandidos-publications.com/ijo/29/1/269/download [4] Nishioka C, Ikezoe T, Yang J, et al. Inhibition of MEK/ERK signaling synergistically potentiates histone deacetylase inhibitor-induced growth arrest, apoptosis and acetylation of histone H3 on p21waf1 promoter in acute myelogenous leukemia cell[J]. Leukemia, 2008, 22 (7): 1449-1452. doi: 10.1038/sj.leu.2405079 [5] Rosato RR, Grant S. Histone deacetylase inhibitors in cancer therapy[J]. Cancer Biol Ther, 2003, 2(1): 30-37. doi: 10.1200/jco.2009.22.1291 [6] Camins A, Pallas M, Silvestre JS. Apoptotic mechanisms involved in neurodegenerative diseases: experimental and therapeutic approaches[J]. Methods Find Exp Clin Pharmacol, 2008, 30(1): 43-65. doi: 10.1358/mf.2008.30.1.1090962 [7] Yadav V, Zhang X, Liu J, et al. Reactivation of Mitogen-Activated Protein Kinase (MAPK) Pathway by FGF Receptor 3 (FGFR3)/Ras Mediates Resistance to Vemurafenib in Human B-RAF V600E Mutant Melanoma[J]. J Biol Chem, 2012. [Epub ahead of print] http://annonc.oxfordjournals.org/cgi/ijlink?linkType=ABST&journalCode=jbc&resid=287/33/28087 [8] Im SR, Jang YJ. Aspirin enhances TRAIL-induced apoptosis via regulation of ERK1/2 activation in human cervical cancer cells[J]. Biochem Biophys Res Commun, 2012. [Epub ahead of print] http://www.onacademic.com/detail/journal_1000035358929310_036e.html [9] Hoekstra AV, Ward EC, Hardt JL. Chemosensitization of endometrial cancer cells through AKT inhibition involves FOXO1[J]. Gynecol Oncol, 2008, 108(3): 609-618. doi: 10.1016/j.ygyno.2007.11.007 [10] Wiman KG. Strategies for therapeutic targeting of the p53 pathway in cancer. Cell Death Differ, 2006, 13(6): 921-926. doi: 10.1038/sj.cdd.4401921 -




 下载:
下载: