Clinical observation of postoperative hepatic arterial infusion and systemic adjuvant chemotherapy in patients with stage Ⅲ colorectal cancer
-
摘要:
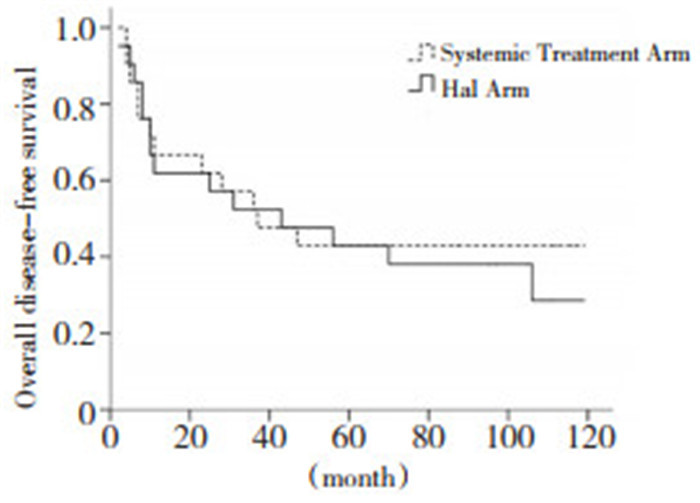
目的 通过与静脉辅助化疗对照, 观察经肝动脉灌注联合静脉辅助化疗对Ⅲ期结直肠癌术后肝转移、无病生存期及总生存期的影响。 方法 2002年1月至2006年3月, 21例Ⅲ期结直肠癌患者作为治疗组, 术后给予肝动脉灌注FUDR联合静脉应用草酸铂化疗, 同期对照21例Ⅲ期结直肠癌患者, 术后给予草酸铂联合CF/5-FU静脉化疗。主要观察终点为肝转移率及DFS, 次要终点为OS和用药安全性。 结果 中位随访65(9~119)个月, 治疗组肝转移发生率较低(9.5%vs.28.6%, P=0.109), 肺转移发生率略高(28.6%vs.14.3%, P=0.256)。2组5年DFS(38.1%vs.42.9%, P=0.671)及OS(47.9%vs.45.0%, P=0.784)无统计学差异。化疗副反应多为Ⅰ~Ⅱ度血白细胞减少、恶心呕吐及感觉神经障碍。 结论 Ⅲ期结直肠癌术后给予经肝动脉联合静脉系统化疗, 与静脉化疗相比, 可能会降低肝转移的发生率, DFS及OS无统计学差异, 化疗副反应较轻, 可耐受。 Abstract:Objective This study aims to investigate whether postoperative hepatic arterial infusion combined with systemic adjuvant chemotherapy is more effective compared with systemic adjuvant chemotherapy alone in terms of survival and liver metastasis for patients with stage Ⅲ colorectal cancer. Methods From January 2002 to March 2006, 21 patients with stage Ⅲ colorectal cancer were assigned to postoperative locoregional chemotherapy with fluorodeoxyuridine and systemic adjuvant chemotherapy with oxaliplatin(treatment group).Another group of 21 patients were assigned to the systemic adjuvant chemotherapy with oxaliplatin as well as fluorouracil and folinic acid(control goup).The primary endpoint of the observation included 5-year disease-free survival(DFS) and liver metastasis.The secondary endpoint was the overall survival(OS) and toxic effects. Results Median follow-up was 65 months(range: 9 months to 119 months).The incidence rate of liver metastasis in patients who underwent hepatic arterial infusion and systemic chemotherapy was 9.5%, whereas that in patients who underwent systemic chemotherapy alone was calculated at 28.6%(P=0.109).By contrast, the incidence of lung metastasis was relatively higher; however, the difference was not significant(28.6% vs.14.3%, P =0.256).No significant difference in the 5-year DFS(38.1% vs.42.9%, P=0.671) and the 5-year OS(47.9%vs.45.0%, P=0.784) was indicated between the two groups.Toxicities such as reduction in leukocytes as well as nausea and peripheral neurosensory toxicity were mild and could be treated by medication. Conclusion Postoperative hepatic arterial infusion combined with systemic chemotherapy can possibly reduce the occurrence of liver metastasis, but may add no further benefit to the DFS and OS of the patients with stage Ⅲ cancer obtaining systemic chemotherapy alone.The toxicities were mild and tolerable. -
Key words:
- colorectal cancer /
- hepatic arterial infusion /
- chemotherapy
-
表 1 Ⅲ期结直肠癌术后辅助化疗中治疗组与对照组的一般资料比较
Table 1. Comparison of the clinical data in patients undergoing adjuvant chemotherapy after surgery of stage Ⅲ colorectal cancer between the two groups

表 2 化疗不良反应
Table 2. Toxicity and complications after chemotherapy

表 3 治疗组与对照组不同首发转移部位
Table 3. Various primary sites of metastasis in the treatment and control groups

-
[1] Taylor L. Adjuvant chemotherapy after resection of liver metasta ses from colorectal cancer[J]. Eur J Cancer, 2008, 44(9): 1198-1201. doi: 10.1016/j.ejca.2008.04.007 [2] Nordlinger B, Rougier P, Arnaud JP, et al. Adjuvant regional che motherapy and systemic chemotherapy versus systemic chemother apy alone in patients with stage Ⅱ-Ⅲ colorectal cancer: a multicen tre randomised controlled phase Ⅲ trial[J]. Lancet Oncol. 2005, 6(7): 459-468. [3] Bolton JS, O'Connell MJ, Mahoney MR, et al. Hepatic arterial infusion and systemic chemotherapy after multiple metastasectomy in patients with colorectal carcinoma metastatic to the liver: a North Central Cancer Treatment Group(NCCTG) phase Ⅱ study, 92-46-52[J]. Clin Colorectal Cancer, 2012, 11(1): 31-37. doi: 10.1016/j.clcc.2011.03.029 [4] Xu J, Zhong Y, Weixin N, et al. Preoperative hepatic and regional arterial chemotherapy in the prevention of liver metastasis aftercolorectal cancer surgery[J]. Ann Surg, 2007, 245(4): 583-590. doi: 10.1097/01.sla.0000250453.34507.d3 [5] Sadahiro S, Suzuki T, Ishikawa K, et al. Prophylactic hepatic arteri al infusion chemotherapy for the prevention of liver metastasis inpatients with colon carcinoma: a randomized control trial[J]. Cancer, 2004, 100(3): 590-597. [6] Jonker DJ, Spithoff K, Maroun J, et al. Adjuvant systemic chemo therapy for Stage Ⅱ and Ⅲ colon cancer after complete resection: an updated practice guideline[J]. Clin Oncol, 2011, 23(5): 314-322. doi: 10.1016/j.clon.2011.02.010 [7] 马力文, 肖宇, 张照辉, 等. 氟脲苷肝动脉持续灌注联合草酸铂静脉化疗治疗肝转移癌[J]. 中国肿瘤临床与康复, 2005, 12(6): 513-516. doi: 10.3969/j.issn.1005-8664.2005.06.011 [8] Samaras P, Breitenstein S, Haile SR, et al. Selective intra-arterial chemotherapy with floxuridine as second-or third-line approach in patients with unresectable colorectal liver metastases[J]. Ann Surg Oncol, 2011, 18(7): 1924-1931. [9] Bouchahda M, Lévi F, Adam R, et al. Modern insights into hepatic arterial infusion for liver metastases from colorectal cancer[J]. Eur J Cancer, 2011, 47(18): 2681-2690. doi: 10.1016/j.ejca.2011.06.037 [10] 王墨培, 马力文, 张淑兰, 等. 含草酸铂化疗方案治疗大肠癌神经毒性的临床研究[J]. 肿瘤防治杂志, 2004, 11(2): 168-170. https://www.cnki.com.cn/Article/CJFDTOTAL-QLZL200402020.htm -




 下载:
下载: