DHA-inhibited proliferation through the PTEN/PI3K/Akt pathway in gastric cancer SGC7901 cells
-
摘要:
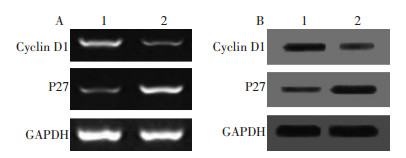
目的 探讨二氢青蒿素(dihydroartemisinin, DHA)通过PTEN/PI3K/Akt通路对人胃癌细胞株SGC7901细胞周期的影响及其分子机制。 方法 不同浓度(6.25、12.5、25、50、100μmol/L)DHA作用SGC7901细胞24、48、72h后, 细胞计数法检测SGC7901细胞增殖的情况。不同浓度DHA作用SGC7901细胞24 h后, 流式细胞术测定细胞周期的分布; RT-PCR和Western blotting分别测定cyclin D1、P27的mRNA和蛋白的表达水平; Western blotting测定PTEN、PI3K、p-Akt的表达水平。分别以PTEN特异性小干扰RNA(PTEN-siRNA)及无关序列对照siRNA(non-specific siRNA, NS-siRNA)转染细胞, 加入100μmol/L DHA, 作用SGC7901细胞24 h后, Western blotting测定cyclin D1、P27、PTEN、PI3K、p-Akt的表达水平。 结果 DHA剂量和时间依赖性抑制SGC7901细胞的增殖, 使细胞周期阻滞于G1期(P < 0.05)。RT-PCR和Western blotting分析结果显示, 100μmol/L DHA作用SGC7901细胞24 h后, cyclin D1 mRNA和蛋白表达显著下降, P27 mRNA和蛋白表达显著上升(P < 0.05)。PTEN的蛋白表达显著增加, PI3K和p-Akt的表达水平逐渐下降(P < 0.05)。敲低PTEN表达后, DHA对PI3K和p-Akt的表达水平的影响明显减弱, 与此同时, cyclin D1表达水平升高, P27表达有所下降(P < 0.05)。 结论 DHA通过抑制PTEN/PI3K/Akt信号通路的活化, 影响细胞增殖相关基因cyclin D1和P27的表达, 进而使细胞阻滞于G0/G1期, 抑制人胃癌SGC7901细胞的增殖。 Abstract:Objective This study aims to elucidate the mechanism of anti-proliferation induced by dihydroartemisinin(DHA) on human gastric cancer SGC7901 cells through the PTEN/PI3K/Akt pathway. Methods SGC7901 cells were treated with DHA at various concentrations(6.25, 12.5, 25, 50, and 100 μmol/L) in different lengths of time(24, 48, and 72 h). We detected the changes in proliferation through cell count. The cell cycles were measured through flow cytometry. The cells were treated with 100 μmol/L DHA and were cultured for 24 h. The expressions of cyclin D1 and P27 were detected through reverse transcription-polymerase chain reaction and Western blot analysis, respectively. The expressions of PTEN, PI3K, and p–Akt were also detected through Western blot analysis. The PTEN expression was downregulated by the RNAi technology. The cells were subsequently treated with 100 μmol/L DHA for 24 h, followed by a Western blot analysis for the expressions of PTEN, PI3K, p–Akt, cyclin D1, and P27. Results Resultsof the cell count showed that DHA greatly inhibited the growth and proliferation of SGC7901 cells in a dose-and time-dependent manner(P < 0.05). The DHA inhibited the proliferation of SGC7901 cells through the induced cell cycle G1phrase arrest. The expressions of cyclin D1, PI3K, and p–Akt were downregulated, and the expressions of P27 and PTEN were upregulated after DHA treatment(P < 0.05). The DHA-elicited decrease in cyclin D1, PI3K, and p-Akt expression was significantly induced, and the DHA-elicited increase in P27 and PTEN expression was significantly reduced(P < 0.05) when the SGC7901 cells were transfected with PTEN-specific siRNA to block the endogenous PTEN expression induced by DHA. Conclusion DHA induces the cell cycle G0/G1 phase arrest through the regulation of cyclin D1 and P27 expression by activating the PTEN/ PI3K/Akt signaling pathway in human gastric cancer SGC7901 cells. -
Key words:
- dihydroartemisinin /
- gastric cancer /
- proliferation /
- Akt signal transduction pathway /
- cell cycle
-
表 1 DHA作用后SGC7901细胞周期分布的比较(%,x±s)
Table 1. Comparison of the SGC7901 cell cycle distributions after treatment with DHA(%, x±s)

表 2 DHA对SGC7901细胞cyclin D1、P27 mRNA和蛋白表达的影响(n=6,x±s)
Table 2. Effects of DHA on mRNA and protein levels of cyclin D1 and P27 in SGC7901 cells(n=6, x±s)

表 3 DHA对SGC7901细胞PTEN/PI3K/Akt信号通路的影响(n=6,x±s)
Table 3. Effects of DHA on the PTEN/PI3K/Akt signaling pathway(n=6, x±s)

表 4 DHA对转染PTEN-siRNA后SGC7901细胞中PTEN/PI3K/Akt通路及相关基因表达的影响(n=6,x±s)
Table 4. Effects of PTEN siRNA on the PTEN/PI3K/Akt pathway and related gene expression(n=6, x±s)

-
[1] Palesty JA, Wang W, Javle MM, et al. Side effects of therapy: Case 3. Gastric cancer after radiotherapy of pediatric Hodgkin's disease[J]. J Clin Oncol, 2004, 22(12): 2507-2509. doi: 10.1200/JCO.2004.09.168 [2] Jeyadevan JP, Bray PG, Chadwick J, et al. Antimalarial and antitumor evaluation of novel C-10 non-acetal dimers of 10beta- (2-hydroxyethyl) deoxoartemisinin[J]. J Med Chem, 2004, 47(5): 1290-1298. doi: 10.1021/jm030974c [3] Aung W, Sogawa C, Furukawa T, et al. Anticancer effect of dihydroartemisinin (DHA) in a pancreatic tumor model evaluated by conventional methods and optical imaging[J]. Anticancer Res, 2011, 31 (5): 1549-1558. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_PM21617209 [4] Lu JJ, Chen SM, Zhang XW, et al. The anti-cancer activity of dihydroartemisinin is associated with induction of iron-dependent endoplasmic reticulum stress in colorectal carcinoma HCT116 cells[J]. Invest New Drugs, 2011, 29(6): 1276-1283. doi: 10.1007/s10637-010-9481-8 [5] Lu YY, Chen TS, Wang XP, et al. The JNK inhibitor SP600125 enhances dihydroartemisinin-induced apoptosis by accelerating Bax translocation into mitochondria in human lung adenocarcinoma cells[J]. FEBS Lett, 2010, 584(18): 4019-4026. doi: 10.1016/j.febslet.2010.08.014 [6] Zhong Z, Yeow WS, Zou C, et al. Cyclin D1/cyclin-dependent kinase 4 interacts with filamin A and affects the migration and invasion potential of breast cancer cells[J]. Cancer Res, 2010, 70(5): 2105-2114. doi: 10.1158/0008-5472.CAN-08-1108 [7] Stocker G, Ott K, Henningsen N, et al. Cyclin D1 and interleukin-1 receptor antagonist polymorphisms are associated with prognosis in neoadjuvant-treated gastric carcinoma[J]. Eur J Cancer, 2009, 45(18): 3326-3335. doi: 10.1016/j.ejca.2009.09.021 [8] Zhang W, Tong Q, Wu Q, et al. Upregulated p27kip1 can downregulate survivin expression and inhibit telomerase activity in gastric carcinoma cells[J]. Cancer Invest, 2009, 27(9): 898-900. doi: 10.3109/07357900902918478 [9] Cox SN, Sallustio F, Serino G, et al. Altered modulation of WNT-beta-catenin and PI3K/Akt pathways in IgA nephropathy[J]. Kidney Int, 2010, 78(4): 396-407. doi: 10.1038/ki.2010.138 [10] Wang LY, Wang Y, Xu DS, et al. MDG-1, a polysaccharide from Ophiopogon japonicus exerts hypoglycemic effects through the PI3K/Akt pathway in a diabetic KKAy mouse model[J]. J Ethnopharmacol, 2012, 143(1): 347-354. doi: 10.1016/j.jep.2012.06.050 [11] Carnero A, Blanco-Aparicio C, Renner O, et al. The PTEN/PI3K/ AKT signalling pathway in cancer, therapeutic implications[J]. Curr Cancer Drug Targets, 2008, 8(3): 187-198. doi: 10.2174/156800908784293659 [12] Osaki M, Oshimura M, Ito H. PI3K-Akt pathway: Its functions and alterations in human cancer[J]. Apoptosis, 2004, 9(6): 667-676. doi: 10.1023/B:APPT.0000045801.15585.dd [13] Prasad R, Vaid M, Katiyar SK. Grape Proanthocyanidin Inhibit Pancreatic Cancer Cell Growth In Vitro and In Vivo through Induction of Apoptosis and by Targeting the PI3K/Akt Pathway[J]. PLoS One, 2012, 7(8): e43064. doi: 10.1371/journal.pone.0043064 [14] Li J, Yen C, Liaw D, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer [J]. Science, 1997, 275(5308): 1943-1947. doi: 10.1126/science.275.5308.1943 [15] Leslie NR, Yang X, Downes CP, et al. The regulation of cell migration by PTEN[J]. Biochem Soc Trans, 2005, 33(6): 1507-1508. doi: 10.1042/BST0331507 [16] Gautam A, Li ZR, Bepler G. RRM1-induced metastasis suppression through PTEN-regulated pathways[J]. Oncogene, 2003, 22(14): 2135-2142. doi: 10.1038/sj.onc.1206232 [17] Capodanno A, Camerini A, Orlandini C, et al. Dysregulated PI3K/Akt/ PTEN pathway is a marker of a short disease-free survival in node-negative breast carcinoma[J]. Hum Pathol, 2009, 40(10): 1408-1417. doi: 10.1016/j.humpath.2009.02.005 [18] Despoix N, Walzer T, Jouve N, et al. Mouse CD146/MCAM is a marker of natural killer cell maturation[J]. Eur J Immunol, 2008, 38 (10): 2855-2864. doi: 10.1002/eji.200838469 [19] Graupera M, Guillermet-Guibert J, Foukas LC, et al. Angiogenesis selectively requires the p110alpha isoform of PI3K to control endothelial cell migration[J]. Nature, 2008, 453(7195): 662-666. doi: 10.1038/nature06892 [20] Jiang BH, Zheng JZ, Aoki M, et al. Phosphatidylinositol 3-kinase signaling mediates angiogenesis and expression of vascular endothelial growth factor in endothelial cells[J]. Proc Natl Acad Sci USA, 2000, 97 (4): 1749-1753. doi: 10.1073/pnas.040560897 -




 下载:
下载: