Correlation of WIF-1 expression and clinicopathologic significance in breast cancer with aberrant DNA methylation
-
摘要:
目的 研究WIF-1(Wnt inhibitory factor-1)mRNA在乳腺癌组织中的表达及其启动子区域甲基化情况,进一步探讨WIF-1基因甲基化与乳腺癌临床病理特征的关系。 方法 收集2009年9月1日至2009年12月30日青岛大学附属医院乳腺外科手术切除新鲜组织标本69例,其中良性病变组织9例,乳腺癌及癌旁组织各30例,应用RT-PCR及甲基化特异性PCR(methylation specific PCR,MSP)检测乳腺癌组织、相应癌旁组织和乳腺良性病变组织中WIF-1mRNA表达及其启动子甲基化情况。 结果 癌组织中WIF-1基因表达率明显低于相应癌旁组织及乳腺良性病变组织,具有显著性差异(χ2=41.786,P < 0.05);与其他两组相比甲基化率在癌组织中明显升高(矫正χ2=16.484,P < 0.05);WIF-1基因表达下降与其异常甲基化存在明显关联(P=0.023);WIF-1异常甲基化与乳腺癌发病年龄、肿瘤分级、组织分型和淋巴结转移无相关性(P>0.05)。 结论 异常甲基化可能是乳腺癌WIF-1基因表达下降的重要原因,是乳腺癌发生、发展的重要机制。 Abstract:Objective To investigate the mRNA expression of the WIF-1 gene and the methylation of its promoter in breast cancer, and to determine the correlation between the epigenetic aberrant WIF-1 DNA methylation and the clinicopathological significance of WIF-1 in breast cancer. Methods RT-PCR and sensitive methylation-specific-PCR (MSP) were used to detect WIF-1 mRNA expression and the methylation of the WIF-1 promoter in 30 breast cancer samples as well as in tumor-adjacent tissue samples and 9 benign breast tissues. Results The WIF-1 mRNA expression in 30 breast cancer samples significantly decreased compared with those of the other two groups. In addition, WIF-1 methylation was more frequent in breast-tumor tissues compared with those in tumor-free tissues. Meanwhile, WIF-1 mRNA expression in breast cancer tissues involved the abnormal methylation of its promoter. Clinicopathological correlation analysis showed that the abnormal methylation of the WIF-1 gene promoter was not associated with age, TNM stage, histotype, or lymph node metastasis. Conclusion WIF-1 mRNA expression loss due to abnormal methylation may be a crucial factor in breast cancer development and can thus be used in the prognosis and progression of the disease. -
Key words:
- WIF-1 /
- gene expression /
- breast cancer /
- methylation /
- TNM stage /
-
-
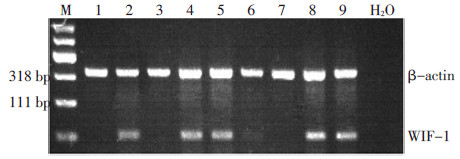
图 1 乳腺癌组织、乳腺癌癌旁组织及乳腺良性病变组织中的WIF-1mRNA表达
Figure 1. mRNA analysis of WIF-1 in breast cancer tissue, adjacent non-cancerous tissue, and hyperplasic breast tissue
M: DNA marker; 1, 3, 5, and 7:breast cancer tissue; 2, 4, 6, and 8: Tumor-adjacent normal tissue; 9:Benign-breast disease tissue; H2O: Blank control
图 2 乳腺癌组织、乳腺癌癌旁组织及乳腺良性病变组织中的WIF-1甲基化状况
Figure 2. Analysis of WIF-1 methylation in breast cancer tissue, adjacent non-cancerous tissue, and hyperplasic breast tissue
P: Positive control; T: Breast cancer tissue; N: Tumor-adjacent normal tissue; B: Benign-breast disease tissue; H2O: Blank control; U: Un⁃ methylated DNA; M: Methylated DNA
表 1 PCR检测引物序列
Table 1. Primer sequences and PCR conditions

3 BGS检测WIF-1基因启动子CpG位点甲基化状态结果
3. BGS analysis of the methylation of the WIF-1 promoter region

表 3 乳腺癌组织WIF-1甲基化与临床特征的关系
Table 3. Relationship between WIF-1 methylation in breast cancer tissue and clinical features

-
[1] Herman JG, Baylin SB. Gene silencing in cancer in association with Promoter hypermethylation[J]. N Engl J Med, 2003, 349(21): 2042-2054. doi: 10.1056/NEJMra023075 [2] 方娟娟, 余卫平, 安艳丽, 等. 乳腺癌中RIZ1基因启动子甲基化状态的探讨[J]. 中国肿瘤临床, 2007, 34(12): 664-669. http://www.cjco.cn/cn/article/doi/ [3] van Hoesel AQ, Sato Y, Elashoff DA, et al. Assessment of DNA methylation status in early stages of breast cancer development[J]. Br J Cancer, 2013, 5(7): 1024-1029. [4] Trifa F, Karray-Chouayekh S, Jmal E, et al. Loss of WIF-1 and Wnt5a expression is related to aggressiveness of sporadic breast cancer in Tunisian patients[J]. Tumour Biol, 2013, 2(16): 812-816. [5] Park SY, Kwon HJ, Lee HE, et al. Promoter CpG island hypermethylation during breast cancer progression[J]. Virchows Arch, 2011, 458(1): 73-84. doi: 10.1007/s00428-010-1013-6 [6] Fendri A, Khabir A, Hadri-Guiga B, et al. Epigenetic alteration of the Wnt inhibitory factor-1 promoter is common and occurs in advanced stage of Tunisian nasopharyngeal carcinoma[J]. Cancer Invest, 2010, 28(9): 896-903. [7] Wissmann C, Wild PJ, Kaiser S, et al. WIF1, a component of the Wnt pathway, is downregulated in prostate, breast, lung, and bladder cancer[J]. J Pathol, 2003, 201(2): 204-212. doi: 10.1002/path.1449 [8] Jürgen Veeck, Peter J Wild, Thomas Fuchs, et al. Prognostic relevance of Wnt-inhibitory factor-1(WIF1) and Dickkopf-3 (DKK3) promoter methylation in human breast cancer[J]. BMC Cancer, 2009, 9(217): 278-393. [9] Urakami S, Shiina H, Enokida H, et al. Epigenetic inactivation of Wnt inhibitory factor-1 plays an important role in bladder cancer through aberrant canonical Wnt/beta-catenin signaling pathway[J]. Clin Cancer Res, 2006, 12(2): 383-391. doi: 10.1158/1078-0432.CCR-05-1344 [10] Gao Z, Xu Z, Hung MS, et al. Procaine and procainamide inhibit the Wnt canonical pathway by promoter demethylation of WIF-1 in lung cancer cells[J]. Oncol Rep, 2009, 22(6): 1479-1484. [11] He B, Reguart N, You L, et al. Blockade of Wnt-1 signaling induces apoptosis in human colorectal cancer cells containing downstream mutations[J]. Oncogene, 2005, 24(18): 3054-3058. doi: 10.1038/sj.onc.1208511 [12] Batra S, Shi Y, Kuchenbecker KM, et al. Wnt inhibitory factor-1, a Wnt antagonist, is silenced by promoter hypermethylationin malignant pleural mesothelioma[J]. Biochem Biophys Res Commun, 2006, 342(4): 1228-1232. doi: 10.1016/j.bbrc.2006.02.084 [13] Chim CS, Fung TK, Wong KF, et al. Infrequent Wnt inhibitory factor-1 (Wif-1) methylation in chronic lymphocytic leukemia[J]. Leuk Res, 2006, 30(9): 1135-1139. doi: 10.1016/j.leukres.2005.12.005 [14] Hou HA, Kuo YY, Liu CY, et al. Distinct association between aberrant methylation of Wnt inhibitors and genetic alterations in acute myeloid leukaemia[J]. Br J Cancer, 2011, 105(12): 1927-1933. doi: 10.1038/bjc.2011.471 [15] Yang Z, Wang Y, Fang J, et al. Expression and aberrant promoter methylation of Wnt inhibitory factor-1 in human astrocytomas[J]. J Exp Clin Cancer Res, 2010, 29(26): 1123-1128. -




 下载:
下载: