Apoptosis -promoting effect of splice-switching oligonucleotides targeting Bcl-x pre-mRNA of the human glioma cell line
-
摘要:
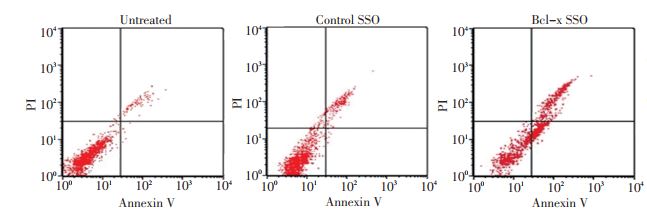
目的 探讨Bcl-x mRNA前体剪接转换寡核苷酸(Bcl-x splice-switching oligonucleotides,Bcl-x SSO)对胶质瘤细胞株U251 增殖和凋亡的影响。 方法 设计靶向 Bcl-x基因下游选择性 5'端剪接位点的 Bcl-x SSO,β-globin SSO 作为阴性对照 SSO。SSO经2'-甲氧乙基(2'-O-methoxyethyl,MOE)、全硫代磷酸化(phosphorothioate,PS)修饰。采用阳离子脂质体将不同的SSO转染至U251细胞。采用四甲基偶氮唑蓝比色法(MTT法)检测Bcl-x SSO对U251细胞的增殖抑制率。流式细胞术定量检测U251细胞的凋亡率。逆转录聚合酶链反应(RT-PCR)方法检测Bcl-x SSO对Bcl-xL、Bcl-xS mRNA表达水平的影响,Western blot方法检测Bcl-x SSO对Bcl-xL、Bcl-xS蛋白表达的影响。 结果 MTT结果显示Bcl-x SSO显著抑制U251细胞的增殖,且抑制作用呈剂量依赖性。流式细胞术检测 Bcl-x SSO 能够明显促进 U251 细胞凋亡。RT-PCR 检测 Bcl-x SSO 处理组 Bcl-xL mRNA 表达水平下降,Bcl-xS mRNA表达水平升高。Western blot检测Bcl-x SSO处理组Bcl-xL蛋白表达水平下降,Bcl-xS蛋白表达水平升高。 结论 在胶质瘤细胞U251中,Bcl-x SSO可特异性的作用于Bcl-x mRNA前体,调节其选择性剪接模式从Bcl-xL转换至Bcl-xS,进而促进U251细胞凋亡。 Abstract:Objective To investigate the proliferation inhibition and apoptosis -promoting effects of splice-switching oligonucleotides targeting the Bcl-x pre-mRNA (Bcl-x SSO) of the human glioma cell line U251. Methods Bcl-x SSO was designed to bind tothe 5'-splice site of exon Ⅱ in Bcl-x pre-mRNA. An oligonucleotide targeted to aberrantly splice human β-globin intron was used as acontrol SSO. SSOs were modified using 2'-O-methoxyethyl and phosphorothioate, and were delivered together with lipofectamine intothe human glioma cell line U251 via cationic liposomes. The proliferation inhibition rate of the cell human cell line U251 was assessedvia MTT assay. Flow cytometry was performed to detect the apoptosis rate. Modulation from Bcl-xL to Bcl-xS was analyzed via reverse transcription polymerase chain reaction and Western blot. Results The study showed that Bcl-x SSO caused proliferation inhibition and induced apoptosis in a dose-dependent manner in the human glioma cell line U251, whereas the control SSO did not show anyevident effect. The expression of Bcl-xL mRNA and protein increased, whereas the expression level of Bcl-xS mRNA decreased in thehuman glioma cell line U251 treated with Bcl-x SSO. Conclusion The study demonstrated that Bcl-x SSO can induce apoptosis in human glioma cell line U251. The mechanism involves the redirection of Bcl-x splicing from Bcl-xl to Bcl-xs. Bcl-x SSOs have the potential to be used as anti-cancer drugs for glioma therapy. -
Key words:
- alternative splicing /
- oligonucleotides /
- apoptosis /
- flow cytometry /
- glioma cell line
-
表 1 不同浓度的Bcl-x SSO和Control SSO作用于U251细胞48h后的增殖抑制率 (%)
Table 1. Proliferation inhibition rate(%)after 48 hours of human glioma U251 cells treated with Bcl-x SSO,and control SSO with different concentrationstions

-
[1] Wang ET, Sandberg R, Luo S, et al. Alternative isoform regulation in human tissue transcription[J]. Nature, 2008, 456(7221):470-476. doi: 10.1038/nature07509 [2] Kim YJ, Kim HS. Alternative splicing and its impact as a cancer diagnostic marker[J]. Genomics Inform, 2012, 10(2):74-80. doi: 10.5808/GI.2012.10.2.74 [3] Zhang MX, Xu YJ, Zhu MC, et al. Overexpressed ostepontin-c as a potential biomarker for esophageal squamous cell carcinoma[J]. Asian Pac J Cancer Prev, 2013, 14(12):7315-7319. doi: 10.7314/APJCP.2013.14.12.7315 [4] Moore MJ, Wang Q, Kennedy CJ, et al. An alternative splicing network links cell cycle control to apoptosis[J]. Cell, 2010, 142(4): 625-636. doi: 10.1016/j.cell.2010.07.019 [5] Miura K, Fujibuchi W, Unno M. Splice variants in apoptotic pathway[J]. Exp Oncol, 2012, 34(3):212-217. [6] Mercatante DR, Bortner CD, Cidlowski JA, et al. Modification of alternative splicing of Bcl-x pre-mRNA in prostate and breast cancer cells. Analysis of apoptosis and cell death[J]. J Biol Chem, 2001, 276(19):16411-16417. doi: 10.1074/jbc.M009256200 [7] Mercatante DR, Mohler JL, Kole R. Cellular response to an antisense-mediated shift of Bcl-x pre-mRNA splicing and antineoplastic agents[J]. J Biol Chem, 2002, 277(51):49374-49382. doi: 10.1074/jbc.M209236200 [8] Shchelkunova A, Ermolinsky B, Boyle M, et al. Tuning of alternative splicing-switch from proto-oncogene to tumor suppressor[J]. Int J Biol Sci, 2013, 9(1):45-54. doi: 10.7150/ijbs.5194 [9] Surget S, Khoury MP, Bourdon JC. Uncovering the role of p53 splice variants in human malignancy: a clinical perspective[J]. Onco Targets Ther, 2013, 7(12):57-68. http://cn.bing.com/academic/profile?id=1cbeebb6524e0043dd1e12020f1a34cd&encoded=0&v=paper_preview&mkt=zh-cn [10] Zhang J, Manley JL. Misregulation of pre-mRNA alternative splicing in cancer[J]. Cancer Discov, 2013, 3(11):1228-1237. doi: 10.1158/2159-8290.CD-13-0253 [11] Ghigna C, Riva S, Biamonti G. Alternative splicing of tumor suppressors and oncogenes[J]. Cancer Treat Res, 2013, 158(4): 95-117. [12] Bauman J, Jearawiriyapaisarn N, Kole R. Therapeutic potential of splice-switching oligonucleotides[J]. Oligonucleotides, 2009, 19(1): 1-13. doi: 10.1089/oli.2008.0161 [13] Bauman JA, Li SD, Yang A, et al. Anti-tumor activity of splice switching oligonucleotides[J]. Nucleic Acids Res, 2010, 38(22): 8348-8356. doi: 10.1093/nar/gkq731 [14] Wan J, Bauman JA, Graziewicz MA, et al. Oligonucleotide therapeutics in cancer[J]. Cancer Treat Res, 2013, 158(3):213-233. https://www.ncbi.nlm.nih.gov/pubmed/24222360 [15] Wong MS, Chen L, Foster C, et al. Regulation of telomerase alternative splicing: a target for chemotherapy[J]. Cell Rep, 2013, 3(4): 1028-1035. https://www.sciencedirect.com/science/article/pii/S2211124713001186 -




 下载:
下载: