In vitro synergistic effect of arsenic trioxide with conventional or new drugs on the proliferation of cutaneous T cell lymphoma cells Hut-78 and Hut-102
-
摘要:
目的 探讨三氧化二砷(arsenic trioxide,As2O3)与传统药物地塞米松(Dexamethasone,DXM)、依托泊苷(Etoposide,VP-16)、甲氨蝶呤(Methotrexate,MTX)和新型药物硼替佐米(Bortezomib,BTZ)、组蛋白去乙酰化酶抑制剂-辛二酰苯胺异羟肟酸(suberoylanilide hydroxamic acid,SAHA)联合对人皮肤T细胞淋巴瘤(CTCL)细胞系Hut-78、Hut-102细胞的增殖抑制作用。 方法 不同浓度的As2O3单药或与DXM/VP-16/MTX/BTZ/SAHA联合作用于Hut-78、Hut-102细胞48 h后,采用MTT法检测细胞增殖抑制率,应用联合指数分析两药是否具有协同效应。 结果 As2O3、DXM、VP-16、MTX、BTZ、SAHA单药对Hut-78、Hut-102细胞的增殖均具有显著的抑制作用,呈剂量依赖性,培养48h的半数抑制浓度分别为5 μmol/L、500 μg/L、2.5 μg/L、1 μg/L、10 μmol/L、2.5 μmol/L。As2O3与DXM/VP-16/MTX/BTZ/SAHA联合时具有协同效应,抗肿瘤作用更为显著。 结论 As2O3单药及其与DXM/VP-16/MTX/ BTZ/SAHA联合在体外可有效抑制CTCL细胞增殖。As2O3是一种很有前景的治疗CTCL的药物,As2O3与DXM/VP-16/MTX/BTZ/ SAHA等传统或新型药物联合可为CTCL临床的治疗提供实验依据。 -
关键词:
- 皮肤T细胞淋巴瘤细胞系 /
- 三氧化二砷 /
- 化疗药物 /
- 硼替佐米 /
- 组蛋白去乙酰化酶抑制剂-辛二酰苯胺异羟肟酸药物协同作用
Abstract:Objective To investigate the in vitro effect of arsenic trioxide (As2O3) alone and in combination with dexamethasone (DXM), etoposide (VP-16), methotrexate (MTX), bortezomib (BTZ), and suberoylanilide hydroxamic acid (SAHA) on the growth of human cutaneous T cell lymphoma (CTCL) cells Hut-78 and Hut-102. Methods Hut-78 and Hut-102 cells were cultured with different concentrations of As2O3, DXM, VP-16, MTX, BTZ, and SAHA alone and As2O3 in combination with DXM, VP-16, MTX, BTZ, or SAHA for 48 h. The effects of the different samples on Hut-78 and Hut-102 cell proliferation were determined by MTT assay. Analyses using CalcuSyn software were performed to determine whether the combination of As2O3 with DXM, VP-16, MTX, BTZ, or SAHA induced synergistic cytoxicity. Results As2O3, DXM, VP-16, MTX, BTZ, and SAHA alone significantly inhibited the growth of Hut-78 and Hut-102 cells in a dose-dependent manner, with a 50% inhibiting concentration of 5 μmol/L, 500 μg/mL, 2.5 μg/mL, 1 μg/mL, 10 μ mol/L, and 2.5 μmol/L individually after 48 h of culture. As2O3 with DXM, VP-16, MTX, BTZ, or SAHA showed remarkable antitumor efficacy compared with that of individual applications. Conclusion As2O3 alone or combined with DXM, VP-16, MTX, BTZ, or SAHA significantly inhibited Hut-78 and Hut-102 cell growth in vitro. This study demonstrated that As2O3 with DXM, VP-16, MTX, BTZ, or SAHA presents synergistic antitumor effects on CTCL cells and may be an optimal regimen in clinical trials of CTCL. -
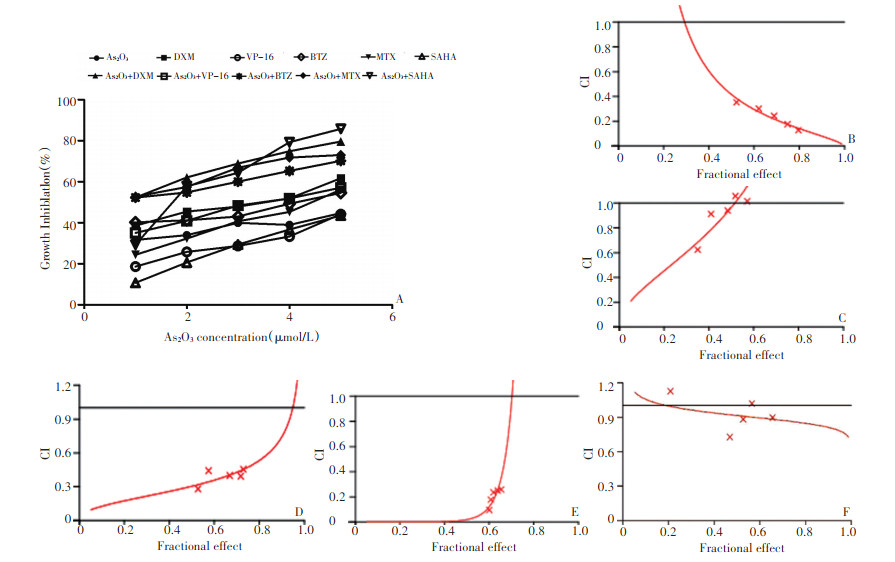
图 1 As2O3与DXM、VP-16、MTX、BTZ、SAHA单药及联合作用于Hut-78的抑制曲线及CI图
Figure 1. Growth inhibition curves of Hut-78 cells cultured in As2O3, DXM, VP-16, MTX, BTZ, SAHA, and As2O3 with DXM, VP-16, MTX, BTZ, or SAHA. The CIs of As2O3 with other drugs were analyzed
▶A: Growth inhibition curves of Hut-78 cultured in different concentrations of As2O3, DXM, VP-16, MTX, BTZ, SAHA and As2O3 with DXM, VP-16, MTX, BTZ, or SAHA; B: Combination index (CI) for Hut-78 cultured in As2O3 with DXM; C: CI for Hut-78 cultured in As2O3 with VP-16; D: CI for Hut-78 cultured in As2O3 with MTX; E: CI for Hut-78 cultured in As2O3 with BTZ; F: CI for Hut-78 cultured in As2O3 with SAHA
-
[1] 李晓武, 张翼鷟.原发皮肤T细胞淋巴瘤治疗的最新进展[J].山东医药, 2012, 52(20):91-94. doi: 10.3969/j.issn.1002-266X.2012.20.036Li XW, Zhang YZ. The latest progress in primary cutaneous T-cell lymphoma[J]. ShanDong Medical Journal, 2012, 52(20): 91-94. doi: 10.3969/j.issn.1002-266X.2012.20.036 [2] Vose JM. The international PTCL Project, international peripheral T-cell lymphoma(PTCL)clinical and pathologic review project: poor outcome by prognostic indices and lack of efficacy with anthracyclines[J]. Blood, 2005, 106(11):611-613. doi: 10.1182/blood.V106.11.611.611 [3] Lengfelder E, Hofmann WK, Nowak D. Impact of arsenic trioxide in the treatment of acute promyelocytic leukemia[J]. Leukemia, 2012, 26(3):433-442. doi: 10.1038/leu.2011.245 [4] Keyhani M. Use of arsenic trioxide as a first-line single agent in the treatment of acute promyelocytic leukemia[J]. J CIin Oncol, 2012, 30(2):217-222. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=fe77afadafc1d2bb33efdd2c7605f486 [5] Hoffman E, Mielicki WP, Arsenic trioxide: impact on the growth and differentiation of cancer cells and possible use in cancer therapy [J]. Postepy Hig Med Dosw(Online), 2013, 67:817-827. doi: 10.5604/17322693.1061640 [6] Emadi A, Gore SD. Arsenic trioxide, an old drug rediscovered[J]. Blood Rev, 2010, 24(4-5):191-199. doi: 10.1016/j.blre.2010.04.001 [7] Gao YH, Zhang HP, Yang SM, Inactivation of Akt by arsenic trioxide induces cell death via mitochondrial-mediated apoptotic signaling in SGC-7901 human gastric cancer cells[J]. Oncol Rep, 2014, 31(4):1645-1652. doi: 10.3892/or.2014.2994 [8] Tun-Kyi A, Qin JZ, Oberholzer PA, et al. Arsenic trioxide down-regulates antiapoptotic genes and induces cell death in mycosis fungoides tumors in a mouse model[J]. Ann Oncol, 2008, 19(8): 1488-1494. doi: 10.1093/annonc/mdn056 [9] 张学美, 王婷, 李晓进, 等.一定浓度下三氧化二砷对T细胞淋巴瘤Hut-78细胞株增殖的影响[J].中国组织工程研究与临床康复, 2011, 15(23):4325-4329. doi: 10.3969/j.issn.1673-8225.2011.23.033Zhang XM, Wang T, Li XJ, et al. Effect of arsenic trioxide on proliferation of T cell lymphoma cell line Hut-78[J]. Journal of Clinical Rehabilitative Tissue Engineering Research, 2011, 15(23): 4325-4329. doi: 10.3969/j.issn.1673-8225.2011.23.033 [10] 钱正子, 王华庆, 付凯, 等.硼替佐米与吡喃阿霉素联合对T细胞淋巴瘤细胞系Hut-78细胞增殖和凋亡的影响[J].中华血液学杂志, 2011, 32(1):47-51. doi: 10.3760/cma.j.issn.0253-2727.2011.01.013Qian ZZ, Wang HQ, Fu K, et al. In vitro synergistic effect of bortezomib and pirarubicin on proliferation and apoptosis of T cell lymphoma cell line Hut-78[J]. Chin J Hematol, 2011, 32(1):47-51. doi: 10.3760/cma.j.issn.0253-2727.2011.01.013 [11] Nakaoka T, Ota A, Ono T, et al. Combined arsenic trioxide-cisplatin treatment enhances apoptosis in oral squamous cell carcinoma cells[J]. Cell Oncol(Dordr), 2014, 37(2):119-129. doi: 10.1007/s13402-014-0167-7 [12] Hideshima T, Catley L, Yasui H, et al. Perifosine, an oral bioactive novel alkylphospholipid, inhibits Akt and induces in vitro and vivo cytotoxicity in human multiple myeloma cells[J]. Blood, 2006, 107 (10):4053-4062. doi: 10.1182/blood-2005-08-3434 [13] Hayashi T, Hideshima T, Akiyama M, et al. Arsenic trioxide inhibits growth of human multiple myeloma cells in the bone marrow microenvironment[J]. Mol Cancer Ther, 2002, 1(10):851-860. [14] Duvic M, Olsen EA, Breneman D, et al. Evaluation of the long-term tolerability and clinical benefit of vorinostat in patients with advanced cutaneous T-cell lymphoma[J]. Clin Lymphoma Myeloma, 2009, 9(6):412-416. doi: 10.3816/CLM.2009.n.082 [15] Frye R, Myers M, Axelrod KC, et al. Romidepsin: a new drug for the treatment of cutaneous T-cell lymphoma[J]. Clin J Oncol Nurs, 2012, 16(2):195-204. http://cn.bing.com/academic/profile?id=aa48fd0648d540e5f0fb6f851c56691a&encoded=0&v=paper_preview&mkt=zh-cn [16] Duvic M, Tetzlaff H, Gangar P, et al. Phase Ⅱtrial of brentuximab vedotin(SGN-35) for CD30 + cutaneous T-cell lymphomas and lymphoproliferative disorders[J]. J Invest Dermatol, 2013, 133:S180. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3873074/ [17] Jia Y, Liu D, Xiao D, et al. Expression of AFP and STAT3 is involved in arsenic trioxide-induced apoptosis and inhibition of proliferation in AFP-producing gastric cancer cells[J]. PLoS One, 2013, 8(1):e54774. doi: 10.1371/journal.pone.0054774 [18] Kodigepalli KM, Dutta PS, Bauckman KA, et al. SnoN/SkiL expression is modulated via arsenic trioxide-induced activation of the PI3K/AKT pathway in ovarian cancer cells[J]. FEBS Lett, 2013, 587 (1):5-16. doi: 10.1016/j.febslet.2012.11.003 [19] Hussein MA. Nontraditional cytotoxic therapies for relapsed/refractory multiple myeloma[J]. Oncologist, 2002, 7(Suppl 1):20-29. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=Open J-Gate000001644905 [20] Hayashi T, Hideshima T, Akiyama M, et al. Arsenic trioxide inhibits growth of human multiple myeloma cells in the bone marrow microenvironment[J]. Mol Cancer Ther, 2002, 1(10):851-860. https://www.researchgate.net/publication/10982501_Arsenic_trioxide_inhibits_growth_of_human_multiple_myeloma_cells_in_the_bone_marrow_microenvironment [21] Zinzani PL, Musuraca G, Tani M, et al. PhaseⅡtrial of proteasome inhibitor bortezomib in patients with relapsed or refractory cutaneous T-cell lymphoma[J]. J Clin Oncol, 2007, 25(27):4293-4297. doi: 10.1200/JCO.2007.11.4207 [22] Marks PA, Discovery and development of SAHA as an anticancer agent[J]. Oncogene, 2007, 26(9):1351-1356. doi: 10.1038/sj.onc.1210204 [23] Zhou Q, Dalgard CL, Wynder C, et al. Histone deacetylase inhibitors SAHA and sodium butyrate block G1-to-S cell cycle progression in neurosphere formation by adult subventricular cells[J]. BMC Neurosci, 2011, 12(1):50-62. http://cn.bing.com/academic/profile?id=e40da1da638bd4eb1e5a5635a44390a1&encoded=0&v=paper_preview&mkt=zh-cn [24] Madeleine D, Youn HK, Timothy MK, et al. Vorinostat (Suberoylanilide Hydroxamic Acid, SAHA)Provides Prolonged Clinical Benefit to Advanced Cutaneous T-Cell Lymphoma Patients:Updated Results of the Phase Ⅱ b Multicenter Clinical Tria1[J]. Blood(ASH Annual Meeting Abstracts), 2006, 108:Abstract 399. [25] Tong A, Zhang H, Li z, et al. Proteomic analysis of liver cancer cells treated with suberoylanilide hydroxamic acid[J]. Cancer Chemother Pharmacol, 2008, 61(5):791-802. doi: 10.1007/s00280-007-0536-2 -




 下载:
下载: