Correlation between activated circulating endothelial cells and efficacy of anti-angiogenic therapy in non-small cell lung cancer patients
-
摘要:
目的 检测非小细胞肺癌(NSCLC)患者外周血中活化血管内皮细胞(aCECs)数量,分析其与抗血管生成疗效的关系,以期寻找能够早期反映抗血管生成疗效的标志物。 方法 将142例NSCLC患者分为化疗组和联合组(化疗+恩度),应用流式细胞术检测患者各个治疗周期外周血中CD105和CD146的表达状态,观察以其标记的aCECs变化趋势并分析其与疗效的相关性。 结果 联合组在第8天、第29天、第2个周期后、第50天、第71天、第4个周期后aCEC均较基线明显升高(P < 0.05),其中疾病进展(progression disease,PD)患者于治疗后aCECs升高更为显著,治疗后较基线升高均有统计学意义(P < 0.05)。治疗周期与治疗前后aCECs差值呈负相关(r=-0.970,P=0.001),治疗后aCECs的变化差值与TTP间呈负相关(r=-0.351,P=0.039)。 结论 CD105和CD146能够反映内皮细胞的活化状态,对药物治疗反应敏感,可作为aCECs的理想标志物;肿瘤进展时aCECs呈上升趋势,抗血管生成有效治疗使其波动下降;检测aCECs数目,可以帮助预判抗血管生成治疗疗效。 Abstract:Objective This study aimed to examine the number of activated circulating endothelial cells (aCECs) in the peripheral blood of patients with non-small cell lung cancer (NSCLC), and investigate the relationship among aCECs, anti-angiogenic therapy, and prognosis of NSCLC patients. This study also aimed to identify novel predictive markers for anti-angiogenic therapy, and provide basic data and experimental basis for establishing an evaluation system for this therapy. Methods A total of 142 NSCLC patients were randomly divided into the chemotherapy group (Group 1) and combined therapy group (i.e., chemotherapy plus endostatin, Group 2). The number of aCECs was measured using flow cytometry by detecting the expression status of CD105 and CD146 in the peripheral blood. The correlation between the changes in aCECs and efficacy of drug treatment was statistically analyzed using SPSS software. Results The number of aCECs in Group 2 increased significantly at 8 and 29 d, two cycles, 50 and 71 d, and four cycles after treatment, respectively (P < 0.05). In particular, aCECs amount in cases of progressive disease increased more significantly after combined therapy (P < 0.05). A negative correlation was found between the treatment cycle and difference in aCECs amount before and after therapy (r= -0.970, P=0.001). A negative correlation was also observed between the difference in aCECs amount and time to tumor progression (TTP) (r=-0.351, P=0.039). Therefore, the difference in aCECs amount before and after therapy could serve as an important predictor for TTP in NSCLC patients. Conclusion CD105 and CD146 reflected the activation status of endothelial cells, and responded to the drug treatment. Thus, CD105 and CD146 could act as ideal markers for aCECs. The number of aCECs increased during cancer progression, but significantly decreased after long-term treatment. Therefore, the change in aCECs amount may be a useful marker in predicting the efficacy of anti-angiogenic therapy. -
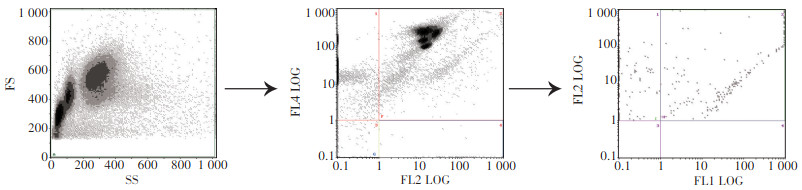
图 1 逻辑设门:在左图中采集10万个外周血细胞为研究背景;中图纵坐标为CD45,横坐标为CD146,其中左上相限和右上相限为CD45阳性细胞即白细胞予以排除,右下相限为CD146单阳性细胞;右图纵坐标为CD146,横坐标为CD105;右图右上相限为CD146、CD105双阳性细胞即活化血管内皮细胞(CD45-CD146+CD105+)
Figure 1. Gating logic: The left illustration shows the gate used to exclude the platelets, dead cells, and debris. We collected 100, 000 peripheral blood cells for study. In the middle illustration, the ordinate was CD45, and the abscissa was CD146. The upper left quadrant and upper right quadrant exhibit leu⁃ kocytes, which were excluded, and the lower right quadrant illustrates CD146+ cells. The ordinate of the right illustration was CD146, and the abscissa was CD105. The upper right quadrant shows CD45+ CD146+ CD105+ cells, namely, aCECs
表 1 aCECs差值与NSCLC患者TTP间的相关性
Table 1. Correlation between △aCECs and TTP

-
[1] Beaudry P, Force J, Naumov GN, et al. Differential effects of vascu lar endothelial growth factor receptor-2 inhibitor ZD6474 on circu lating endothelial progenitors and mature circulating endothelial cells: implications for use as a surrogate marker of antiangiogenic activity[J]. Clin Cancer Res, 2005, 11(9):3514-3522. doi: 10.1158/1078-0432.CCR-04-2271 [2] Mancuso P, Bertolini F. Circulating endothelial cells as biomarkers in clinical oncology[J]. Microvasc Res, 2010, 79(3):224-228. doi: 10.1016/j.mvr.2010.02.007 [3] Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evalua tion criteria in solid tumours: revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2):228-247. http://www.tandfonline.com/servlet/linkout?suffix=CIT0004&dbid=8&doi=10.1080%2F10717544.2018.1440666&key=19097774 [4] Mancuso P, Burlini A, Pruneri G, et al. Resting and activated endo thelial cells are increased in the peripheral blood of cancer patients [J]. Blood, 2001, 97(11):3658-3661. doi: 10.1182/blood.V97.11.3658 [5] Ali AM, Ueno T, Tanaka S, et al. Determining circulating endothe lial cells using Cell Search system during preoperative systemic che motherapy in breast cancer patients[J]. Eur J Cancer, 2011, 47(15): 2265-2272. doi: 10.1016/j.ejca.2011.06.015 [6] Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer [J]. N Engl J Med, 2002, 346(2):92-98. doi: 10.1056/NEJMoa011954 [7] Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer[J]. N Engl J Med, 2006, 355(24):2542-2550. doi: 10.1056/NEJMoa061884 [8] Han B, Xiu Q, Wang H, et al. A multicenter, randomized, dou ble-blind, placebo-controlled study to evaluate the efficacy of paclitaxel-carboplatin alone or with endostar for advanced non-small cell lung cancer[J]. J Thorac Oncol, 2011, 6(6):1104-1109. doi: 10.1097/JTO.0b013e3182166b6b [9] Wang J, Xiao J, Wei X, et al. Circulating endothelial cells and tu mor blood volume as predictors in lung cancer[J]. Cancer Sci, 2013, 104(4):445-452. doi: 10.1111/cas.12097 [10] 李斌, 包婺安, 胡福军, 等.恩度联合化疗治疗转移性鼻咽癌的Ⅱ期临床研究[J].中国肿瘤临床, 2012, 39(24):2022-2025. http://www.cnki.com.cn/Article/CJFDTotal-ZGZL201224015.htmLi B, Bao WA, Hu FJ, et al. A Phase-Ⅱ clinical study on endostar in combination with chemotherapy for metastatic nasopharyngeal carcinoma[J]. Chin J Clin Oncol, 2012, 39(24):2022-2025. http://www.cnki.com.cn/Article/CJFDTotal-ZGZL201224015.htm [11] Li H, Raia V, Bertolini F, et al. Circulating endothelial cells as a therapeutic marker for thalidomide in combined therapy with che motherapy drugs in a human prostate cancer model[J]. BJU Int, 2008, 101(7):884-888. doi: 10.1111/j.1464-410X.2007.07342.x [12] Kawaishi M, Fujiwara Y, Fukui T, et al. Circulating endothelial cells in non-small cell lung cancer patients treated with carboplatin and paclitaxel[J]. J Thorac Oncol, 2009, 4(2):208-213. doi: 10.1097/JTO.0b013e318193030d [13] Tong RT, Boucher Y, Kozin SV, et al. Vascular normalization by vascular endothelial growth factor receptor 2 blockade induces a pressure gradient across the vasculature and improves drug penetra tion in tumors[J]. Cancer Res, 2004, 64(11):3731-3736. doi: 10.1158/0008-5472.CAN-04-0074 [14] Li N, Zheng D, Wei X, et al. Effects of recombinant human end ostatin and its synergy with cisplatin on circulating endothelial cells and tumorvascular normalization in A549 xenograft murine model [J]. J Cancer Res Clin Oncol, 2012, 138(7):1131-1144. doi: 10.1007/s00432-012-1189-z [15] Winkler F, Kozin SV, Tong RT, et al. Kinetics of vascular normal ization by VEGFR2 blockade governs brain tumor response to radi ation: role of oxygenation, angiopoietin-1, and matrix metallopro teinases[J]. Cancer Cell, 2004, 6(6):553-563. http://www.ncbi.nlm.nih.gov/pubmed/15607960 -




 下载:
下载: