-
摘要:
目的 研究Hes1对急性髓系白血病(AML)患者骨髓CD34+CD38-细胞的作用及其机制。 方法 收集初治AML患者及正常供者骨髓样本后,通过密度梯度离心法获取单个核细胞,流式细胞术检测CD34+CD38-细胞比例及其细胞周期。通过免疫磁珠法分选CD34+CD38-细胞后,体外集落形成实验(CFC)检测其增殖能力,并通过Realtime PCR检测其Hes1的表达量。构建Hes1过表达逆转录病毒载体,感染正常供者骨髓CD34+细胞后,流式细胞术分析其细胞周期的改变,CFC检测其增殖的改变。 结果 AML患者骨髓CD34+CD38-细胞比例明显低于正常对照,流式细胞术结果显示患者来源CD34+CD38-细胞大多数进入静止期,CFC结果显示患者CD34+CD38-细胞体外扩增能力下降。Realtime PCR结果发现患者CD34+CD38-细胞中Hes1表达上调。提高正常供者CD34+细胞中Hes1的表达后,细胞增殖减少,进入静止期。 结论 在AML中CD34+CD38-细胞比例下降,进入静止期,与Hes1的表达上调有关。 -
关键词:
- AML白血病 /
- CD34+CD38-细胞 /
- Hes1 /
- 细胞周期 /
- 增殖
Abstract:Objective To determine the effect of Hes1 on bone marrow CD34+ cells in acute myeloid leukemia (AML). Methods Bone marrow mononuclear cells were isolated by using Ficoll. Then, the proportion and cell cycle of CD34+ cells were analyzed by using fluorescence-activated cell sorting (FACS). CD34+ cells were cultured in vitro for colony-forming cells (CFC). The expression of Hes1 in CD34+ cells was evaluated by using real-time polymerase chain reaction. After upregulating the expression of Hes1 in CD34+ cells, the cell cycle was analyzed through FACS, and the colony formation of CD34+Hes1+ cells was analyzed by CFC. Results The ratio of CD34+ cells in the bone marrow was lower in the AML group than in the control group. In addition, more CD34+ cells underwent quiescence in the AML group than in the control group. In vitro assay showed that the colony formation of CD34+ cells was lower in the AML group than in the control group. The expression of Hes1 was higher in the CD34+ cells from the AML patients than that in the CD34+ cells from normal donors. After Hes1 transduction, more CD34+ cells underwent quiescence and showed weak proliferation. Conclusion The proportion of CD34+ cells in the bone marrow was lower in AML patients than in normal donors. A large proportion of CD34+ cells underwent quiescence, which was related to Hes1, in AML patients. -
Key words:
- T-ALL /
- CD34+CD38-cell /
- Hes1 /
- cell cycle /
- proliferation
-
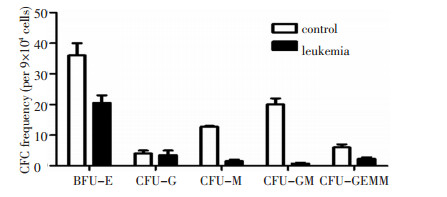
图 1 患者来源CD34+CD38-细胞向各系分化形成的集落数少于对照
Figure 1. CFC test results showed that the clone numbers of CD34+ cells were lower in the AML group than in the control group
BFU-E: burst-forming unit-erythroid; CFU-G: colony-forming unit-granulocyte; CFU-M: colony-forming unit-megakaryocyte; CFU-GM: colony-forming unit-granulocyte macrophage; CFU-GEMM: colony-forming unit-granulocyte, erythroid, macrophage, and monocyte
图 3 Realtime PCR和Western检测Hes1的表达量
Figure 3. Expression of Hes1 was tested through real-time polymerase chain reaction and Western blot
A and B. Relative expression of Hes1 mRNA in T cell acute lymphoblastic leukemia (T-ALL) patients and normal donors compared with the GAPDH gene. C. Relative expression of Hes1 protein in T-ALL patients and normal donors
-
[1] Notta F, Mullighan CG, Wang JC, et al. Evolution of human BCR-ABL1 lymphoblastic leukaemia-initiating cells[J]. Nature, 2011, 469(7330):362-367. doi: 10.1038/nature09733 [2] Coskun S, Hirschi KK. Establishment and regulation of the HSC niche: Roles of osteoblastic and vascular compartments[J]. Birth Defects Res C Embryo Today, 2010, 90(4):229-242. doi: 10.1002/bdrc.20194 [3] Warr MR, Pietras EM, Passegué E. Mechanisms controlling hematopoietic stem cell functions during normal hematopoiesis and hematological malignancies[J]. Wiley Interdiscip Rev Syst Biol Med, 2011, 3(6):681-701. doi: 10.1002/wsbm.145 [4] Chatterjee S, Dutta RK, Basak P, et al. Alteration in marrow stromal microenvironment and apoptosis mechanisms involved in aplastic anemia: an animal model to study the possible disease pathology[J]. Stem Cells Int, 2010, 2010:932354. [5] Notta F, Mullighan CG, Wang JC, et al. Evolution of human BCR-ABL1 lymphoblastic leukaemia-initiating cells[J]. Nature, 2011, 469(7330):362-367. doi: 10.1038/nature09733 [6] Ladipot T, Sirard C, Vormoor J, et al. A cell initiating human acute myeloid leukemia after transplantation in SCID mice[J]. Nature, 1994, 367(6464):645-648. doi: 10.1038/367645a0 [7] de Jonge HJ, Valk PJ, Veeger NJ, et al. High VEGFC expression is associated with unique gene expression profiles and predicts adverse prognosis in pediatric and adult acute myeloid leukemia[J]. Blood, 2010, 116(10):1747-1754. doi: 10.1182/blood-2010-03-270991 [8] Vandercappellen J, Van Damme J, Struyf S. The role of CXC chemokines and their receptors in cancer[J]. Cancer Letters, 2008, 267(2): 226-244. doi: 10.1016/j.canlet.2008.04.050 [9] Kaklamani VG, Sadim M, Hsi A, et al. Variants of the adiponectin and adiponectin receptor 1 genes and breast cancer risk[J]. Cancer Res, 2008, 68(9):3178-3184. doi: 10.1158/0008-5472.CAN-08-0533 [10] Nakayama J, Yamamoto M, Hayashi K, et al. BLNK suppresses pre-B-cell leukemogenesis through inhibition of JAK3[J]. Blood, 2009, 113(7):1483-1492. doi: 10.1182/blood-2008-07-166355 [11] Colmone A, Amorim M, Pontier AL, et al. Leukemic Cells Create Bone Marrow Niches That Disrupt the Behavior of Normal Hematopoietic Progenitor Cells[J]. Science, 2008, 322(5909): 1861-1865. doi: 10.1126/science.1164390 [12] Yuan Y, Yu H, Boyer JM, et al. Hematopoietic stem cells are not the direct target of spontaneous leukemic transformation in p18INK4c-nu ll reconstituted mice[J]. Cancer Res, 2006, 66(1): 343-351. doi: 10.1158/0008-5472.CAN-05-2945 [13] Hu X, Shen H, Tian C, et al. Kinetics of normal hematopoietic stem and progenitor cells in a Notch1-induced leukemia model[J]. Blood, 2009, 114(18):3783-3792. doi: 10.1182/blood-2009-06-227843 [14] Tian C, Zheng G, Cao Z, et al. Hes1 mediates the different responses of hematopoietic stem and progenitor cells to T cell leukemic environment[J]. Cell Cycle, 2013, 12(2):322-331. doi: 10.4161/cc.23160 -




 下载:
下载: