Mechanisms of differentiation of omental-adipose stromal cells promoted by gastric cancer cells
-
摘要:
目的 本实验主要研究在胃癌条件培养基(conditioned medium,CM)诱导下网膜脂肪干细胞(omental-adipose stromal cells,O-ASCs)是否能分化为癌相关成纤维细胞(carcinoma-associated fibroblasts,CAFs),及ERK信号通路在其中的作用。 方法 通过诱导分化成骨、成脂及流式细胞鉴定O-ASCs,将O-ASCs与MGC803和SGC7901 CM共培养,通过RT-PCR和Western-blot检测O-ASCs细胞CAFs标志物α-SMA、FSP-1、vimentin,旁分泌因子VEGFA、TGFβ-1、FAP、SDF-1的表达水平。将O-ASCs分为对照组,SGC7901-CM实验组,SGC7901-CM+U0126处理组,12 h后收集细胞。Western blot检测O-ASCs细胞CAFs标志物α-SMA、FSP-1及ERK1/2、p-ERK1/2的表达水平。 结果 经鉴定原代培养出的细胞为O-ASCs,在SGC7901 CM和MGC803 CM作用下,CAFs标志物α-SMA、FSP-1、vimentin及旁分泌因子SDF-1、VEGFA、TGFβ-1、FAP表达均有明显增加(P < 0.05)。与对照组比较SGC7901-CM组α-SMA、FSP-1、p-ERK1/2表达明显增加(P < 0.05),ERK表达未见明显变化(P > 0.05)。SGC7901-CM+U0126组与SGC7901-CM组比较,α-SMA、FSP-1及p-ERK1/2的蛋白表达水平明显降低(P < 0.05),ERK表达变化无统计学意义(P > 0.05)。 结论 O-ASCs通过分化为CAFs及旁分泌作用参与胃癌腹膜转移,ERK信号通路在该过程中发挥了重要作用。 Abstract:Objective To investigate whether the omental-adipose stromal cells (O-ASCs) exposing to gastric cancer-conditioned medium (CM) could be inducted to differentiate into carcinoma-associated fibroblasts (CAFs) and the effect of ERK signaling pathway in the process. Methods We identified O-ASCs by examining their ability to differentiate osteogenic and adipogenic lineages and through flow cytometry. O-ASCs were co-cultured with MGC803 and SGC7901CM. The expression of CAFs markers (α-SMA, FSP-1, and vimentin) and paracrine factors (VEGFA, TGF-β, FAP, and SDF-1) were evaluated by RT-PCR and Western blot. In vitro cultures of OASCs were divided into three groups: the control, SGC7901-CM, and SGC7901-CM+U0126 groups. Cells were collected after 12 h. Western blot was performed to evaluate the expression of α-SMA, FSP-1, ERK, and p-ERK1/2. Results The primary cells were O-ASCs. The expression levels of CAFs markers (α-SMA, FSP-1, and vimentin) and O-ASC paracrine factors (VEGFA, TGF-β, FAP, and SDF-1) clearly increased (P < 0.05). In comparison with the control, the expression of ERK in SGC7901-CM group did not change (P > 0.05), while the expression of p-ERK1/2, α-SMA, and FSP-1 significantly improved (P < 0.05). Comparison of SGC 7901-CM + U0126 and SGC 7901-CM groups showed that the expression levels of ERK had no statistical difference (P > 0.05), while the expression levels of p-ERK1/2, α-SMA, and FSP-1 decreased (P < 0.05). Conclusion O-ASCs participate in the peritoneal metastasis of gastric cancer through differentiation by CAF and paracrine factors. The ERK signaling pathway is important in the differentiation of O-ASCs towards CAFs. -
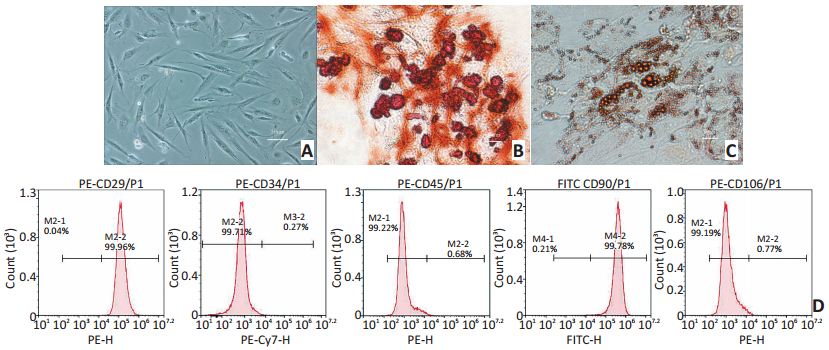
图 1 O-ASCs的培养及鉴定
Figure 1. Culture and identification of O-ASCs
A. Morphological analysis with passage 2 O-ASCs by the light microscope (200×); B. After adipogenic and osteogenic induction for 21 days, alizarin redstaining showed that typical calcified nodules were indicated in red-brown (400×); C. After adipogenic induction for 21 days, oil red O-staining showed red-brown fat droplets of uneven sizes (400×); D. Expression of stromal markers on the cellular membrane of O-ASCs analyzed by flow cytometry
图 3 胃癌CM促进O-ASCs分化为CAFs
Figure 3. Gastric cancer CM-induced differentiation of O-ASCs towards CAFs
A. In comparison with the control group, expression of α-SMA, FSP-1, and vimentin in SGC7901-CM and MGC803-CM groups significantly increased at the RNA level; B. Confirmation of the trend by Western blot; C. Histogram representations of quantitative Western blot (n=3). *P < 0.01, **P < 0.05
图 4 胃癌CM促进O-ASCs相关旁分泌因子增加
Figure 4. Gastric cancer CM promoted the expression of paracrine factors
A. In comparison with the control group, expression of SDF-1, VEGFA, TGF-β1, and FAP in SGC7901-CM and MGC803-CM groups significantly increased at the RNA level; B. Western blot of VEGFA and TGF-β1 showed that expression levels of paracrine factors were higher in SGC 7901-CM and MGC803-CM groups than in the control group; C. Histogram representations of quantitative Western blot (n=3). *P < 0.01, **P < 0.05
图 5 O-ASCs在U0126作用后向CAFs分化受抑制
Figure 5. O-ASC differentiation towards CAFs inhibited by U0126
▶A. In comparison with the control group, the expression of ERK in SGC7901-CM group did not show a significant change, whereas the expression levels of p-ERK1/2, α-SMA and FSP-1 were significantly increased. No significant difference was observed in the expression of ERK between the SGC 7901-CM + U0126 and SGC7901-CM groups, whereas the expression levels of p-ERK1/2, α-SMA, and FSP-1 were decreased; B. Histogram representations of quantitative Western blots. (n=3). *P < 0.01, #P > 0.05
表 1 引物序列
Table 1. Primer sequences

-
[1] Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2):115-132. doi: 10.3322/caac.21338 [2] 武海涛, 姬忠贺, 张倩, 等.洛铂联合多西他赛行肿瘤细胞减灭术加腹腔热灌注化疗治疗同时性胃癌腹膜癌[J].中国肿瘤临床, 2016, 43(4): 146-151. http://www.cjco.cn/CN/abstract/abstract13298.shtmlWu HT, Ji ZH, Zhang Q, et al. Treatment of synchronous peritoneal carcinomatosis fromgastric cancer with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy with lobaplatin and docetaxel[J]. Chin J Clin Oncol, 2016, 43(4):146-151. http://www.cjco.cn/CN/abstract/abstract13298.shtml [3] 李雁, 杨国梁, 杨肖军.细胞减灭术加腹腔热灌注化疗治疗腹膜种植瘤的研究进展[J].中国肿瘤临床, 2007, 34(21): 1257-1260. doi: 10.3969/j.issn.1000-8179.2007.21.017Li Y, Yang GL, Yang XJ, et al. Cytoreductive Surgery with Intraoperative Peritoneal Hyperthermo-chemotherapy for Peritoneal Carcinomatosis [J]. Chin J Clin Oncol, 2007, 34(21):1257-1260. doi: 10.3969/j.issn.1000-8179.2007.21.017 [4] Jotzu C, Alt E, Welte G, et al. Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors[J]. Cell Oncol (Dordr), 2011, 34(1): 55-67. doi: 10.1007/s13402-011-0012-1 [5] Muehlberg FL, Song YH, Krohn A, et al. Tissue-resident stem cells promote breast cancer growth and metastasis[J]. Carcinogenesis, 2009, 30(4):589-597. doi: 10.1093/carcin/bgp036 [6] Zhang C, Rexrode KM, Van DRM, et al. Abdominal obesity and the risk of all-cause, cardiovascular, and cancer mortality: sixteen years of follow-up in US women[J]. Circulation, 2008, 117(13):1658-1667. doi: 10.1161/CIRCULATIONAHA.107.739714 [7] Zhao BC, Zhao B, Han JG, et al. Adipose-derived stem cells promote gastric cancer cell growth, migration and invasion through SDF-1/ CXCR4 axis[J]. Hepatogastroenterology, 2010, 57(104):1382-1389. https://www.researchgate.net/publication/50890226_Adipose-Derived_Stem_Cells_Promote_Gastric_Cancer_Cell_Growth_Migration_and_Invasion_Through_SDF-1CXCR4_Axis [8] Rezaeifard S, Razmkhah M, Robati M, et al. Adipose derived stem cells isolated from omentum: a novel source of chemokines for ovarian cancer growth[J]. J Cancer Res Ther, 2014, 10(1):159-164. doi: 10.4103/0973-1482.131451 [9] Chu Y, Tang H, Guo Y, et al. Adipose-derived mesenchymal stem cells promote cell proliferation and invasion of epithelial ovarian cancer[J]. Exp Cell Res, 2015, 337(1):16-27. doi: 10.1016/j.yexcr.2015.07.020 [10] Salimian RB, Caneba C, Nowicka A, et al. Nitric oxide mediates metabolic coupling of omentum-derived adipose stroma to ovarian and endometrial cancer cells[J]. Cancer Res, 2015, 75(2):456-471. doi: 10.1158/0008-5472.CAN-14-1337 [11] Kalluri R, Zeisberg M. Fibroblasts in cancer[J]. Nature Reviews Cancer, 2006, 6(5):392-401. doi: 10.1038/nrc1877 [12] Yamaguchi H, Sakai R. Direct Interaction between Carcinoma Cells and Cancer Associated Fibroblasts for the Regulation of Cancer Invasion[J]. Cancers (Basel), 2015, 7(4):2054-2062. doi: 10.3390/cancers7040876 [13] Rasmussen JG, Frobert O, Pilgaard L, et al. Prolonged hypoxic culture and trypsinization increase the pro-angiogenic potential of human adipose tissue-derived stem cells[J]. Cytotherapy, 2011, 13(3):318-328. doi: 10.3109/14653249.2010.506505 [14] Willis BC, Borok Z. TGF-beta-induced EMT: mechanisms and implications for fibrotic lung disease[J]. Am J Physiol Lung Cell Mol Physiol, 2007, 293(3):L525-534. doi: 10.1152/ajplung.00163.2007 [15] Cohen SJ, Alpaugh RK, Palazzo I, et al. Fibroblast activation protein and its relationship to clinical outcome in pancreatic adenocarcinoma[J]. Pancreas, 2008, 37(2):154-158. doi: 10.1097/MPA.0b013e31816618ce [16] Cho JA, Park H, Lim EH, et al. Exosomes from breast cancer cells can convert adipose tissue-derived mesenchymal stem cells into myofibroblast-like cells[J]. Int J Oncol, 2012, 40(1):130-138. doi: 10.3892/ijo.2011.1193/download [17] Cho HH, Kyoung KM, Seo MJ, et al. Overexpression of CXCR4 increases migration and proliferation of human adipose tissue stromal cells[J]. Stem Cells Dev, 2006, 15(6):853-864. doi: 10.1089/scd.2006.15.853 -




 下载:
下载: