Prognostic value of the combined preoperative plasma levels of fibrinogen and lymphocyte to monocyte ratio (F-LMR) in patients with non-small cell lung cancer
-
摘要:
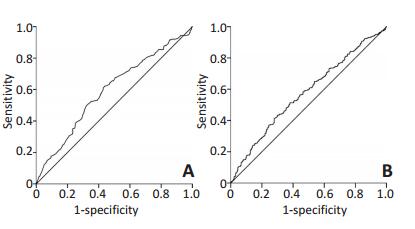
目的 探讨术前纤维蛋白原(fibrinogen,Fbg)联合淋巴细胞与单核细胞比值(lymphocyte to monocyte ratio,LMR)评分(FLMR)与非小细胞肺癌(non-small cell lung cancer,NSCLC)患者预后的相关性。 方法 回顾性分析2006年1月至2009年12月天津医科大学肿瘤医院行根治性手术切除的589例NSCLC患者的临床病例资料。根据受试者工作特征(ROC)曲线判定Fbg及LMR的界限值,根据F-LMR评分原则将患者分为3组:F-LMR 0分、F-LMR 1分、F-LMR 2分。通过χ2检验分析其与患者临床病理因素的关系,Kaplan-Meier法进行生存分析,运用Log-rank法进行差异性检验,Cox比例风险模型进行单因素与多因素回归分析。 结果 根据ROC曲线,3.48 g/L、3.23分别作为Fbg和LMR的界值。F-LMR 0分215例、F-LMR 1分228例、F-LMR 2分146例,术前F-LMR评分与年龄、性别、吸烟史、肿瘤位置、手术类型、病理分期、病理类型、肿瘤大小有显著相关(P < 0.05)。单因素分析显示:肿瘤位置、手术类型、病理分期、肿瘤大小、F-LMR评分、LMR及Fbg水平是影响NSCLC患者预后的危险因素(P < 0.05);多因素分析结果表明:病理分期(DFS:HR:1.700,95%CI:1.483~1.950,P < 0.001;OS:HR:1.703,95%CI:1.486~1.952,P < 0.001)和F-LMR评分(DFS:HR:1.264,95%CI:1.077~1.484,P=0.004;OS:HR:1.301,95%CI:1.107~1.528,P=0.001)是影响患者预后的独立危险因子。 结论 术前F-LMR评分可作为预测行根治性切除的非小细胞肺癌患者预后的指标。 -
关键词:
- 非小细胞肺癌 /
- 纤维蛋白原 /
- 淋巴细胞与单核细胞比例 /
- F-LMR评分 /
- 预后
Abstract:Objective This study aims to evaluate the correlation of combined preoperative plasma levels of fibrinogen (Fbg) and lymphocyte to monocyte ratio (LMR) (F-LMR) with the prognosis of patients with non-small cell lung cancer (NSCLC) after complete resection. Methods The clinical data of 589 patients with NSCLC who underwent complete resection in our hospital were retrospectively analyzed. Receiver operating characteristic curve (ROC) analysis was used to select the cut-off values of Fbg and LMR. Based on the criteria of F-LMR, we divided the patients into three groups: F-LMR 0 score, F-LMR 1 score, and F-LMR 2 score. The association between F-LMR and the clinicopathological characteristics was analyzed by the χ2 test. Kaplan-Meier analysis was used to analyze the prognostic factors, and the log-rank test was used to determine the differences in survival rates. Prognostic factors were assessed by univariate and mu#60ivariate analyses (Cox's proportional hazards regression model). Results According to the ROC curve, the cut-off values of Fbg and LMR were 3.48 g/L and 3.23, respectively. F-LMR 0 score had n=215, F-LMR 1 score had n=228, and F-LMR 2 score had n=146. Preoperative F-LMR was closely related to age, gender, smoking history, tumor location, surgical type, pathological stage, pathological type, and tumor size (P < 0.05). Univariate analysis showed that tumor location, surgical type, pathological stage, tumor size, F-LMR score, LMR, and Fbg were associated with survival (P < 0.05). Mu#60ivariate analysis showed that the pathological stage [disease-free survival (DFS): hazard ratio (HR)= 1.700, 95% confidence interval (CI)= 1.483-1.950, P < 0.001; overall survival (OS): HR= 1.703, 95% CI=1.486-1.952, P < 0.001] and F-LMR score (DFS: HR= 1.264, 95% CI= 1.077-1.484, P=0.004; OS: HR= 1.301, 95% CI= 1.107-1.528, P=0.001) were the independent prognostic factors of NSCLC patients. Conclusion The preoperative F-LMR score may be a useful blood marker for predicting the prognosis of patients with NSCLC with radical resection. -
Key words:
- non-small cell lung cancer, /
- fibrinogen /
- ymphocyte to monocyte ratio /
- F-LMR score /
- prognosis
-
图 2 The correlation between preoperative F-LMR score and clinicopathologic characteristics of patients with NSCLC n (%)
Figure 2. The correlation between preoperative LMR or Fbg level and prognosis of patients with NSCLC
A.Survival curve of patients for DFS in LMR; B. Survival curve of patients for OS in LMR; C. Survival curve of patients for DFS in Fbg; D. Survival curve of patients for OS in Fbg
表 1 非小细胞肺癌患者术前F-LMR评分与临床病理特征的相关性 例数(%)
Table 1. The correlation between preoperative F-LMR score and clinicopathologic characteristics of patients with NSCLC n (%)

表 2 影响肺癌预后的单因素Cox回归分析
Table 2. Univariate Cox regression analysis of prognostic factors in patients with NSCLC

表 3 影响肺癌预后的多因素Cox回归分析
Table 3. Multivariate Cox regression analysis of prognostic factors in patients with NSCLC

-
[1] Siegel R, Ma J, Zou Z, et al. Cancer statistics, 2014[J]. CA Cancer J Clin, 2014, 64(1):9-29. doi: 10.3322/caac.21208 [2] Bystricky B, Reuben JM, Mego M. Circulating tumor cells and coagulation-Minireview[J]. Crit Rew Oncol Hematol, 2017, 114:33-42. doi: 10.1016/j.critrevonc.2017.04.003 [3] Mantovani A, Allavena P, Sica A, et al. Cancer-related inflammation[J]. Nature, 2008, 454(7203):436-444. doi: 10.1038/nature07205 [4] Jiang HG, Li J, Shi SB, et al. Value of fibrinogen and D-dimer in predicting recurrence and metastasis after radical surgery for nonsmall cell lung cancer[J]. Med Oncol, 2014, 31 (7):22. doi: 10.1007/s12032-014-0022-8 [5] Suzuki T, Shimada H, Nanami T, et al. Hyperfibrinogenemia is associated with inflammatory mediators and poor prognosis in patients with gastric cancer[J]. Surg Today, 2016, 46(12):1394-1401. doi: 10.1007/s00595-016-1339-z [6] Zhang D, Zhou X, Bao W, et al. Plasma fibrinogen levels are correlated with postoperative distant metastasis and prognosis in esophageal squamous cell carcinoma[J]. Oncotarget, 2015, 6(35):38410-38420. doi: 10.18632/oncotarget.v6i35 [7] Sun ZQ, Han XN, Wang HJ, et al. Prognostic significance of preoperative fibrinogen in patients with colon cancer[J]. World J Gastroenterol, 2014, 20(26):8583-8591. doi: 10.3748/wjg.v20.i26.8583 [8] Hu P, Shen H, Wang G, et al. Prognostic Significance of systemic inflammation-based lymphocyte-monocyte ratio in patients with lung cancer: based on a large cohort study[J]. PLoS One, 2014, 9(9):e108062. http://europepmc.org/abstract/MED/25275631 [9] Li J, Jiang R, Liu WS, et al. A large cohort study reveals the association of elevated peripheral blood lymphocyte-to-monocyte ratio with favorable prognosis in nasopharyngeal carcinoma[J]. PLoS One, 2013, 8(12):e83069. doi: 10.1371/journal.pone.0083069 [10] Stotz M, Szkandera J, Stojakovic T, et al. The lymphocyte to monocyte ratio in peripheral blood represents a novel prognostic marker in patients with pancreatic cancer[J]. Clin Chem Lab Med, 2015, 53(3):499-506. http://pubmed.cn/25389993 [11] Brambilla E, Travis WD, Colby TV, et al. The new World Hea#60h Organization classification of lung tumors[J]. Eur Respir J, 2001, 18(6):1059-1068. doi: 10.1183/09031936.01.00275301 [12] Detterbeck FC, Boffa DJ, Tanoue LT. The new lung cancer staging system[J]. Chest, 2009, 136(1):260-271. doi: 10.1378/chest.08-0978 [13] Goad KE, Gralnick HR.Coagulation disorders in cancer[J]. Hematol Oncol Clin North Am, 1996, 10(2):457-484. doi: 10.1016/S0889-8588(05)70347-6 [14] Marinho FC, Takagaki TY. Hypercoagulability and lung cancer[J]. J Bras Pneumol, 2008, 34(5):312-322. doi: 10.1590/S1806-37132008000500011 [15] Tian Y, Hong M, Jing S, et al. Clinical and prognostic effect of plasma fibrinogen in renal cell carcinoma: A meta-analysis[J]. Biomed Res Int, 2017, DOI: 10.1155/2017/9591506. [16] Yamaguchi T, Yamamoto Y, Yokota S, et al. Involvement of interleukin-6 in the elevation of plasma fibrinogen levels in lung cancer patients[J]. Jpn J Clin Oncol, 1998, 28(12):740-744. doi: 10.1093/jjco/28.12.740 [17] 高翠红, 高庆双, 彭利红.卵巢癌患者外周血D-二聚体、FIB水平与IL-6动态变化的研究[J].中国免疫学杂志, 2015, 31(4):534-536. http://www.cnki.com.cn/Article/CJFDTOTAL-ZMXZ201504023.htmGao CH, Gao QS, Peng LH. Study of dynamic changes of peripheral blood D-dimer, IL-6 and Fibrinogen of patients with ovarian cancer[J]. Chinese Journal of Immunology, 2015, 31(4):534-536. http://www.cnki.com.cn/Article/CJFDTOTAL-ZMXZ201504023.htm [18] Sahni A, Simpson-Haidaris PJ, Sahni SK, et al. Fibrinogen synthesized by cancer cells augments the proliferative effect of fibroblast growth factor-2 (FGF-2)[J]. J Thromb Haemost, 2008, 6(1):176-183. doi: 10.1111/j.1538-7836.2007.02808.x/epdf [19] Sahni A, Francis CW. Vascular endothelial growth factor binds to fibrinogen and fibrin and stimulates endothelial cell proliferation[J].Blood, 2000, 96(12):3772-3778. http://www.bloodjournal.org/content/104/2/409?sso-checked=1 [20] Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation[J]. Cell, 2011, 144(5):646-674. doi: 10.1016/j.cell.2011.02.013 [21] Petruzzi MN, Cherubini K, Salum FG, et al. Role of tumour-associated macrophages in oral squamous cells carcinoma progression: an update on current knowledge[J]. Diagn Pathol, 2017, 12(1):32. doi: 10.1186/s13000-017-0623-6 [22] Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer[J]. Cell, 2010, 140(6):883-899. doi: 10.1016/j.cell.2010.01.025 [23] Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis[J]. Cell, 2010, 141(1):39-51. doi: 10.1016/j.cell.2010.03.014 [24] Douglas E, McMillan DC. Towards a simple objective framework for the investigation and treatment of cancer cachexia: The Glasgow Prognostic Score[J]. Cancer Treat Rev, 2014, 40(6):685-691. doi: 10.1016/j.ctrv.2013.11.007 -




 下载:
下载: