Berberine exerts pro-apoptotic effects on PC-9 cells via activation of JNK/FOXO3 signaling
-
摘要:
目的 探讨黄连素(berberine,Ber)诱导肺腺癌PC-9细胞凋亡及c-Jun氨基末端激酶(c-Jun N-terminal kinase,JNK)/转录因子叉头蛋白3(forkhead box protein O3,FOXO3)信号通路的作用机制。 方法 实验采用完全随机化的分组方法,分为对照组、Ber组(30、60 μM),分别检测各组PC-9细胞活力值、凋亡率、ROS含量、caspase 3活性、线粒体膜电位,以及JNK/FOXO3通路和凋亡相关蛋白含量的变化。SP600125特异性抑制JNK(磷酸化)激活后,Ber(0、60 μM)处理细胞24 h,重复上述检测。 结果 Ber有效抑制PC-9细胞活力,促进细胞凋亡(P < 0.05),显著降低PC-9细胞线粒体膜电位,增加ROS含量和caspase 3活性(P < 0.05),并呈浓度依赖效应;Western blot检测结果显示Ber上调p-JNK、FOXO3和Bax的含量(P < 0.05),下调p-FOXO3和Bcl-2的含量(P < 0.05);SP600125特异性抑制JNK激活后,拮抗Ber下调p-FOXO3含量及其促PC-9细胞凋亡作用(P < 0.05)。 结论 Ber可有效抑制肺腺癌PC-9细胞活力、促进细胞凋亡和氧化应激损伤,作用机制可能与上调p-JNK、FOXO3含量和抑制FOXO3磷酸化有关。 Abstract:Objective To investigate the pro-apoptotic effect of berberine (Ber) on human lung adenocarcinoma PC-9 cell line, and to detect the role of c-Jun N-terminal kinase (JNK)/forkhead box protein O3 (FOXO3) signaling in this process. Methods The PC-9 cells were randomly divided into the control group and the Ber group, which was treated with 30 and 60 μM Ber. The survival rate, apoptotic rate, ROS generation, caspase-3 activity, and mitochondrial membrane potential of cells were detected. Western blot was performed to detect the expression of JNK/FOXO3 signaling and apoptosis-related proteins. A JNK-specific activation inhibitor, SP600125, was used to block the phosphorylation of FOXO3 in PC-9 cells, and then treated with Ber (30 and 60 μM) for further detection after 24h. Results Ber treatment resulted in an obvious reduction in cell viability, promotion of cell apoptosis, downregulation of mitochondrial membrane potential, and an increase of ROS and caspase-3 in a dose-dependent manner. Western blot analysis demonstrated that Ber treatment resulted in a significant upregulation of p-JNK, FOXO3, and Bax expression, and a downregulation of p-FOXO3 and Bcl2 levels. Moreover, the inhibition of JNK activation by SP600125 antagonized the anti-FOXO3 phosphorylation role and the pro-apoptotic role of Ber on PC-9 cells. Conclusion Ber treatment effectively inhibits the viability of PC-9 cells and enhances apoptosis and oxidative stress injury, which may be related to the upregulation of p-JNK and FOXO3 and the downregulation of p-FOXO3. -
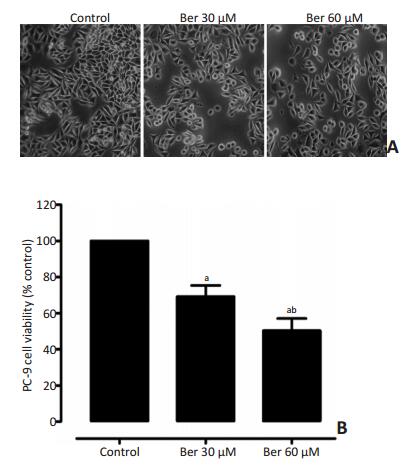
图 1 不同浓度Ber处理24 h抑制PC-9细胞活力
Figure 1. Ber treatment inhibited PC-9 cell viability (24h)
A.Effect of Ber treatment on the morphology of PC9 cells (24 h, 200× resolution); B. Analyses of cell viability detected by MTT assay. The control group is 100%. aP<0.05, versus control, abP<0.05, versus Ber, 30 μM
图 2 不同浓度Ber处理24 h对各组细胞凋亡率的影响
Figure 2. Effect of Ber treatment on the apoptosis of PC9 cells (24 h)
A.Figures of TUNEL assay captured by a confocal microscope, displaying the cell nucleus (blue) and the apoptotic cells (green) at the lower panel; B. Analyses of apoptotic index. aP<0.05, versus control, abP<0.05, versus Ber, 30 μM
图 3 不同浓度Ber处理24 h对各组细胞JC-1线粒体膜电位、ROS含量及caspase3活性的影响
Figure 3. Effect of Ber treatment on JC-1 mitochondrial membrane potential, ROS content, and caspase-3 activity of PC9 cells (24 h)
A.Effect of Ber treatment on the JC-1 mitochondrial membrane potential (MMP) of PC9 cells (24 h). Green regions represent low MMP and red regions represent high MMP. The upper right figure shows the ratio of low MMP; B, C. Analyses of ROS content and caspase-3 activity. The control group is 100%. aP<0.05 vs. control; abP<0.05 vs. Ber (30 μM)
-
[1] Vuddanda PR, Chakraborty S, Singh S. Berberine: a potential phytochemical with multispectrum therapeutic activities[J]. Expert Opin Investig Drugs, 2010, 19(10):1297-1307. doi: 10.1517/13543784.2010.517745 [2] Wang N, Tan HY, Li L, et al. Berberine and Coptidis Rhizoma as potential anticancer agents: Recent updates and future perspectives[J]. J Ethnopharmacol, 2015, 176:35-48. doi: 10.1016/j.jep.2015.10.028 [3] Li-Weber M. Targeting apoptosis pathways in cancer by Chinese medicine[J]. Cancer Lett, 2013, 332(2):304-312. doi: 10.1016/j.canlet.2010.07.015 [4] Ortiz LM, Lombardi P, Tillhon M, et al. Berberine, an epiphany against cancer[J]. Molecules, 2014, 19(8):12349-12367. doi: 10.3390/molecules190812349 [5] Coomans de Brachene A, Demoulin JB. FOXO transcription factors in cancer development and therapy[J]. Cell Mol Life Sci, 2016, 73(6):1159-1172. doi: 10.1007/s00018-015-2112-y [6] Bullock M. FOXO factors and breast cancer: outfoxing endocrine resistance[J]. Endocr Relat Cancer, 2016, 23(2):R113-130. [7] Liu J, Lin A. Role of JNK activation in apoptosis: a double-edged sword[J]. Cell Res, 2005, 15(1): 36-42. doi: 10.1038/sj.cr.7290262 [8] Wagner EF, Nebreda AR. Signal integration by JNK and p38 MAPKpathways in cancer development[J]. Nat Rev Cancer, 2009, 9(8):537-549. doi: 10.1038/nrc2694 [9] Shukla S, Rizvi F, Raisuddin S, et al. FoxO proteins' nuclear retention and BH3-only protein Bim induction evoke mitochondrial dysfunction-mediated apoptosis in berberine-treated HepG2 cells[J].Free Radic Biol Med, 2014, 76:185-199. doi: 10.1016/j.freeradbiomed.2014.07.039 [10] Xie J, Xu Y, Huang X, et al. Berberine-induced apoptosis in human breast cancer cells is mediated by reactive oxygen species generation and mitochondrial-related apoptotic pathway[J]. Tumour Biol, 2015, 36(2):1279-1288. doi: 10.1007/s13277-014-2754-7 [11] Pham TP, Kwon J, Shin J. Berberine exerts anti-adipogenic activity through up-regulation of C/EBP inhibitors, CHOP and DEC2[J]. Biochem Biophys Res Commun, 2011, 413(2):376-382. doi: 10.1016/j.bbrc.2011.08.110 [12] Eom KS, Kim HJ, So HS, et al. Berberine-induced apoptosis in human glioblastoma T98G cells is mediated by endoplasmic reticulum stress accompanying reactive oxygen species and mitochondrial dysfunction[J]. Biol Pharm Bull, 2010, 33(10):1644-1649. doi: 10.1248/bpb.33.1644 [13] Zheng F, Tang Q, Wu J, et al. p38alpha MAPK-mediated induction and interaction of FOXO3a and p53 contribute to the inhibitedgrowth and induced-apoptosis of human lung adenocarcinoma cells by berberine[J]. J Exp Clin Cancer Res, 2014, 33:36. doi: 10.1186/1756-9966-33-36 [14] Martins R, Lithgow GJ, Link W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity[J]. Aging Cell, 2016, 15(2):196-207. doi: 10.1111/acel.2016.15.issue-2 [15] Wang X, Chen WR, Xing D. A pathway from JNK through decreased ERK and Akt activities for FOXO3a nuclear translocation in response to UV irradiation[J]. J Cell Physiol, 2012, 227(3):1168-1178 doi: 10.1002/jcp.22839 -




 下载:
下载: