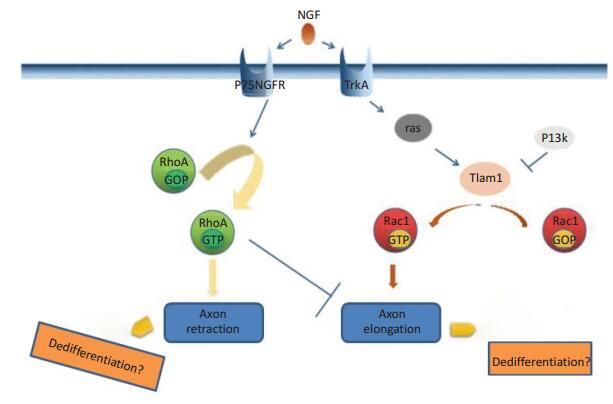
-
摘要: 随着分子诊断技术的不断发展,精准治疗已进入恶性肿瘤的治疗范畴,靶向治疗是近年来较为热门的研究方向。神经母细胞瘤(neuroblastoma,NB)是儿童期最常见的颅外实体瘤。研究表明,很多基因参与NB的发生发展过程,T淋巴瘤侵袭转移诱导因子1(T lymphoma invasion and metastasis inducing factor 1,TIAM1)是其中之一。TIAM1主要与下游的RAC1(Ras-related C3 botu?linum toxin substrate 1)结合,作用于TrkA/TIAM1/RAC1通路,激活下游相关因子,参与调节神经元轴突的分化过程。因此,深入的研究和实验或许可以更透彻的阐明具体的机制,为NB未来的诊疗提供一个新的方向。Abstract: With continuous development of molecular diagnosis, "precision treatment" has entered the therapeutic category for malignant tumors, and targeted gene therapy has been an intense research topic in recent years. Neuroblastoma is the most common extracranial solid tumor that develops during childhood. Studies show that many genes, such as TIAM1, are involved in the development and progression of neuroblastoma. TIAM1 mainly combines with RAC1 to activate downstream factors that mediate differentiation via the TrkA/TIAM1/RAC1 signaling pathway, which is involved in the regulation of neurite. Therefore, further studies and experiments may reveal the specific mechanisms and provide a new direction for the future treatment and development of neuroblastoma.
-
Key words:
- neuroblastoma /
- children /
- TIAM1 /
- differentiation /
- research progress
-
表 1 TIAM1主要结构域的基本功能及信号通路
Table 1. The basic functions and signaling pathways of the main domain of the TIAM1

-
[1] Coughlan D, Gianferante M, Lynch CF, et al. Treatment and survival of childhood neuroblastoma: evidence from a population-based study in the United States[J]. Pediatr Hematol Oncol, 2017:1-11. [2] He J, Zou Y, Wang T, et al. Genetic variations of GWAS-identified genes and neuroblastoma susceptibility: a replication study in southern Chinese children[J]. Transl Oncol, 2017, 10(6):936-941. doi: 10.1016/j.tranon.2017.09.008 [3] Li Z, Yu X, Wang Y, et al. By downregulating TIAM1 expression, microRNA-329 suppresses gastric cancer invasion and growth[J]. Oncotarget, 2015, 6(19):17559-17569. doi: 10.18632/oncotarget.v6i19 [4] Chen G, Lu L, Liu C, et al. MicroRNA-377 suppresses cell proliferation and invasion by inhibiting TIAM1 expression in hepatocellular carcinoma[J]. PLoS One, 2015, 10(3):e0117714. doi: 10.1371/journal.pone.0117714 [5] Engers R, Mueller M, Walter A, et al. Prognostic relevance of Tiam1 protein expression in prostate carcinomas[J]. Br J Cancer, 2006, 95 (8):1081-1086. doi: 10.1038/sj.bjc.6603385 [6] Guo X, Wang M, Zhao Y, et al. Par3 regulates invasion of pancreatic cancer cells via interaction with Tiam1[J]. Clin Exp Med, 2016, 16 (3):357-365. doi: 10.1007/s10238-015-0365-2 [7] Li Z, Liu Q, Piao J, et al. Clinicopathological implications of Tiam1 overexpression in invasive ductal carcinoma of the breast[J]. BMC Cancer, 2016, 16(1):681. doi: 10.1186/s12885-016-2724-0 [8] Wang S, Li S, Tang Q, et al. Overexpression of Tiam1 promotes the progression of laryngeal squamous cell carcinoma[J]. Oncol Rep, 2015, 33(4):1807-1814. doi: 10.3892/or.2015.3785 [9] Liu S, Li Y, Qi W, et al. Expression of Tiam1 predicts lymph node metastasis and poor survival of lung adenocarcinoma patients[J]. Diagn Pathol, 2014, (9):69. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3973616/ [10] Li H, Cui X, Chen D, et al. Clinical implication of Tiam1 overexpression in the prognosis of patients with serous ovarian carcinoma[J]. Oncol Lett, 2016, 12(5):3492-3498. doi: 10.3892/ol.2016.5091/download [11] Habets GG, Ra VDK, Jenkins NA, et al. The invasion-inducing TIAM1 gene maps to human chromosome band 21q22 and mouse chromosome 16[J]. Cytogene Geno Res, 2008, 70(1-2):48-51. https://content.karger.com/Article/Abstract/133989 [12] Xu Z, Gakhar L, Bain FE, et al. The Tiam1 guanine nucleotide exchange factor is auto-inhibited by its pleckstrin homology coiledcoil extension domain[J]. 2017, 292(43):17777-17793. [13] Yoo S, Kim Y, Lee H, et al. A gene trap knockout of the Tiam-1 protein results in malformation of the early embryonic brain[J]. Mole Cells, 2012, 34(1):103-108. doi: 10.1007/s10059-012-0119-x [14] Boissier P, Huynhdo U. The guanine nucleotide exchange factor Tiam1: a janus-faced molecule in cellular signaling[J]. Cellular Signalling, 2014, 26(3):483-491. doi: 10.1016/j.cellsig.2013.11.034 [15] Xu Z, Gakhar L, Bain FE, et al. The Tiam1 guanine nucleotide exchange factor is auto-inhibited by its pleckstrin homology coiledcoil extension domain[J]. 2017. [Epub ahead of print]. [16] Stam JC, Sander EE, Michiels F, et al. Targeting of Tiam1 to the plasma membrane requires the cooperative function of the N-terminal pleckstrin homology domain and an adjacent protein interaction domain[J]. J Biol Chem, 1997, 272(45):28447-28454. doi: 10.1074/jbc.272.45.28447 [17] Crompton AM, Foley LH, Wood A, et al. Regulation of Tiam1 nucleotide exchange activity by pleckstrin domain binding ligands[J]. J Biol Chem, 2000, 275(33):25751-25759. doi: 10.1074/jbc.M002050200 [18] Worthylake DK, Rossman KL, Sondek J. Crystal structure of Rac1 in complex with the guanine nucleotide exchange region of Tiam1[J]. Nature, 2000, 408(6813):682-688. doi: 10.1038/35047014 [19] Shepherd TR, Klaus SM, Liu X, et al. The Tiam1 PDZ domain couples to Syndecan1 and promotes cell-matrix adhesion[J]. J Mol Biol, 2010, 398(5):730-746. doi: 10.1016/j.jmb.2010.03.047 [20] Fard SS, Kele J, Vilar M, et al. Tiam1 as a signaling mediator of nerve growth factor-dependent neurite outgrowth[J]. PLoS One, 2010, 5 (3):e9647. doi: 10.1371/journal.pone.0009647 [21] Kawauchi T, Chihama K, Nabeshima Y, et al. The in vivo roles of STEF/Tiam1, Rac1 and JNK in cortical neuronal migration[J]. Embo J, 2003, 22(16):4190-4201. doi: 10.1093/emboj/cdg413 [22] Zhang H, Macara IG. The polarity protein PAR-3 and TIAM1 cooperate in dendritic spine morphogenesis[J]. Nat Cell Biol, 2006, 8(3): 227-237. doi: 10.1038/ncb1368 [23] Tolias KF, Bikoff JB, Burette A, et al. The Rac1-GEF Tiam1 couples the NMDA receptor to the activity-dependent development of dendritic arbors and spines[J]. Neuron, 2005, 45(4):525-538. doi: 10.1016/j.neuron.2005.01.024 [24] Tolias KF, Bikoff JB, Kane CG, et al. The Rac1 guanine nucleotide exchange factor Tiam1 mediates EphB receptor-dependent dendritic spine development[J]. Proc Natl Acad Sci USA, 2007, 104(17):7265-7270. doi: 10.1073/pnas.0702044104 [25] Sanmartin E, Yanez Y, Fornes-Ferrer V, et al. TIAM1 variants improve clinical outcome in neuroblastoma[J]. Oncotarget, 2017, 8(28):45286-45297. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5542186/ [26] Pugh TJ, Morozova O, Attiyeh EF, et al. The genetic landscape of high-risk neuroblastoma[J]. Nat Genet, 2013, 45(3):279-284. doi: 10.1038/ng.2529 [27] Nishimura T, Yamaguchi T, Kato K, et al. PAR-6-PAR-3 mediates Cdc42-induced Rac activation through the Rac GEFs STEF/Tiam1[J]. Nat Cell Bio, 2005, 7(3):270. doi: 10.1038/ncb1227 [28] Cao Y, Jin Y, Yu J, et al. Clinical evaluation of integrated panel testing by nextgeneration sequencing for somatic mutations in neuroblastomas with MYCN unamplification[J]. Oncotarget, 2017. [Epub ahead of print]. https://www.ncbi.nlm.nih.gov/pubmed/28591696 [29] Genau HM, Huber J, Baschieri F, et al. CUL3-KBTBD6/KBTBD7Ubiquitin ligase cooperates with GABARAP proteins to spatially restrict TIAM1-RAC1 signaling[J]. Molecular Cell, 2015, 57(6):995-1010. doi: 10.1016/j.molcel.2014.12.040 [30] Ferrero M, Avivar A, Garcíamacías MC, et al. Phosphoinositide 3-kinase/AKT signaling can promote AIB1 stability independently of GSK3 phosphorylation[J]. Cancer Res, 2008, 68(13):5450. doi: 10.1158/0008-5472.CAN-07-6433 [31] Barbara H, Yania Y, Sarai P, et al. Targeting neuroblastoma stem cells with retinoic acid and proteasome inhibitor[J]. PLoS One, 2013, 8 (10):e76761-e76761. doi: 10.1371/journal.pone.0076761 [32] Keely PJ, Westwick JK, Whitehead IP, et al. Cdc42 and Rac1 induce integrin-mediated cell motility and invasiveness through PI(3)K[J]. Nature, 1997, 390(6660):632. doi: 10.1038/37656 [33] Matsuo N, Terao M, Nabeshima Y, et al. Roles of STEF/Tiam1, guanine nucleotide exchange factors for Rac1, in regulation of growth cone morphology[J]. Mol Cell Neurosci, 2003, 24(1):69-81. doi: 10.1016/S1044-7431(03)00122-2 [34] Leeuwen FN, Kain HE, Kammen RA, et al. The guanine nucleotide exchange factor Tiam1 affects neuronal morphology; opposing roles for the small GTPases Rac and Rho[J]. J Cell Biol, 1997, 139(3):797-807. doi: 10.1083/jcb.139.3.797 [35] Yamauchi J, Miyamoto Y, Tanoue A, et al. Ras activation of a Rac1 exchange factor, Tiam1, mediates neurotrophin-3-induced Schwann cell migration[J]. Neurosci Res, 2005, 102(41):14889-14894. http://www.ncbi.nlm.nih.gov/pubmed/16203995 [36] Miyamoto Y, Yamauchi J, Tanoue A, et al. TrkB binds and tyrosine-phosphorylates Tiam1, leading to activation of Rac1 and induction of changes in cellular morphology[J]. Proce Nat Acade Sci USA, 2006, 103(27):10444-10449. doi: 10.1073/pnas.0603914103 [37] Molenaar JJ, Koster J, Zwijnenburg DA, et al. Sequencing of neuroblastoma identifies chromothripsis and defects in neuritogenesis genes[J]. Nat Int Week J Sci, 2012, 483(7391):589-593. http://adsabs.harvard.edu/abs/2012Natur.483..589M [38] Speleman F, Park JR, Henderson TO. Neuroblastoma: a tough nut to crack[J]. Am Soci Clin Oncol Edu Book Am Soci Clin Oncol Meet, 2016, (35):e548. https://www.ncbi.nlm.nih.gov/pubmed/27249766?tool=MedlinePlus -




 下载:
下载: