Profiling the dynamic changes of PBMC immune-related indicators in patients with lung cancer after chemotherapy cycles
-
摘要:
目的 探索免疫治疗与化疗相结合的有利切入点,为临床免疫治疗介入提供试验依据。 方法 收集2015年11月至2016年12月新乡医学院第一附属医院初诊的23例肺癌患者,全部患者均完成连续5个周期的化疗,采用流式细胞术方法检测化疗过程中患者外周血免疫相关指标如CD4+T细胞、CD8+T细胞、CD19+B细胞和CD16+、CD56+、NK细胞比率,T细胞表面共信号分子PD-1、PD-L1、CD137、CTLA-4、CCR-4、LAG-3以及细胞因子表达情况。分析多周期过程中,上述指标动态变化趋势。 结果 在肺癌患者多个周期的化疗过程中,机体CD8+T淋巴细胞、CD19+B淋巴细胞和CD16+、CD56+、NK细胞水平下调,CD4+T淋巴细胞数量增加,差异具有统计学意义(P<0.05);T细胞表面共抑制分子PD-1、CTLA-4和CCR-4随治疗进行表达下调,差异具有统计学意义(P<0.05)。 结论 肺癌患者在多个周期的化疗中,实施淋巴细胞亚群数量及T细胞表面共抑制分子监测具有重要意义,化疗中后期针对免疫检查点高表达患者,联合应用PD-1抗体、PD-L1抗体、CTLA-4抗体或CCR-4抗体可能具有更优的治疗效果。 -
关键词:
- 免疫治疗 /
- T细胞表面共信号分子 /
- 肺癌 /
- 多个周期的化疗
Abstract:Objective To explore the optimal time point for combining chemotherapy and immunotherapy and provide an experimental basis for immunotherapy intervention in clinical. Methods Twenty-three lung cancer patients who completed five chemotherapy cycles between November 2015 and December 2016 in the First Affiliated Hospital of Xinxiang Medical University were enrolled in this study. Numbers of T lymphocyte subsets, B lymphocytes, and NK lymphocytes in peripheral blood were counted. Expression levels of T lymphocyte co-suppression molecule and cytokines in the peripheral blood mononuclear cell were detected using flow cytometry to analyze the dynamic changes of such indicators from one cycle to five cycles of chemotherapy. Results Significant decreases in the levels of CD8+ T lymphocytes, CD19+ B lymphocytes, and CD16+ CD56+ NK cells and an increase in CD4+ T lymphocytes were observed in the course of multi-cycle chemotherapy for patients with lung cancer. Differences were statistically significant (P < 0.05). The co-suppression molecular expression of PD-1, CTLA-4, and CCR-4 with T lymphocytes was downregulated, and the differences were significant (P < 0.05). Conclusion Profiling the dynamic changes of lymphocyte subsets and the expression of T lymphocyte co-suppression molecule are significant in multiple chemotherapy cycles for patients with lung cancer. In the later stage, the combined application of PD-1, PDL1, CTLA-4, or CCR-4 antibody may exert good therapeutic effects for patients with a high expression level of related immune checkpoints. -
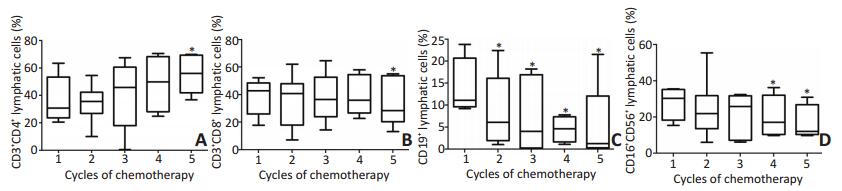
图 1 淋巴细胞亚群的变化趋势
Figure 1. Variation in lymphocyte subgroups
A. CD3+CD4+T lymphocyte level was significantly up regulated; B. CD3+CD8+T lymphocyte level was downregulated; C. CD19+B lymphocyte level was downregulated; D. CD16+CD56+ NK lymphocyte level was downregulated;*. indicates a statistical difference compared with the initial results, P < 0.05
图 2 CD4+T淋巴细胞表面相关免疫检查点变化趋势
Figure 2. Expression of immune checkpoints in CD4+T cell surface
A. The PD-1 expression on CD4+ T cells was significantly up regulated; B. The PD-L1 expression of CD4+ T cells was significantly down regulated; C. The change of CD137 on CD4+ T cells was not significant; D. The change of CTLA4 on CD4+ T cells was not significant; E. The change of CCR-4 on CD4+ T cells was not significant; F. The change of LAG-3 on CD4+ T cells was not significant;*. indicates a statistical difference compared with the initial results, P < 0.05
图 3 CD8+T淋巴细胞表面相关免疫检查点变化趋势
Figure 3. Expression of immune checkpoints in CD8+ T cell surface
A. The PD-1 expression on CD8+ T cells was significantly up regulated; B. The PD-L1 expression on CD8+ T cells was significantly down regulated; C. The change of CD137 on CD8+ T cells was not significant; D. The change of CTLA4 on CD8+ T cells was significant; E. CCR-4 on CD8+ T cells was significant up-regulated; F. The change of LAG-3 on CD8+ T cells was not significant;*. indicates a statistical difference compared with the initial results, P < 0.05
表 1 入组患者临床病理资料
Table 1. Clinical and pathological data of patients enrolled in the group

表 2 多个周期的化疗前外周血常规变化趋势
Table 2. Changes in peripheral blood routine in the pretreatment of chemotherapeutic treatment

表 3 多个周期的化疗前外周血生化指标变化
Table 3. Statistical analysis of peripheral blood biochemical changes in the pretreatment of chemotherapeutic treatment

-
[1] Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016[J]. CA J Clin, 2016, 66(1):7-30. doi: 10.3322/caac.21332 [2] Sateia HF, Choi Y, Stewart RW, et al. Screening for lung cancer[J]. Seminar in Oncol, 2017, 44(1):74-82. doi: 10.1053/j.seminoncol.2017.02.003 [3] Gasparri ML, Ruscito I, Taghavi K, et al. The Immunobiology of cancer: from tumor escape to cancer immunoediting towards immunotherapyin gynecologic oncology[M]. Mole Oncol: underlying mechanisms and translational advancements. springer international publishing, 2017:193-204. [4] Chacon JA, Schutsky K. The impact of chemotherapy, radiation and epigenetic modifiers in cancer cell expression of immune inhibitory and stimulatory molecules and anti-tumor efficacy[J]. Vaccines, 2016, 4(4):43. doi: 10.3390/vaccines4040043 [5] Seyedin SN, Schoenhals JE, Lee DA, et al. Strategies for combining immunotherapy with radiation for anticancer therapy[J]. Immun, 2015, 7(9):967. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4825325/figure/F1/ [6] 李军, 石殿鹏.肺癌与T淋巴细胞亚群相关性研究[J].中华肺部疾病杂志电子版, 2015, 8(2):110-112. http://d.wanfangdata.com.cn/Periodical/zhonghfbjbzz201502030Li J, Shi DP. Correlation study of lung cancer and T-lymphocyte subgroup[J]. China Lung Dis J, 2015, 8(2):110-112. http://d.wanfangdata.com.cn/Periodical/zhonghfbjbzz201502030 [7] 支旺旺, 孙超, 王淑珍.靶向T细胞共信号的肿瘤免疫治疗研究进展[J].药学进展, 2015, (9):659-665. http://www.wenkuxiazai.com/doc/68b8e49eb307e87100f69671.htmlZhi WW, Sun chao, Wang SZ. Progress research on tumor immunotherapy of targeted T cell co-signal[J]. Pharmac Pro, 2015, (9):659-665. http://www.wenkuxiazai.com/doc/68b8e49eb307e87100f69671.html [8] Lippitz BE. Cytokine patterns in patients with cancer: a systematic review[J]. The Lancet Oncol, 2013, 14(6):e218-e228. doi: 10.1016/S1470-2045(12)70582-X [9] 王传金, 纪秀英.肿瘤化疗药物的不良反应[J].现代肿瘤医学, 2006, 14(10):1321-1323. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=sxzl200610064&dbname=CJFD&dbcode=CJFQWang CJ, Ji XY. Adverse reactions of chemotherapy drugs in tumor [J]. Mod Oncol Medi, 2006, 14(10):1321-1323 http://kns.cnki.net/KCMS/detail/detail.aspx?filename=sxzl200610064&dbname=CJFD&dbcode=CJFQ [10] Calì B, Molon B, Viola A. Tuning cancer fate: the unremitting role of host immunity.[J]. Open Biol, 2017, 7(4). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5413907/ [11] Miceli MC, Parnes JR. Role of CD4 and CD8 in T cell activation and differentiation[J]. Adv Immun, 1993, 53(53):59-122. https://www.ncbi.nlm.nih.gov/pubmed/8512039 [12] Pieper K, Grimbacher B, Eibel H. B-cell biology and development[J]. J Alle Clin Immun, 2013, 131(4):959-971. doi: 10.1016/j.jaci.2013.01.046 [13] Vivier E, Tomasello E, Baratin M, et al. Functions of natural killer cells[J]. Nat Immu, 2008, 9(5):503-510. doi: 10.1038/ni1582 [14] Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition.[J]. Nat Rev Immun, 2013, 13(4):227-42. doi: 10.1038/nri3405 [15] Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy[J]. Nat Rev Cancer, 2012, 12(4):252-64. doi: 10.1038/nrc3239 [16] Meniawy TM, Lake RA, Mcdonnell AM, et al. PD-L1 on peripheral blood T lymphocytes is prognostic in patients with non-small cell lung cancer (NSCLC) treated with EGFR inhibitors[J]. Lung Cancer, 2016, (93):9. https://www.sciencedirect.com/science/article/pii/S0169500215301252 -




 下载:
下载: