The association between the expression and activity of indoleamine 2, 3-dioxygenase and the efficacy of neoadjuvant chemotherapy in patients with breast cancer
-
摘要:
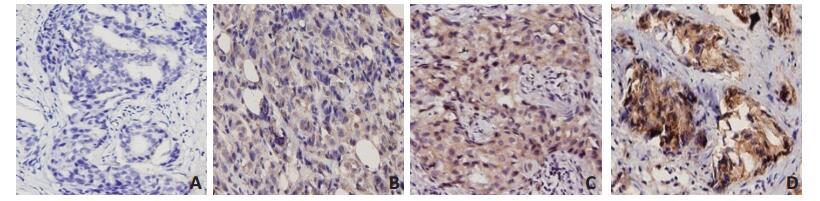
目的 分析吲哚胺2,3-双加氧酶(indoleamine 2,3-dioxygenase,IDO)在乳腺癌患者癌组织中表达及其在外周血中活性与新辅助化疗疗效的关系。 方法 收集2015年9月至2016年12月天津医科大学肿瘤医院53例行新辅助化疗乳腺癌患者的肿瘤穿刺标本和血液标本,采用免疫组织化学法及高效液相色谱法检测癌组织中IDO表达及外周血中色氨酸(tryptophan,Trp)、犬尿氨酸(kynurenine,Kyn)浓度与IDO活性,分析IDO表达及活性与化疗疗效的相关性。 结果 新辅助化疗前乳腺癌组织中IDO表达与临床T分期(P=0.006)、N分期(P=0.020)、临床分期(P=0.045)及ER状态(P=0.014)有关。新辅助化疗前外周血中IDO高活性伴随癌组织中IDO高表达(P=0.004),并与临床T分期(P=0.019)及N分期(P=0.047)有关。单因素分析显示新辅助化疗临床疗效与化疗前临床T分期(P=0.049)、ER状态(P=0.025)及分子分型(P=0.014)有关;病理完全缓解(pathologic complete response,pCR)与化疗前临床T分期(P=0.014)有关。更重要的是新辅助化疗临床疗效及pCR均与化疗前IDO表达及活性有关(均P < 0.05)。多因素分析显示新辅助化疗前外周血中IDO活性是影响pCR的唯一独立因素(P=0.032)。 结论 新辅助化疗前乳腺癌组织中IDO表达和外周血中IDO活性与化疗疗效相关,可以为临床预测化疗是否敏感提供一定信息。 -
关键词:
- 乳腺癌 /
- 吲哚胺2,3-双加氧酶 /
- 新辅助化疗 /
- 化疗疗效 /
- 病理完全缓解
Abstract:Objective To investigate the association between indoleamine 2, 3-dioxygenase (IDO) expression in tumor tissue, its peripheral blood activity, and the efficacy of neoadjuvant chemotherapyin patients with breast cancer. Methods Immunohistochemistry (IHC) and high-performance liquid chromatography (HPLC) were used to measure IDO protein expression in tumor tissue, and kynurenine (Kyn), tryptophan (Trp), and IDO activity (Kyn/Trp) in peripheral blood before neoadjuvant chemotherapy in 53 patients with breast cancer from Tianjin Medical University Cancer Institute and Hospital between September 2015 and December 2016. The correlations between the expression and activity of IDO and the efficacy of chemotherapy were analyzed. Results In tumor tissue, IDO expressionbefore neoadjuvant chemotherapy was related to clinical tumor stages (P=0.006), node stages (P=0.020), clinical stages (P=0.045), and estrogen receptor (ER) status (P=0.014). High IDO activity before neoadjuvant chemotherapy in peripheral blood was associated with high IDO expression in tumor tissue (P=0.004), and was also correlated with clinical tumor stages (P=0.019) and node stages (P=0.047). Univariate analysis showed that the clinical efficacy of neoadjuvant chemotherapy was associated with pre-chemotherapeutic clinical tumor stages (P=0.049), ER status (P=0.025), and molecular subtype (P=0.014), while pathologic complete response (pCR) was related to pre-chemotherapeutic clinical tumor stages (P=0.014). Importantly, the clinical efficacy of neoadjuvant chemotherapy and pCR were both related to IDO expression and activity before chemotherapy (all P < 0.05). Multivariate analysis showed that pre-chemotherapeutic IDO activity in peripheral blood was the only independent factor that affected pCR (P=0.032). Conclusions Tumor tissue IDO expression and peripheral blood IDO activity before chemotherapy were associated with chemotherapy efficacy, and could provide promising information for the clinical prediction of chemotherapy sensitivity. -
表 1 53例乳腺癌患者新辅助化疗前癌组织中IDO表达及外周血中IDO活性与临床病理特征的相关性

表 2 53例乳腺癌患者新辅助化疗前癌组织中IDO表达及外周血中IDO活性和化疗疗效的关系

表 3 53例乳腺癌患者新辅助化疗疗效的单因素分析

表 4 乳腺癌患者新辅助化疗疗效的多因素二元Logistic回归分析

-
[1] Okamoto A, Nikaido T, Ochiai K, et al. Indoleamine 2, 3-dioxygenase serves as amarker of poor prognosis in gene expression profiles of serous ovarian cancer cells[J]. Clin Cancer Res, 2005, 11(16):6030-6039. doi: 10.1158/1078-0432.CCR-04-2671 [2] Lyon DE, Walter JM, Starkweather AR, et al. Tryptophan degradation in women withbreast cancer: a pilot study[J]. BMC Res Notes, 2011, 4:156. doi: 10.1186/1756-0500-4-156 [3] Sakurai K, Fujisaki S, Nagashima S, et al. Indoleamine 2, 3-dioxygenase activityduring neoadjuvant chemotherapy in patients with breast cancer[J]. Gan To Kagaku Ryoho, 2013, 40(12):1578-1580. https://www.sigmaaldrich.com/catalog/papers/24393854 [4] Ino K, Yoshida N, Kajiyama H, et al. Indoleamine 2, 3-dioxygenase is a novel prognostic indicator for endometrial cancer[J]. Br J Cancer, 2006, 95(11):1555-1561. doi: 10.1038/sj.bjc.6603477 [5] Liu JT, Wei LJ, Yu JP, et al. Expression of indoleamine 2, 3-dioxygenase and its correlation with prognosis in breast cancer patients[J]. Zhong Hua Zhong Liu Za Zhi, 2011, 33(7):513-516. [6] Isla Larrain MT, Rabassa ME, Lacunza E, et al. IDO is highly expressed in breast cancer and breast cancer-derived circulating microvesicles and associated to aggressive types of tumors by in silico analysis[J]. Tumour Biol, 2014, 35(7):6511-6519. doi: 10.1007/s13277-014-1859-3 [7] Hascitha J, Priya R, Jayavelu S, et al. Analysis of Kynurenine/Tryptophan ratio and expression of IDO1 and 2mRNA in tumour tissue of cervical cancer patients[J]. Clin Biochem, 2016, 49(12):919-924. doi: 10.1016/j.clinbiochem.2016.04.008 [8] Sakurai K, Amano S, Enomoto K, et al. Study of indoleamine 2, 3-dioxygenaseexpression in patients with breast cancer[J]. Gan To Kagaku Ryoho, 2005, 32(11):1546-1549. https://www.researchgate.net/publication/7452889_Study_of_indoleamine_23-dioxygenase_expression_in_patients_with_breast_cancer [9] Sakurai K, Fujisaki S, Suzuki S, et al. Indoleamine 2, 3-dioxygenase activity in breast cancer patients with local recurrence or distant metastases[J]. Gan To Kagaku Ryoho, 2014, 41(10):1304-1306. [10] Ninomiya S, Narala N, Huye L, et al. Tumor indoleamine 2, 3-dioxygenase (IDO) inhibits CD19-CAR T cells and is downregulated by lymphodepleting drugs[J]. Blood, 2015, 125(25):3905-3916. doi: 10.1182/blood-2015-01-621474 [11] Sim SH, Ahn YO, Yoon J, et al. Influence of chemotherapy on nitric oxide synthase, indole-amine-2, 3-dioxygenase and CD124 expression in granulocytes and monocytes ofnon-small cell lung cancer [J]. Cancer Sci, 2012, 103(2):155-160. doi: 10.1111/cas.2012.103.issue-2 [12] Creelan BC, Antonia S, Bepler G, et al. Indoleamine 2, 3-dioxygenase activity andclinical outcome following induction chemotherapy and concurrent chemoradiation in stage Ⅲnon-small cell lung cancer[J]. Oncoimmunology, 2013, 2(3):e23428. doi: 10.4161/onci.23428 [13] Sakurai K, Fujisaki S, Suzuki S, et al. Indoleamine 2, 3-dioxygenase activity during fulvestrant therapy for aromatase inhibitor-resistant metastatic breast cancer[J]. Gan ToKagaku Ryoho, 2015, 42(10): 1225-1227. -




 下载:
下载: