The influence of genetic variation of cytidine deaminase on hand-foot syndrome among colorectal cancer patients treated with capecitabine-based adjuvant chemotherapy regimens
-
摘要:
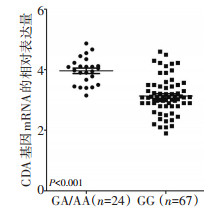
目的 探讨胞苷脱氨酶(cytidine deaminase,CDA)基因变异对接受含卡培他滨方案治疗的结直肠癌患者的3级手足综合征(hand-foot syndrome,HFS)的影响。 方法 选取2014年5月至2017年5月在郑州人民医院接受卡培他滨治疗的结直肠癌患者149例,对卡培他滨代谢通路上的关键基因CDA进行基因分型,对分型的位点和HFS发生的相关性进行分析。另外,从全部患者中随机选取91例患者的外周血单核细胞(peripheral blood mononuclear cell,PBMC)提取RNA,对CDA的mRNA表达水平进行测定,并结合多态性位点进行分析。 结果 位于CDA基因启动子区域的多态性位点-451G > A与HFS相关。基因分型结果为GG型109例(73.15%),GA型38例(25.50%),AA型2型(1.36%),最小等位基因频率为0.14,三种基因型分布频率符合哈迪温伯格平衡(P=0.516)。相关性分析发现-451G > A位点GA/AA型患者与HFS的发生显著相关(OR=2.53,P=0.014)。另外,位于CDA基因启动子区域的插入变异-33delC和-451C > T呈现高度的连锁不平衡(D'=0.92)。在91例PBMC当中提取的RNA表达分析结果表明,-451G > A位点GA/AA型患者的CDA mRNA表达水平显著高于野生型GG型患者(4.01±0.53 vs. 3.13±0.61,P < 0.001)。 结论 TAM的-451G > A多态性位点可能通过介导CDA基因mRNA的表达从而影响卡培他滨引起的HFS。 Abstract:Objective To investigate the association between grade 3 hand-foot syndrome (HFS) in colorectal cancer (CRC) patients treated with capecitabine and variation of cytidine deaminase (CDA) genes. Methods The polymorphisms of the key gene CDA involved in capecitabine metabolism were genotyped and 149 CRC patients were included in this study. The association between these polymorphisms and susceptibility to HFS were analyzed. Additionally, peripheral blood mononuclear cells (PBMCs) of 91 CRC patients were collected for mRNA expression analysis, and the levels of mRNA expression according to different CDA genotypes were compared. Results The prevalence of the polymorphism -451G > A, which is located in the promoter region of CDA, were correlated with HFS. The results were as follows: GG genotype, 109 cases (73.15%); GA genotype, 38 cases (25.50%); and AA genotype, 2 cases (1.36%). The minor allele frequency of -451G > A was 0.14. The distribution of the three genotypes were in accordance with HardyWeinberg Equilibrium (P=0.516). Logistic analysis indicated that GA/AA genotypes were associated with grade 3 HFS (odds ratio=2.53, P=0.011). Additionally, another insert polymorphism-33delC located in the promoter region of CDA was in linkage disequilibrium with -451G > A (D' =0.92). Of the 91 PBMC mRNA expression analyses, the GA/AA genotype of -451G > A was associated with higher CDA mRNA expression compared with GG genotypes (4.01±0.53 vs. 3.13±0.61, P < 0.001). Conclusions The polymorphism -451G > A of CDA may influence occurance of grade 3 HFS induced by capecitabine by influencing CDA mRNA expression. -
Key words:
- capecitabine /
- cytidine deaminase /
- polymorphism /
- hand-foot syndrome
-
表 1 149例结直肠癌患者的基线临床资料

-
[1] Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2):115-132. doi: 10.3322/caac.21338 [2] Rosen LS, Jacobs IA, Burkes RL. Bevacizumab in colorectal cancer: current role in treatment and the potential of biosimilars[J]. Target Oncol, 2017, 12(5):599-610. doi: 10.1007/s11523-017-0518-1 [3] Li J, Qin S, Xu R, et al. Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, doubleblind, placebo-controlled, phase 3 trial[J]. Lancet Oncol, 2015, 16(6): 619-629. doi: 10.1016/S1470-2045(15)70156-7 [4] Lonardi S, Sobrero A, Rosati G, et al. Phase Ⅲ trial comparing 3-6 months of adjuvant FOLFOX4/XELOX in stage Ⅱ-Ⅲ colon cancer: safety and compliance in the TOSCA trial[J]. Ann Oncol, 2017, 28(12): 3110. doi: 10.1093/annonc/mdx021 [5] 白羽, 吴海伟, 刘颖, 等.二氢嘧啶脱氢酶和亚甲基四氢叶酸还原酶基因多态性与卡培他滨方案治疗结直肠癌患者药物不良反应的相关性[J].中国临床药理学杂志, 2017, 33(17):1637-1640. http://www.cnki.com.cn/Article/CJFDTotal-SHYW201507025.htm [6] Chen M, Chen J, Peng X, et al. The contribution of keratinocytes in capecitabine-stimulated hand-foot-syndrome[J]. Environ Toxicol Pharmacol, 2017, 49:81-88. doi: 10.1016/j.etap.2016.12.001 [7] Cacabelos R, Meyyazhagan A, Carril JC, et al. Pharmacogenetics of vascular risk factors in alzheimer's disease[J]. J Pers Med, 2018, 8(1): E3. doi: 10.3390/jpm8010003 [8] Conyers R, Devaraja S, Elliott D. Systematic review of pharmacogenomics and adverse drug reactions in paediatric oncology patients[J]. Pediatr Blood Cancer, 2017, 2018, 64(4): 1-12. http://www.ncbi.nlm.nih.gov/pubmed/29286579 [9] Caronia D, Martin M, Sastre J, et al. A polymorphism in the cytidine deaminase promoter predicts severe capecitabine-induced hand-foot syndrome[J]. Clin Cancer Res, 2011, 17(7):2006-2013. doi: 10.1158/1078-0432.CCR-10-1741 [10] Rachar V, Czejka M, Kitzmueller M, et al. Assessment of pharmacokinetic interaction between capecitabine and cetuximab in metastatic colorectal cancer patients[J]. Anticancer Res, 2016, 36(9):4715-4723. doi: 10.21873/anticanres [11] Mu Y, Zelazowska MA, McBride KM. Phosphorylation promotes activation-induced cytidine deaminase activity at the Myc oncogene[J]. J Exp Med, 2017, 214(12):3543-3552. doi: 10.1084/jem.20170468 [12] Yue L, Saikawa Y, Ota K, et al. A functional single-nucleotide polymorphism in the human cytidine deaminase gene contributing to araC sensitivity[J]. Pharmacogenetics, 2003, 13(1):29-38. doi: 10.1097/00008571-200301000-00005 [13] Yonemori K, Ueno H, Okusaka T, et al. Severe drug toxicity associated with a single-nucleotide polymorphism of the cytidine deaminase gene in a Japanese cancer patient treated with gemcitabine plus cisplatin[J]. Clin Cancer Res, 2005, 11(7):2620-2624. doi: 10.1158/1078-0432.CCR-04-1497 [14] Fitzgerald SM, Goyal RK, Osborne WR, et al. Identification of functional single nucleotide polymorphism haplotypes in the cytidine deaminase promoter[J]. Hum Genet, 2006, 119(3):276-283. doi: 10.1007/s00439-006-0142-0 [15] Nikolaou V, Syrigos K, Saif MW. Incidence and implications of chemotherapy related hand-foot syndrome[J]. Expert Opin Drug Saf, 2016, 15(12):1625-1633. doi: 10.1080/14740338.2016.1238067 [16] 张莎, 陈自平, 杜文军, 等.DNA修复基因XPD XPC XRCC4基因多态性与结直肠癌易感性的关联性研究[J].中国肿瘤临床, 2017, 44(8):365-370. http://cpfd.cnki.com.cn/Article/CPFDTOTAL-ZGBH201111001252.htm -




 下载:
下载: