Expression and prognostic value of immune checkpoint molecule A2aR in tumor tissue and on the surface of CD8+T cells in diffuse large B cell lymphoma
-
摘要:
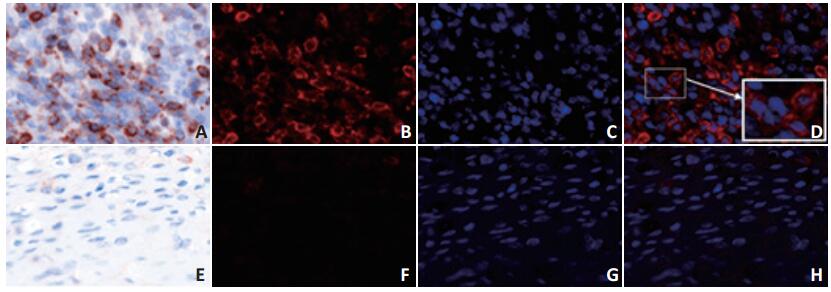
目的 探讨A2aR在弥漫大B细胞淋巴瘤(diffuse large B cell lymphoma,DLBCL)组织和CD8+T细胞上的表达及与患者临床病理特征和预后的相关性。 方法 回顾性分析2010年1月1日至2015年12月31日就诊于天津医科大学肿瘤医院初治的72例DLBCL患者临床病理资料。采用多重免疫组织化学法检测组织中A2aR、CD8的表达,采用χ2检验分析其与患者临床病理特征的相关性,应用Kaplan-Meier法进行生存资料分析。 结果 DLBCL组织中A2aR总表达阳性率为41.7%(30/72),与患者分期、IPI评分、Ki-67、β2-MG、B症状相关(P < 0.05);A2aR+组患者总生存期(overall survival,OS)较A2aR-组差(P < 0.05)。DLBCL组织中CD8+T细胞上A2aR表达阳性率为22.2%(16/58),与患者分期、IPI评分、Ki-67、β2-MG、乳酸脱氢酶(lactate dehydrogenase,LDH)、结外受累数目、B症状相关(P < 0.05);A2aR+CD8+组患者OS较A2aR-CD8+组显著更差(P < 0.001)。 结论 A2aR总表达及CD8+T细胞上A2aR表达与DLBCL患者临床病理特征及预后密切相关。检测CD8+T细胞上A2aR表达可更好地评价DLBCL患者的预后。 -
关键词:
- 弥漫大B细胞淋巴瘤 /
- A2aR /
- CD8阳性T淋巴细胞 /
- 预后
Abstract:Objective To investigate the expression of adenosine A2A receptor (A2aR) in tumor tissue and on the surface of CD8+T cells and its correlation with clinicopathologic characteristics and prognosis of patients with diffuse large B cell lymphoma (DLBCL). Methods A retrospective analysis was performed on the clinicopathologic characteristics of 72 newly diagnosed patients at Tianjin Medical University Cancer Institute and Hospital from January 2010 to December 2015. The expression of A2aR and CD8 was evaluated by multiple immunohistochemical staining assays. The associations between the expression of A2aR in tumor tissue and on the surface of CD8+T cells and clinicopathologic characteristics were then assessed using the Chi-square test. The Kaplan-Meier method was used for the analysis of survival. Results The positive rate of A2aR total expression in DLBCL was 41.7% (30/72), which was associated with several clinicopathologic characteristics including stage, International Prognostic Index (IPI) score, Ki-67 index score, β-2 microglobulin (MG), and B symptoms (P < 0.05). Survival analysis showed that patients with positive A2aR expression had poorer overall survival (OS) than those with negative A2aR expression (P < 0.05). The rate of positive A2aR expression on the surface of CD8+T cells was 22.2% (16/58). The expression of A2aR on the surface of CD8+T cells was correlated with several clinicopathologic characteristics including stage, IPI, Ki-67, β2-MG, lactate dehydrogenase levels (LDH), extranodal involvement, and B symptoms (P < 0.05). Survival analysis showed that patients with A2aR+CD8+ status had significantly poorer OS than those with A2aR-CD8+ status (P < 0.001). Conclusions Positive expression of A2aR in tumor tissue and on the surface of CD8+T cells is significantly correlated with clinicopathologic characteristics and prognosis of patients with DLBCL. The detection of A2aR on the surface of CD8+T cells is superior to evaluate the prognosis of patients with DLBCL. -
Key words:
- diffuse large B cell lymphoma /
- A2aR /
- CD8-positive T-lymphocyte /
- prognosis
-
表 1 A2aR总表达与患者临床病理特征之间的关系

表 2 CD8+T细胞上A2AR表达与患者临床病理特征之间的关系

-
[1] Coiffier B, Sarkozy C. Diffuse large B-cell lymphoma: R-CHOP failurewhat to do[J]? Hematol Am Soc Hematol Educ Program, 2017, 2016(1): 366-378. http://www.ncbi.nlm.nih.gov/pubmed/27913503 [2] Leone RD, Ying-Chun L, Powell JD. A2aR antagonists: Next generation checkpoint blockade for cancer immunotherapy[J]. Comput Struct Biotechnol J, 2015, 13(8):265-272. http://cn.bing.com/academic/profile?id=742649986814bed968c6c879fb3d357e&encoded=0&v=paper_preview&mkt=zh-cn [3] Ma SR, Deng WW, Liu JF, et al. Blockade of adenosine A2A receptor enhances CD8+T cells response and decreases regulatory T cells in head and neck squamous cell carcinoma[J]. Mole Cancer, 2017, 16 (1):99. doi: 10.1186/s12943-017-0665-0 [4] Mediavilla-Varela M, Castro J, Chiappori A, et al. A novel antagonist of the immune checkpoint protein adenosine A2a receptor restores tumor-infiltrating lymphocyte activity in the context of the tumor microenvironment[J]. Neoplasia, 2017, 19(7):530-536. doi: 10.1016/j.neo.2017.02.004 [5] Sitkovsky MV, Hatfield S, Abbott R, et al. Hostile, hypoxia-A2-adenosinergic tumor biology as the next barrier to the tumor immunologists[J]. Cancer Immunol Res, 2014, 2(7):598-605. doi: 10.1158/2326-6066.CIR-14-0075 [6] Iannone R, Miele L, Maiolino P, et al. Adenosine limits the therapeutic effectiveness of anti-CTLA4 mAb in a mouse melanoma model[J]. Am J Cancer Res, 2014, 4(2):172-181. http://cn.bing.com/academic/profile?id=3f5c9fea6ec490397a37cfc81255da85&encoded=0&v=paper_preview&mkt=zh-cn [7] Inoue Y, Yoshimura K, Kurabe N, et al. Prognostic impact of CD73 and A2A adenosine receptor expression in non-small-cell lung cancer[J]. Oncotarget, 2017, 8(5):8738-8751. http://cn.bing.com/academic/profile?id=de8d2e664a0e63c49081a0a2d4831f0d&encoded=0&v=paper_preview&mkt=zh-cn [8] Sevigny CP, Li L, Awad AS, et al. Activation of adenosine 2A receptors attenuates allograft rejection and alloantigen recognition[J]. J Immunol, 2007, 178(7):4240-4249. doi: 10.4049/jimmunol.178.7.4240 [9] Murphy KA, Griffith TS. CD8+T cell-independent antitumor response and Its potential for treatment of malignant gliomas[J]. Cancers, 2016, 8(8):71. doi: 10.3390/cancers8080071 [10] Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy[J]. Nat Rev Cancer, 2012, 12(4):252-264. doi: 10.1038/nrc3239 -




 下载:
下载: