Effect of long non-coding RNA FOXD2-AS1 on proliferation and migration of ovarian cancer cells
-
摘要:
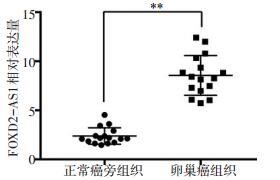
目的 观察长链非编码RNA(lncRNA)FOXD2-AS1对卵巢癌细胞增殖和迁移的影响。 方法 收集2017年2月至2017年9月华中科技大学同济医学院附属武汉儿童医院(武汉市妇幼保健院)手术切除的16例卵巢癌患者的组织标本,采用荧光实时定量聚合酶链式反应(quantitative PCR,qPCR)检测组织标本、人卵巢癌细胞系及人正常卵巢上皮细胞系中的FOXD2-AS1表达水平。将HO-8910细胞分为实验组和对照组,分别转染siRNA-FOXD2-AS1和阴性对照RNA。细胞计数CCK-8法、Transwell迁移实验分别检测沉默FOXD2-AS1表达对卵巢癌HO-8910细胞增殖活性和迁移能力的影响。生物信息学方法预测FOXD2-AS1的下游靶基因,qPCR检测下游靶基因表达,Western blot法检测葡萄糖调节蛋白94(glucose regulated protein94,GRP94)和信号通路Wnt/β-catenin的蛋白表达。 结果 卵巢癌组织中的FOXD2-AS1相对表达量8.56±0.51明显高于正常癌旁组织的2.38±0.21(P < 0.01),FOXD2-AS1在卵巢癌细胞系OC3、HO-8910、A2780、SKOV-3中表达明显增加(P < 0.05)。下调FOXD2-AS1表达明显抑制卵巢癌HO-8910细胞的增殖活性和迁移能力(均P < 0.01)。生物信息学方法显示,FOXD2-AS1靶向结合miR-150-5p后,可靶向结合GRP94基因。实验组和对照组miR-150-5p相对表达量分别为5.45±0.91和1.01±0.08(P < 0.01),GRP94 mRNA相对表达量分别为0.32±0.05和1.02±0.11(P < 0.01)。GRP94和Wnt/β-catenin信号通路蛋白表达减少。 结论 FOXD2-AS1在卵巢癌组织和细胞系呈高表达,下调FOXD2-AS1可调控miR-150-5p表达,抑制卵巢癌HO-8910细胞的增殖和迁移,可发挥抑癌基因的功能。 -
关键词:
- FOXD2-AS1 /
- 卵巢癌 /
- miR-150-5p /
- 细胞增殖 /
- 细胞迁移
Abstract:Objective This study was conducted to determine the effect of long-chain non-coding RNA FOXD2-AS1 on the proliferation and migration of ovarian cancer cells. Methods Tissue specimens were collected from 16 patients with ovarian cancer undergoing surgical resection at Wuhan Children's Hospital between February 2017 and September 2017. The expression of FOXD2-AS1 in tissues, human ovarian cancer cell lines, and human normal ovarian epithelial cell line IOSE80 were detected by quantitative PCR. HO-8910 cells were divided into experimental and control groups and transfected with siRNA-FOXD2-AS1 and negative control RNA, respectively. The effect of low expression of FOXD2-AS1 on the proliferation and cell migration of ovarian cancer HO-8910 cells was detected by cell counting CCK-8 and Transwell migration assays. Bioinformatic methods were used to predict the target genes of FOXD2-AS1; quantitative PCR was used to detect the expression of the target genes, and Western blot was performed to detect the expression of glucoseregulated protein (GRP94) and Wnt/β-catenin signaling pathway proteins. Results The relative expression of FOXD2-AS1 in ovarian cancer tissues (8.56±0.51) was significantly higher than that in normal paracancerous tissues (2.38 ± 0.21) (P < 0.01). FOXD2-AS1 expression was significantly increased in the ovarian cancer cell lines OC3, HO-8910, A2780, and SKOV-3 (P < 0.05). Down-regulation of FOXD2- AS1 significantly inhibited the proliferative activity (P < 0.05) and migration ability (P < 0.01) of ovarian cancer HO-8910 cells. Bioinformatic analysis showed that FOXD2-AS1 can bind miR-150-5p and then target the GRP94 gene. The relative expression levels of miR- 150-5p in the control and experimental groups were 1.01±0.08 and 5.45±0.91, respectively (P < 0.01). The relative expression levels of GRP94 mRNA were 1.02±0.11 and 0.32±0.05, respectively (P < 0.01). The expression of GRP94 and Wnt/β-catenin signaling pathway proteins was decreased. Conclusions FOXD2- AS1 is highly expressed in ovarian cancer tissues and cell lines. Down- regulation of FOXD2-AS1 inhibits the proliferation and migration of ovarian cancer HO-8910 cells by regulating miR-150-5p expression, thereby functioning as a tumor suppressor gene in ovarian cancer. -
Key words:
- FOXD2-AS1 /
- ovarian cancer /
- miR-150-5p /
- cell proliferation /
- cell migration
-
表 1 引物序列

-
[1] Coni P, Madeddu A, Kuqi L, et al. LncRNA colon cancer-associated transcript 1 (CCAT1) in ovarian cancer[J]. Eur Rev Med Pharmacol Sci, 2018, 22(6):1525-1527. http://europepmc.org/abstract/MED/29630091 [2] Yang CH, Zhang XY, Zhou LN, et al. LncRNA SNHG8 participates in the development of endometrial carcinoma through regulating c-MET expression by miR-152[J]. Eur Rev Med Pharmacol Sci, 2018, 22(6): 1629-1637. [3] Liu S, Lei H, Luo F, et al. The effect of lncRNA HOTAIR on chemoresistance of ovarian cancer through regulation of HOXA7[J]. Biol Chem, 2018, 399(5):485-497. doi: 10.1515/hsz-2017-0274 [4] Bao J, Zhou C, Zhang J, et al. Upregulation of the long noncoding RNA FOXD2-AS1 predicts poor prognosis in esophageal squamous cell carcinoma[J]. Cancer Biomark, 2018, 21(3):527-533. doi: 10.3233/CBM-170260 [5] Su F, He W, Chen C, et al. The long non-coding RNA FOXD2-AS1 promotes bladder cancer progression and recurrence through a positive feedback loop with Akt and E2F1[J]. Cell Death Dis, 2018, 9(2):233. doi: 10.1038/s41419-018-0275-9 [6] Wang Y, Zhang W, Wang Y, et al. HOXD-AS1 promotes cell proliferation, migration and invasion through miR-608/FZD4 axis in ovarian cancer[J]. Am J Cancer Res, 2018, 8(1):170-182. https://reference.medscape.com/viewpublication/18822_2 [7] Tao F, Tian X, Lu M, et al. A novel lncRNA, lnc-OC1, promotes ovarian cancer cell proliferation and migration by sponging miR-34a and miR-34c[J]. J Genet Genomics, 2018, 45(3):137-145. doi: 10.1016/j.jgg.2018.03.001 [8] Wang Z, Yang B, Zhang M, et al. LncRNA epigenetic landscape analysis identifies EPIC1 as an oncogenic lncRNA that interacts with MYC and promotes cell-cycle progression in cancer[J]. Cancer Cell, 2018, 33(4): 706-720. doi: 10.1016/j.ccell.2018.03.006 [9] Yang X, Duan B, Zhou X. Long non-coding RNA FOXD2-AS1 functions as a tumor promoter in colorectal cancer by regulating EMT and Notch signaling pathway[J]. Eur Rev Med Pharmacol Sci, 2017, 21(16): 3586-3591. doi: 10.1186/s12943-018-0812-2 [10] Lai XJ, Cheng HF. LncRNA colon cancer- associated transcript 1 (CCAT1) promotes proliferation and metastasis of ovarian cancer via miR-1290[J]. Eur Rev Med Pharmacol Sci, 2018, 22(2):322-328. doi: 10.1007/s13277-014-2511-y [11] Xia B, Hou Y, Chen H, et al. Long non-coding RNA ZFAS1 interacts with miR- 150- 5p to regulate Sp1 expression and ovarian cancer cell malignancy[J]. Oncotarget, 2017, 8(12):19534-19546. http://cn.bing.com/academic/profile?id=3c606318e7881711127473511780fcd4&encoded=0&v=paper_preview&mkt=zh-cn [12] Peng L, Chen G, Zhu Z, et al. Circular RNA ZNF609 functions as a competitive endogenous RNA to regulate AKT3 expression by sponging miR-150-5p in Hirschsprung's disease[J]. Oncotarget, 2017, 8(1):808- 818. http://cn.bing.com/academic/profile?id=e9628f3de0968846658a0ab6cc4f8870&encoded=0&v=paper_preview&mkt=zh-cn [13] Tan JS, Ong Kc KC, Rhodes A. The role of heat shock proteins and glucose regulated proteins in cancer[J]. Malays J Pathol, 2016, 38(2): 75-82. http://cn.bing.com/academic/profile?id=4e712601b853c1e1fd3b0a85775c0454&encoded=0&v=paper_preview&mkt=zh-cn [14] Nami B, Ghasemi-Dizgah A, Vaseghi A. Overexpression of molecular chaperons GRP78 and GRP94 in CD44(hi)/CD24(lo) breast cancer stem cells[J]. Bioimpacts, 2016, 6(2):105-110. doi: 10.15171/bi.2016.15 [15] Huang CY, Batzorig U, Cheng WL, et al. Glucose- regulated protein 94 mediates cancer progression via AKT and eNOS in hepatocellular carcinoma[J]. Tumour Biol, 2016, 37(4):4295-4304. doi: 10.1007/s13277-015-4254-9 -




 下载:
下载: