Comparison of the efficacy and safety of cisplatin, etoposide, and irinotecan combined with chemotherapy and topotecan monotherapy as second-line treatment for patients with sensitive relapsed SCLC
-
摘要:
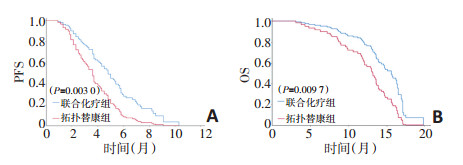
目的 比较顺铂、依托泊苷、伊立替康联合化疗方案和单药拓扑替康二线治疗敏感复发型小细胞肺癌(smalll cell lung cancer, SCLC)的疗效和安全性。 方法 收集2014年9月至2017年9月吉林省肿瘤医院就诊78例患者资料, 筛选敏感复发型小细胞肺癌患者, 其中36例患者给予顺铂、依托泊苷、伊立替康联合化疗方案, 42例患者给予单药拓扑替康化疗。联合化疗组药物用法:顺铂25 mg/m2, 第1天、第8天静脉滴注; 依托泊苷60 mg/m2, 第1、2、3天静脉滴注; 伊立替康90 mg/m2, 第8天静脉滴注, 连续给予5个2周方案的化疗。单药拓扑替康组药物用法:拓扑替康1.5 mg/m2, 第1~5天静脉滴注, 每3周1个周期。评价两组治疗方案的无进展生存时间(progressionfree survival, PFS)、总生存时间(overall survival, OS)及安全性。 结果 联合化疗组中位无进展生存时间(mPFS)5.3个月(95% CI:4.3~ 5.8), 拓扑替康组mPFS 3.2个月(95% CI:2.7~4.0), 差异具有统计学意义(P=0.003 0);联合化疗组中位总生存时间(mOS)16.3个月(95% CI:13.8~19.1), 拓扑替康组mOS 13.1个月, 差异具有统计学意义(P=0.009 7)。联合化疗组和单药拓扑替康组常见的3/4级不良事件主要有中性粒细胞下降[31例(86.1%) vs.28例(66.7%)]、白细胞下降[29例(80.6%) vs.21例(50.0%)]、贫血[26例(72.2%) vs.10例(23.8%)]、血小板下降[13(36.1%) vs.11(26.2%)]。联合化疗组发生1例治疗相关死亡(发热性中性粒细胞下降合并肺部感染), 拓扑替康组无治疗相关的死亡发生。 结论 顺铂、依托泊苷、伊立替康联合化疗方案比单药拓扑替康疗效更好, 可考虑作为敏感复发型SCLC患者二线化疗的备选方案之一。两种化疗方案毒性均可耐受, 但联合化疗组不良事件发生率更高, 应进一步探索更为合适的化疗剂量。 Abstract:Objective To investigate whether combination chemotherapy with cisplatin, etoposide, and irinotecan was better than topotecan alone as second-line chemotherapy in patients with sensitive relapsed small cell lung cancer (SCLC).Method:Between September, 2014 and September, 2017, the patients'data were collected in Jilin Province Cancer Hospital.All patients were diagnosed with sensitive relapsed SCLC.Thirty-six patients received combination chemotherapy containing cisplatin plus etoposide plus irinotecan, and 42 patients received topotecan alone.Combination chemotherapy consisted of five 2-week courses of intravenous cisplatin 25 mg/m2 on days 1 and 8, intravenous etoposide 60 mg/m2 on days 1-3, and intravenous irinotecan 90 mg/m2 on day 8.Topotecan therapy consisted of at least one course of intravenous topotecan 1.5 mg/m2 on days 1-5, every 3 weeks.The primary endpoints were progression-free survival (PFS) and overall survival (OS), and safety was assessed in all patients who received at least one dose of drugs. Results PFS was significantly longer in the combination chemotherapy group[median 5.3 months, 95% confidence interval (CI)4.3-5.8]than in the topotecan group (3.2 months, 95% CI:2.7-4.0; P=0.0030);OS was also significantly increased in the combination chemotherapy group (median 16.3 months, 95% CI:13.8-19.1) than in the topotecan group (13.1 months, 95% CI:10.2-15.4;P=0.0097).The most common grade 3/4 adverse events were neutropenia[31(86.1%) patients in the combination chemotherapy group vs.28(66.7%) patients in the topotecan group], anemia[26(72.2%) vs.10(23.8%)], leucopenia[29(80.6%) vs.21(50.0%)], and thrombocytopenia[13(36.1%) vs.11(26.2%)].One treatment-related death (febrile neutropenia with pulmonary infection) occurred in the combination chemotherapy group, and none occurred in the topotecan group. Conclusions Combination chemotherapy with cisplatin plus etoposide plus irinotecan could be considered a treatment option in second-line chemotherapy for selected patients with sensitive relapsed SCLC.However, the combination chemotherapy group had a higher incidence of adverse events than the topotecan group, and appropriate drug dosages should be explored. -
表 1 两组患者临床特征

表 2 拓扑替康组和联合化疗组完成各周期化疗(例)

表 3 治疗相关的3级/4级不良事件n(%)

-
[1] Bray F, Ferlay J, Soerjomataram I, et al.Global cancer statistics 2018:GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J].CA Cancer J Clin, 2018, 68(6): 394-424. doi: 10.3322/caac.v68.6 [2] Hansen HH.Management of small-cell cancer of the lung[J].Lancet, 1992, 339(8797):846-849. doi: 10.1016/0140-6736(92)90287-D [3] Kim YH, Goto K, Yoh K, et al.Performance status and sensitivity to first-line chemotherapy are significant prognostic factors in patients with recurrent small cell lung cancer receiving second-line chemotherapy[J].Cancer, 2008, 113(9):2518-2523. doi: 10.1002/cncr.v113:9 [4] Giaccone G, Donadio M, Bonardi G, et al.Teniposide in the treatment of small-cell lung cancer:the influence of prior chemotherapy[J].J Clin Oncol, 1988, 6(8):1264-1270. doi: 10.1200/JCO.1988.6.8.1264 [5] Owonikoko TK, Behera M, Chen Z, et al.A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer[J].J Thorac Oncol, 2012, 7(5):866-872. doi: 10.1097/JTO.0b013e31824c7f4b [6] O'Brien ME, Ciuleanu TE, Tsekov H, et al.Phase Ⅲ trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer[J].J Clin Oncol, 2006, 24(34):5441-5447. doi: 10.1200/JCO.2006.06.5821 [7] von Pawel J, Schiller JH, Shepherd FA, et al.Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer[J].J Clin Oncol, 1999, 17(2):658-667. doi: 10.1200/JCO.1999.17.2.658 [8] Eckardt JR, von Pawel J, Pujol JL, et al.Phase Ⅲ study of oral compared with intravenous topotecan as second-line therapy in smallcell lung cancer[J].J Clin Oncol, 2007, 25(15):2086-2092. doi: 10.1200/JCO.2006.08.3998 [9] von Pawel J, Jotte R, Spigel DR, et al.Randomized phase Ⅲ trial of amrubicin versus topotecan as second-line treatment for patients with small-cell lung cancer[J].J Clin Oncol, 2014, 32(35):4012 - 4019. doi: 10.1200/JCO.2013.54.5392 [10] Goto K, Ohe Y, Shibata T, et al.Combined chemotherapy with cisplatin, etoposide, and irinotecan versus topotecan alone as sec-ondline treatment for patients with sensitive relapsed small-cell lung cancer (JCOG0605):a multicentre, open-label, randomised phase 3 trial[J].Lancet Oncol, 2016, 17(8):1147-1157. doi: 10.1016/S1470-2045(16)30104-8 [11] Groen HJ, Fokkema E, Biesma B, et al.Paclitaxel and carboplatin in the treatment of small-cell lung cancer patients resistant to cyclophosphamide, doxorubicin, and etoposide:a non-cross-resistant schedule[J].J Clin Oncol, 1999, 17(3):927-932. doi: 10.1200/JCO.1999.17.3.927 [12] Hoang T, Kim K, Jaslowski A, et al.Phase Ⅱ study of second-line gemcitabine in sensitive or refractory small cell lung cancer[J].Lung Cancer, 2003, 42(1):97-102. http://europepmc.org/abstract/MED/14512193 [13] Jalal S, Ansari R, Govindan R, et al.Pemetrexed in second line and beyond small cell lung cancer:a Hoosier oncology group phase Ⅱ study[J].J Thorac Oncol, 2009, 4(1):493-496. http://www.ncbi.nlm.nih.gov/pubmed/19096313 [14] Einhorn LH, Pennington K, McClean J.PhaseⅡ trial of daily oral VP- 16 in refractory small cell lung cancer:a Hoosier Oncology Group study[J].Semin Oncol, 1990, 17(1 Suppl 2):32-35. http://www.ncbi.nlm.nih.gov/pubmed/2154857 [15] Johnson DH, Greco FA, Strupp J, et al.Prolonged administration of oral etoposide in patients with relapsed or refractory small-cell lung cancer:a phase Ⅱ trial[J].J Clin Oncol, 1990, 8(10):1613-1617. doi: 10.1200/JCO.1990.8.10.1613 [16] Masuda N, Fukuoka M, Kusunoki Y, et al.CPT-11:a new derivative of camptothecin for the treatment of refractory or relapsed smallcell lung cancer[J].J Clin Oncol, 1992, 10(8):1225-1229. doi: 10.1200/JCO.1992.10.8.1225 [17] Postmus PE, Berendsen HH, van Zandwijk N, et al.Retreatment with the induction regimen in small cell lung cancer relapsing after an initial response to short term chemotherapy[J].Eur J Cancer Clin Oncol, 1987, 23(9):1409-1411. doi: 10.1016/0277-5379(87)90128-3 [18] Giaccone G, Ferrati P, Donadio M, et al.Reinduction chemotherapy in small cell lung cancer[J].Eur J Cancer Clin Oncol, 1987, 23(11): 1697-1699. doi: 10.1016/0277-5379(87)90452-4 [19] Sekine I, Nishiwaki Y, Kakinuma R, et al.Phase Ⅰ/Ⅱ trial of weekly cisplatin, etoposide, and irinotecan chemotherapy for metastatic lung cancer:JCOG 9507[J].Br J Cancer, 2003, 88(6):808-813. doi: 10.1038/sj.bjc.6600800 -




 下载:
下载: