-
摘要:
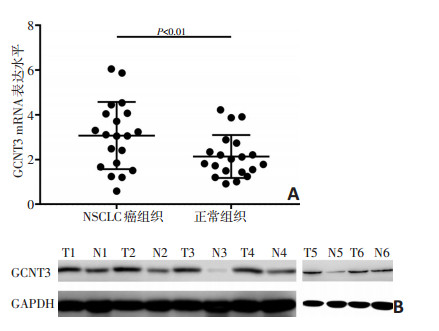
目的 研究葡萄糖胺基转移酶3(glycosyltransferase enzyme 3, GCNT3)在非小细胞肺癌(non-small cell lung cancer, NSCLC)组织及对应正常组织中的表达情况, 探讨其表达水平与NSCLC患者临床病理特征、总生存期(overall survival, OS)和无进展生存期(progression-free survival, PFS)的关系。 方法 分别应用实时定量反转录链式反应(qRT-PCR)和蛋白质印记技术(Western blot, WB)检测GCNT3在2017年3月至2017年7月天津医科大学肿瘤医院20例NSCLC患者癌组织及对应的正常组织中的表达情况; 此外, 收集2010年1月至2014年12月本院164例NSCLC患者的石蜡组织标本, 通过免疫组织化学技术(immunohistochemistry, IHC)评估样本中GCNT3表达, 分析GCNT3表达水平与临床病理学特征之间的关系, 探究GCNT3表达与NSCLC患者OS及PFS的关系, 通过细胞功能实验研究GCNT3对NSCLC细胞增殖能力、侵袭能力和迁移能力的影响。 结果 qRT-PCR与WB结果均显示GCNT3在NSCLC癌组织中的表达水平明显高于正常组织。IHC结果显示GCNT3表达水平与NSCLC患者的性别、吸烟史、组织学类型、病理分期和淋巴结转移相关; Kaplan-Meier分析显示GCNT3高表达NSCLC患者OS和PFS均差于GCNT3低表达患者(P < 0.05)。多因素Cox比例风险回归模型分析显示, GCNT3表达是NSCLC患者预后的独立因素(P < 0.05)。抑制GCNT3表达后, NSCLC细胞的增殖能力、侵袭能力和迁移能力明显减弱(P < 0.05)。 结论 GCNT3在NSCLC癌组织中高表达, GCNT3高表达的NSCLC患者OS和PFS较差, GCNT3有望成为评估NSCLC患者预后的标志物。 Abstract:Objective To investigate the expression of glycosyltransferase enzyme 3 (GCNT3) in non-small cell lung cancer (NSCLC) tissues and corresponding normal tissues, and to further explore the relationship between GCNT3 expression and clinicopathological features, overall survival (OS), and progression-free survival (PFS) in patients with NSCLC. Methods In this study, we used quantitative real-time polymerase chain reaction and Western blot to assess the mRNA and protein expression of GCNT3 in paired NSCLC and non-tumor tissues. In addition, 164 NSCLC patients were estimated for GCNT3 expression by immunohistochemistry, and the correlation between GCNT3 expression and clinicopathological features was evaluated. Further, the effects of GCNT3 on the proliferation, invasion, and migration abilities of NSCLC cells were studied. Results The mRNA and protein expression levels of GCNT3 in NSCLC tissues were both significantly higher than those in the corresponding non-tumor tissues. Among the 164 patients with NSCLC, high GCNT3 expression was associated with gender, smoking, histology, pathological stage, and lymph node metastasis. Kaplan-Meier analysis displayed significant differences in OS and PFS among the groups exhibiting differences in GCNT3 expression (P < 0.05). The NSCLC patients with increased GCNT3 expression showed poor OS and PFS. A multivariate analysis demonstrated that GCNT3 expression was as an independent prognostic factor for NSCLC (P < 0.05). Cell function experiments showed that the proliferation, invasion, and migration abilities of NSCLC cells were significantly attenuated after inhibition of GCNT3 expression (P < 0.05). Conclusions High expression of GCNT3 was associated with unfavorable OS and PFS in patients with NSCLC; GCNT3 might, therefore, act as a prognostic biomarker for NSCLC. Key words glycosyltransferase enzyme 3 (GCNT3), non-small cell lung cancer (NSCLC), immunohistochemistry, prognosis. -
表 1 GCNT3表达与NSCLC临床病理特征的关系

表 2 影响NSCLC患者预后的单因素分析

表 3 影响NSCLC患者预后的多因素分析

-
[1] Torre LA, Bray F, Siegel RL, et al.Global cancer statistics, 2012[J].CA Cancer J Clin, 2015, 65(2):69-90. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0234398683/ [2] Rivera MP.Multimodality therapy in the treatment of lung cancer [J].Semin Respir Crit Care Med, 2004, 1:3-10. http://d.old.wanfangdata.com.cn/OAPaper/oai_pubmedcentral.nih.gov_3463922 [3] Spira A, Ettinger DS.Multidisciplinary management of lung cancer [J].N Engl J Med, 2004, 350(4):379-392. doi: 10.1056/NEJMra035536 [4] Hong Q, Wu G, Qian G, et al.Prevention and management of lung cancer in China[J].Cancer, 2015, 121(S17):3080-3088. doi: 10.1002/cncr.29584 [5] Markine-Goriaynoff N, Gillet L, Karlsen OA, et al.The core 2 beta-1, 6-N-acetylglucosaminyltransferase-M encoded by bovine herpesvirus 4 is not essential for virus replication despite contributing to post-translational modifications of structural proteins[J].J General Virol, 2004, 85(pt2):355-367. http://www.ncbi.nlm.nih.gov/pubmed/14769893/ [6] Huang MC, Chen HY, Huang HC, et al.C2GnT-M is downregulated in colorectal cancer and its re-expression of colon cancer cells[J].Oncogene, 2006, 25(23):3267-3276. doi: 10.1038/sj.onc.1209350 [7] Brockhausen I, Kuhns W, Schachter H, et al.Biosynthesis of O-glycans in leukocytes from normal donors and from patients with leukemia:increase in O-glycan core 2 UDP-GlcNAc:galbeta3GalNacalpha-R (GlcNAc to GalNAc) beta (1-6)-N-acetylglucosaminyltransferase in leukemic cells[J].Cancer Res, 1991, 51(4):1257-1263. http://www.ncbi.nlm.nih.gov/pubmed/1997166 [8] Rao CV, Janakiram NB, Madka V, et al.Small molecule inhibition of gcnt3 disrupts mucin biosynthesis and malignant cellular behaviors in pancreatic cancer[J].Cancer Res, 2016, 76(7):1965. doi: 10.1158/0008-5472.CAN-15-2820 [9] Travis WD, Brambilla E, Nicholson AG, et al.The 2015 world health organization classification of lung tumors:impact of genetic, clinical and radiologic advances since the 2004 classification[J].J Thorac Oncol, 2015, 10(9):1243-1260. doi: 10.1097/JTO.0000000000000630 [10] Goldstraw P, Chansky K, Crowley J, et al.The IASLC lung cancer staging project:proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer[J].J Thorac Oncol, 2016, 11(1):39-51. doi: 10.1016/j.jtho.2015.09.009 [11] Yeh JC, Ong E, Fukuda M.Molecular cloning and expression of a novel beta-1, 6-N-acetylglucosaminyltransferase that forms core 2, core 4, and I branches[J].J Biological Chemistry, 1999, 274(5):3215-3221. doi: 10.1074/jbc.274.5.3215 [12] Schwientek T, Nomoto M, Levery SB, et al.Control of O-glycan branch formation.Molecular cloning of human cDNA encoding a novel beta1, 6-N-acetylglucosaminyltransferase forming core 2 and core 4[J].J Biol Chem, 1999, 274(8):4504-4512. doi: 10.1074/jbc.274.8.4504 [13] Zynda ER, Schott B, Gruener S, et al.An RNA interference screen identifies new avenues for nephroprotection[J].Cell Death Differ, 2015, 23(4):608. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=5c3d38f3fe6598aeb5fce0e0e0ad1368 [14] Jeffries JL, Jing J, Woosuk C, et al.Pseudomonas aeruginosapyocyanin modulates mucin glycosylation with sialyl-Lewisxto increase binding to airway epithelial cells[J].Mucosal Immunology, 2016, 9 (4):1039-1050. doi: 10.1038/mi.2015.119 [15] Liu T, Zhang S, Chen J, et al.The transcriptional profiling of glycogenes associated with hepatocellular carcinoma metastasis[J].Plos One, 2014, 9(9):107941. doi: 10.1371/journal.pone.0107941 [16] Sumardika IW, Youyi C, Kondo E, et al.β-1, 3-galactosyl-O-glycosylglycoprotein ß-1, 6-N-acetylglucosaminyltransferase 3 increases mcam stability, which enhances s100a8/a9-mediated cancer motility[J]. Oncol Res, 2018, 26(3):431-444. doi: 10.3727/096504017X15031557924123 [17] Khan GJ, Gao Y, Gu M, et al.TGF-β1 Causes EMT by regulating NAcetyl glucosaminyl transferases via downregulation of non muscle myosin Ⅱ-A through JNK/P38/PI3K pathway in lung cancer[J].Current Cancer Drug Targets, 2018, 18(2):209-219. http://europepmc.org/abstract/MED/28782471 [18] Gonzálezvallinas M, Vargas T, Morenorubio J, et al.Clinical relevance of the differential expression of the glycosyltransferase gene GCNT3 in colon cancer[J].Eur J Cancer, 2015, 51:1-8. doi: 10.1016/j.ejca.2014.10.021 [19] Taniguchi N, Korekane H.Branched N-glycans and their implications for cell adhesion, signaling and clinical applications for cancer biomarkers and in therapeutics[J].BMB Rep, 2001, 44(4):772-781. http://europepmc.org/abstract/med/22189679 -




 下载:
下载: