Screening and immune activity detection of neoantigen vaccine targeting epidermal growth factor receptor mutation of non-small cell lung cancer
-
摘要:
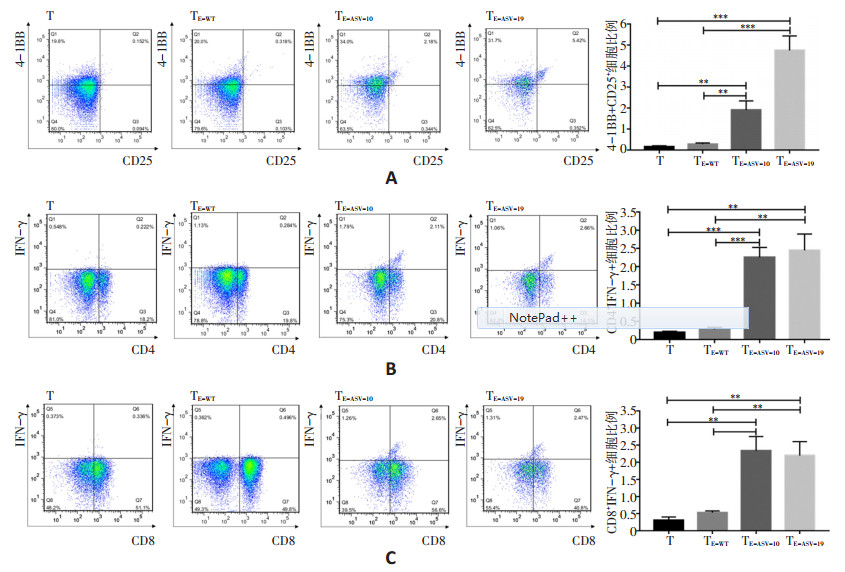
目的 筛选具有免疫源性HLA-A*02限制性表皮生长因子受体(epidermal growth factor receptor,EGFR)exon20插入突变编码的优势表位肽,为携带EGFR exon20插入突变的非小细胞肺癌(non-small cell lung cancer,NSCLC)提供新的免疫治疗手段。 方法 通过IEDB、NetMHC 4.0和SYFPEITHI软件,筛选EGFR exon20插入突变编码的HLA-A*02限制性表位,针对细胞毒性T淋巴细胞表位集中区域设计多肽疫苗,通过体外实验验证其免疫活性。 结果 V769_D770insASV为EGFR exon20插入突变最高频突变位点(19.35%),经软件预测其编码的多肽YVMASVASV与HLA-A*02具有较强的结合力。围绕核心序列设计两条优势表位多肽E-ASV-10和E-ASV-19,体外可诱导HLA-A*02限制性T细胞的扩增和活化,上调4-1BB+CD25+细胞比例,促进CD4+、CD8+T细胞IFN-γ的合成和释放。多肽E-ASV-10可刺激C57BL/6小鼠的T细胞分泌IFN-γ,对携带V769_D770insASV的小鼠NSCLC细胞LLCasv具有杀伤活性。 结论 E-ASV-10和E-ASV-19多肽不仅可以在体外诱导人HLA-A*02限制性T细胞的扩增和活化,而且在C57BL/6小鼠中诱导特异性靶向携带V769_D770insASV突变的小鼠肺腺癌(lewis lung cancer,LLC)的杀伤性T细胞,提示其作为一种新的肿瘤疫苗或可用于治疗携带相应EGFR exon20插入突变的NSCLC,具有潜在临床应用前景。 Abstract:Objective This study aims to screen the optional epitope peptides of HLA-A*02 restricted human epidermal growth factor receptor (EGFR) exon20 insertion mutations in order to provide a novel immunotherapeutic strategy for non-small cell lung cancer (NSCLC). Methods HLA-A*02 restricted epitopes derived from EGFR exon20 insertion mutation were predicted by IEDB, NetMHC4.0 and SYFPEITHI. The cytotoxic T lymphocyte (CTL) epitope-concentrated area was analyzed, and the appropriate length of polypeptides were filtered. Both cell experiments and mice models were implemented to verify the immunogenicity and antitumor efficiency of the candidate polypeptides. Results V769_D770insASV (19.35%) mutation is a high-frequency EGFR exon20 insertion in NSCLC, which encoded YVMASVASV polypeptide with higher MHC class I binding score than other insertions. E-ASV-10 and E-ASV-19, both possessing the core sequence of YVMASVASV polypeptide, were capable to induce the expansion and activation of HLA-A*02 restricted T cells in vitro, displaying increase in the proportion of 4-1BB+CD25+ population and the production and release of IFN-γ in CD4+ and CD8+ T cells. E-ASV-10 stimulated the amplification of cytotoxic T cells against the LLC cells carrying th eEGFR exon20 V769_D770insASVmutation(LLCasv) in C57BL/6 mice. Conclusions E-ASV-10 and E-ASV-19 could induce the expansion and activation of human HLA-A* 02 restricted T cells in vitro, and stimulate specific cytotoxic CTL response against LLCasv in C57BL/6 mice, which might become an promosing immunotherapeutic approach for NSCLC patients carrying EGFR exon20 V769_D770insASV mutation. -
表 1 EGFR V769_D770insASV HLA-A*02:01限制性CTL表位预测结果

表 2 候选表位肽体外诱导人T淋巴细胞分泌IFN-γ的ELISPOT实验结果

-
[1] Graham RP, Treece AL, Lindeman NI, et al. Worldwide frequency of commonly detected egfr mutations[J]. Arch Pathol Lab Med, 2018, 142(2):163-167. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=87f3e9e4fd8fd2eba0fd2e5c2cee2f36 [2] Yatabe Y, Kerr KM, Utomo A, et al. EGFR mutation testing practices within the asia pacific region:Results of a multicenter diagnostic survey[J]. J Thorac Oncol, 2015, 10(3):438-445. http://cn.bing.com/academic/profile?id=28b57d8493fc610febbbcc2401e128b6&encoded=0&v=paper_preview&mkt=zh-cn [3] Hsu WH, Yang JCH, Mok TS, et al. Overview of current systemic management of EGFR-mutant NSCLC[J]. Ann Oncol, 2018, 29(Suppl_1):i3-i9. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=71d86d9e7b0e17453a3ddec3ac06de79 [4] Sequist LV, Yang JC, Yamamoto N, et al. Phase Ⅲ study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations[J]. J Clin Oncol, 2013, 31(27):3327-3334. https://www.ncbi.nlm.nih.gov/pubmed/23816960 [5] Naidoo J, Sima CS, Rodriguez K, et al. Epidermal growth factor receptor exon 20 insertions in advanced lung adenocarcinomas:Clinical outcomes and response to erlotinib[J]. Cancer, 2015, 121(18):3212-3220. http://cn.bing.com/academic/profile?id=2e676f006a6fa8d37ba9b591be3e3ba0&encoded=0&v=paper_preview&mkt=zh-cn [6] Tu HY, Ke EE, Yang JJ, et al. A comprehensive review of uncommon egfr mutations in patients with non-small cell lung cancer[J]. Lung Cancer, 2017, 114:96-102. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0d7e67705d3e61a8ebd2fee15868fc61 [7] Hasegawa H, Yasuda H, Hamamoto J, et al. Efficacy of afatinib or osimertinib plus cetuximab combination therapy for non-small-cell lung cancer with EGFR exon 20 insertion mutations[J]. Lung Cancer, 2019, 127:146-152. http://cn.bing.com/academic/profile?id=be18f6c4362182becf533b98c275c745&encoded=0&v=paper_preview&mkt=zh-cn [8] Ruan Z, Kannan N. Altered conformational landscape and dimerization dependency underpins the activation of EGFR by alphac-beta4 loop insertion mutations[J]. Proc Natl Acad Sci U S A, 2018, 115(35):e8162-e8171. [9] Chen K, Yu X, Wang H, et al. Uncommon mutation types of epidermal growth factor receptor and response to EGFR tyrosine kinase inhibitors in chinese non-small cell lung cancer patients[J]. Cancer Chemother Pharmacol, 2017, 80(6):1179-1187. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=76b5b11bf35c8497ccbc9a61c12c90e0 [10] Byeon S, Kim Y, Lim SW, et al. Clinical outcomes of EGFR exon 20 insertion mutations in advanced non-small cell lung cancer in korea[J]. Cancer Res Treat, 2019, 51(2):623-631. http://cn.bing.com/academic/profile?id=f6494acd95a5d9b2e738f6926ed4aa86&encoded=0&v=paper_preview&mkt=zh-cn [11] Miura Y, Sunaga N. Role of immunotherapy for oncogene-driven nonsmall cell lung cancer[J]. Cancers (Basel), 2018, 10(8):245 [12] Garassino MC, Cho B-C, Kim J-H, et al. Durvalumab as third-line or later treatment for advanced non-small-cell lung cancer (atlantic):An openlabel, single-arm, phase 2 study[J]. The Lancet Oncol, 2018, 19(4):521-536. https://www.thelancet.com/journals/lanonc/article/PIIS1470-2045(18)30144-X/fulltext [13] Yarchoan M, Johnson BA 3rd, Lutz ER, et al. Targeting neoantigens to augment antitumour immunity[J]. Nat Rev Cancer, 2017, 17(4):209-222. http://cn.bing.com/academic/profile?id=7d7571a1baefdb4b4be4595f664380dc&encoded=0&v=paper_preview&mkt=zh-cn [14] Smith CC, Selitsky SR, Chai S, et al. Alternative tumour-specific antigens[J]. Nat Rev Cancer, 2019, 19(8):465-478. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ02995235/ [15] Hartmaier RJ, Charo J, Fabrizio D, et al. Genomic analysis of 63, 220 tumors reveals insights into tumor uniqueness and targeted cancer immunotherapy strategies[J]. Genome Med, 2017, 9(1):16. http://cn.bing.com/academic/profile?id=80da9cfed0f6594e1dd178d663ebe700&encoded=0&v=paper_preview&mkt=zh-cn [16] Akya A, Farasat A, Ghadiri K, et al. Identification of HLA-i restricted epitopes in six vaccine candidates of leishmania tropica using immunoinformatics and molecular dynamics simulation approaches[J]. Infect Genet Evol, 2019.DOI: 10.1016/j.meegid.2019.103953. [17] Li Z, Zhang F, Zhang C, et al. Immunoinformatics prediction of OMP2b and BCSP31 for designing multi-epitope vaccine against brucella[J]. Mol Immunol, 2019, 114:651-660. [18] Kosaka T, Tanizaki J, Paranal RM, et al. Response heterogeneity of egfr and HER2 exon 20 insertions to covalent EGFR and HER2 inhibitors[J]. Cancer Res, 2017, 77(10):2712-2721. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a2c6a66937cc62d1d337e9f77d5a53c6 [19] Kobayashi Y, Mitsudomi T. Not all epidermal growth factor receptor mutations in lung cancer are created equal:Perspectives for individualized treatment strategy[J]. Cancer Sci, 2016, 107(9):1179-1186. http://cn.bing.com/academic/profile?id=3a40617e1ee942f9f1ec06eea010480a&encoded=0&v=paper_preview&mkt=zh-cn [20] Deweerdt S. Calling cancer's bluff with neoantigen vaccines[J]. Nature, 2017, 552(7685):s76-s77. http://cn.bing.com/academic/profile?id=91fcb0076e4b203631f4b0786f07a5e0&encoded=0&v=paper_preview&mkt=zh-cn [21] Ott PA, Hu Z, Keskin DB, et al. An immunogenic personal neoantigen vaccine for patients with melanoma[J]. Nature, 2017, 547(7662):217-221. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c8fe0fcb67a683ef79f11863e9bd550f [22] Sahin U, Derhovanessian E, Miller M, et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer[J]. Nature, 2017, 547(7662):222-226. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d395d402e7b7f8439f612ac6c3f65a73 [23] Hellmann MD, Snyder A. Making it personal:neoantigen vaccines in metastatic melanoma[J]. Immunity, 2017, 47(2):221-223. http://cn.bing.com/academic/profile?id=74e14a1488ef2ccd3c132bcf17c057ed&encoded=0&v=paper_preview&mkt=zh-cn [24] Wu D, Gao Y, Qi Y, et al. Peptide-based cancer therapy:opportunity and challenge[J]. Cancer Lett, 2014, 351(1):13-22. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0210419394/ [25] Oh CY, Klatt MG, Bourne C, et al. ALK and RET inhibitors promote HLA class i antigen presentation and unmask new antigens within the tumor immunopeptidome[J]. Cancer Immunol Res, 2019, 7(12):1984-1997. [26] Akbay EA, Koyama S, Carretero J, et al. Activation of the PD-1 pathway contributes to immune escape in egfr-driven lung tumors[J]. Cancer Discov, 2013, 3(12):1355-1363. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0a2de6fe9760fe6721890ee511dbd38f [27] Forloni M, Albini S, Limongi MZ, et al. Nf-κB, and not MYCN, regulates mhc class i and endoplasmic reticulum aminopeptidases in human neuroblastoma cells[J]. Cancer Res, 2010, 70(3):916-924. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_JJ0215636570 [28] Kitamura H, Ohno Y, Toyoshima Y, et al. Interleukin-6/STAT3 signaling as a promising target to improve the efficacy of cancer immunotherapy[J]. Cancer Sci, 2017, 108(10):1947-1952. http://cn.bing.com/academic/profile?id=df0b4bb9f5a0d4435e0f2d579432301a&encoded=0&v=paper_preview&mkt=zh-cn [29] Mimura K, Kua LF, Shiraishi K, et al. Inhibition of mitogen-activated protein kinase pathway can induce upregulation of human leukocyte antigen class i without pd-l1-upregulation in contrast to interferongamma treatment[J]. Cancer Sci, 2014, 105(10):1236-1244. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4462358/ -




 下载:
下载: