Prospective study on paclitaxel combined with apatinib mesylate in comparison with paclitaxel alone as the second-line treatment for gastric cancer
-
摘要:
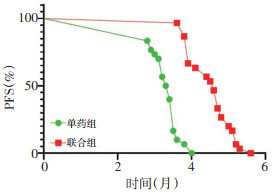
目的 观察紫杉醇联合阿帕替尼对比单药紫杉醇二线治疗胃癌的疗效及安全性。 方法 选取2017年3月至2018年3月就诊于内蒙古医科大学赤峰临床医学院经一线(氟尿嘧啶类联合铂类)治疗后进展的HER-2阴性的晚期胃癌患者60例,分为单药组和联合组,每组30例。单药组给予单药紫杉醇化疗,联合组给予紫杉醇联合阿帕替尼治疗,观察的主要终点为两组患者的无进展生存期(progression-free survival,PFS),次要终点为疾病控制率(disease control rate,DCR)、客观缓解率(objective response rate,ORR)、安全性。 结果 联合组患者的PFS优于单药组患者,两组相比差异有统计学意义(P < 0.001)。联合组DCR、ORR均高于单药组,但差异均无统计学意义(均P>0.05);联合组高血压发生率高于单药组(P < 0.05),其余不良反应发生率差异均无统计学意义(均P>0.05)。 结论 紫杉醇联合甲磺酸阿帕替尼对比单药紫杉醇二线治疗胃癌,联合组PFS、DCR、ORR均优于单药组,虽然联合组DCR、ORR较单药组相比差异无统计学意义,但联合组组患者的PFS较单组延长,差异有统计学意义(P < 0.05),且联合组不良反应可耐受,安全性良好。 Abstract:Objective To investigate the efficacy and safety of paclitaxel combined with apatinib in comparison with paclitaxel alone as the second-line treatment for gastric cancer. Methods Patients with advanced gastric cancer who had been treated at Chifeng Municipal Hospital, Chifeng Clinical Medical School of inner Mongolia Medical University, from March 2017 to March 2018 were enrolled. Inclusion criteria were human epidermal growth factor receptor-2(HER-2)-negative cancer and progression after the first-line treatment with fluorouracil combined with platinum. Patients were divided into groups administered with a single drug and combination of drugs. The single-drug group was administered with paclitaxel chemotherapy, while the combined-drug group with the paclitaxel combined with apatinib treatment. In both groups, the primary endpoint of observation was progression-free survival (PFS), while the secondary endpoint was the disease control rate (DCR), overall response rate (ORR), and safety. Results A total of 60 patients were enrolled, including 30 patients in each of single- and combined-drug groups. PFS was significantly better in the combined-drug group than in the single-drug group (P < 0.001). DCR and ORR were higher in the combined-drug group than in the single-drug group, but without statistical significance (P>0.05). The incidence of hypertension was significantly higher in the combined-drug group than in the single-drug group (P < 0.05), while the incidence of other adverse reactions showed no significant difference between the groups (P>0.05). Conclusions Paclitaxel combined with apatinib mesylate is superior to paclitaxel alone in the second-line treatment of gastric cancer. PFS, DCR, and ORR are superior with paclitaxel combined with apatinib mesylate than with paclitaxel alone. Although DCR and ORR in the combined-drug group were not significantly different from those in the single-drug group, the PFS was significantly longer in the combined-drug group, and the toxic and side effects of paclitaxel combined with apatinib mesylate were tolerable and safe. -
Key words:
- gastric cancer /
- paclitaxel /
- apatinib /
- efficacy /
- safety
-
表 1 患者临床病例资料

表 2 联合组与单药组治疗效果比较

表 3 联合组与单药组血液学毒性观察

表 4 联合组与单药组非血液学毒性观察

-
[1] Mi YJ, Liang YJ, Huang HB, et al. Apatinib (YN968D1) Reverses multidrug resistance by inhibiting the efflux function of multiple ATP-binding cassette transporters[J]. Cancer Res, 2010, 70(20):7981-7991. doi: 10.1158/0008-5472.CAN-10-0111 [2] Li J, Zhao XM, Chen L, et al. Safety and pharmacokinetics of novel selective vascular endothelial growth factor receptor- 2 inhibitor YN968D1 in patients with advanced malignancies[J]. BMC cancer, 2010, 10:529. doi: 10.1186/1471-2407-10-529 [3] Hu X, Zhang J, Xu B, et al. Multicenter Phase Ⅱ study of apatinib, a novel VEGFR inhibitor in heavily pretreated patients with metastatic triple negativebreast cancer[J]. Int J Cancer, 2014, 135(8):1961-1969. doi: 10.1002/ijc.28829 [4] Wang B, Song LJ, Niu PY, et al. Clinical efficacy of Apatinib in treatment of advanced gastric cancer[J]. World Chin J Digestol, 2016, 24(5):759-764. doi: 10.11569/wcjd.v24.i5.759 [5] Siegel RL, Miller KD, Jemal A.Cancer Statistics, 2017[J]. CA Cancer J Clin, 2017, 67(l):7-30. http://cn.bing.com/academic/profile?id=ad58510107c8a5ac70b26bea5a99765f&encoded=0&v=paper_preview&mkt=zh-cn [6] Chen W, Sun K, Zheng R, et al. Cancer incidence and mortality in China, 2014[J]. Clin J Cancer Res, 2018, 30(1):1-12. http://cn.bing.com/academic/profile?id=5b4a7a2075c5dda14222b96845394163&encoded=0&v=paper_preview&mkt=zh-cn [7] Smalley SR, Benedetti JK, Haller DG, et al. Updated analysis of SWOGdirected intergroup study 0116: a phase III trial of adjuvantradiochemotherapy versus observation after curative gastric Cancer resection[J]. J Clin Oncol, 2012, 30(19):2327-2333. doi: 10.1200/JCO.2011.36.7136 [8] Iacovelli R, Pietrantonio F, Maggi C, et al. Combination or single-agent chemotherapy as adjuvant treatment of gastric cancer:A systematic review and meta- analysis of published trials[J]. Crit Rev Oneol Hematol, 2016, 98:24-28. doi: 10.1016/j.critrevonc.2015.09.002 [9] Lordick F, Lorenzen S, Yamam Y, et al. Optimal chemotherapy for advanced gastric cancer:is there a global consensus[J]? Gastric Cancer, 2014, 17(2):213-225. doi: 10.1007/s10120-013-0297-z [10] Kang BW, Kwon OK, Chung HY, et al. Taxanes in the treatment of advanced gastric cancer[J]. Molecules, 2016, 21(5):651. doi: 10.3390/molecules21050651 [11] Savry A, Carre M, Berges R, et al. Bcl-2-enhanced efficacy of microtubuletargeting chemotherapy through bim overexpression:implications for cancer treatment[J]. Neoplasia, 2013, 15(1):49-60. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_PM23358890 [12] Ingemarsdotter CK, Baird SK, Connell CM, et al. Low-dose paclitaxel synergizes with oncolytic adenoviruses via mitotic slippage and apoptosis in ovarian cancer[J]. Oncogene, 2010, 29(45):6051-6063. doi: 10.1038/onc.2010.335 [13] Pan Z, Avila A, Gollahon L. Paclitaxel induces apoptosis in breast cancer cells through different calcium-regulating mechanisms depending on external calcium conditions[J]. Int J Mol Sci, 2014, 15(2):2672-2694. doi: 10.3390/ijms15022672 [14] Wang Y, Liu Y, Li G, et al. Ephrin typeA receptor 2 regulates sensitivity to paclitaxel in nasopharyngeal carcinoma via the phosphoinositide 3kinase/Akt signalling pathway[J]. Mol Med Rep, 2015, 11(2):924-930. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_PM23358890 [15] Hu J, Zhang NA, Wang RL, et al. Paclitaxel induces apoptosis and reduces proliferation by targeting epidermal growth factor receptor signaling pathway in oral cavity squamous cell carcinoma[J]. Oncol Lett, 2015, 10(4):2378-2384. doi: 10.3892/ol.2015.3499 [16] Zhang XY, Zhang YD. Enhanced antiproliferative and apoptosis effect of paclitaxel-loaded polymeric micelles against non-small cell lung cancers[J]. Tumour Biol, 2015, 36(7):4949-4959. doi: 10.1007/s13277-015-3142-7 [17] Kamazwa S, Kigawa J, Kanamori Y, et al. Multidurg resistance gene-l is a useful predictor of Paclitaxel-based chemotherapy for patients with ovarian cancer[J].Gynecol oncol, 2002, 86(2):171-176. doi: 10.1006/gyno.2002.6738 [18] Peter Carmeliet. VEGF as a key mediator of angiogenesis in cancer[J]. Oncology, 2005, 69 (Suppl 3):4-10. http://cn.bing.com/academic/profile?id=092ee865573c0972b2f13e97e6227be0&encoded=0&v=paper_preview&mkt=zh-cn -




 下载:
下载: