Therapeutic evaluation of multidisciplinary treatment for 71 cases of rhabdomyosarcoma in children
-
摘要:
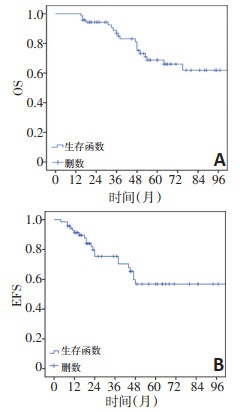
目的 评价横纹肌肉瘤(rhabdomyosarcoma,RMS)多学科诊疗草案重庆方案(CQ-RMS-2009)多学科综合治疗(multidisciplinary treatment,MDT)儿童RMS的有效性,探讨RMS预后因素。 方法 回顾性分析2006年5月至2016年4月于上海交通大学医学院附属新华医院治疗、年龄 <14岁RMS患儿临床资料。比较不同因素对RMS患儿长期生存的影响。 结果 71例RMS患儿纳入研究,男女比例1.84:1;中位诊断年龄为47(2~166)个月,中位随访时间52(15~165)个月。5年总体生存率(5-year overall survival,5y-OS)和5年无事件生存率(5-year event free survival,5y-EFS)分别为(61.9±5.1)%和(56.1±4.2)%。2006年5月至2009年12月收治的患儿5y-OS为(48.1±12.1)%,2010年1月至2016年4月收治患儿的5-OS为(73.3±7.9)%,差异具有统计学意义(P=0.042);5y-EFS分别为(35.9±11.7)%和(69.8±8.7)%,差异无统计学意义(P=0.064)。单因素分析显示,病理类型、术后临床分组、治疗模式、危险度分组是影响RMS患儿总体生存率(overall survival,OS)的重要因素(均P<0.05)。多因素分析显示,术后临床分组及治疗模式是影响患儿预后的独立因素(均P<0.05)。 结论 当RMS确诊时,多数患儿处于中晚期,OS偏低,MDT模式可有效改善RMS患儿的预后,降低晚期患儿复发/疾病进展率,对于疾病复发/进展后的局部控制同样有明显效果。 Abstract:Objective To explore the effectiveness and feasibility of multidisciplinary treatment (MDT) based on the CQ-RMS-2009 protocol and analyze the association of prognostic factors with the long-term survival of children with rhabdomyosarcoma (RMS). Methods Medical records of patients with RMS under 14 years of age at Xinhua Hospital, Shanghai Jiao Tong University School of Medicine, from May 2006 to April 2016 were analyzed. The association of different factors with the long-term survival of children with RMS was compared. Results In total, 71 cases of RMS in children were included. The male to female ratio was 1.84:1, median age at diagnosis was 47 (2-166) months, and median follow-up time was 52 (15-165) months. The 5-year overall survival (5y-OS) and 5-year event free survival (5y-EFS) was 61.9%±5.1% and 56.1%±4.2%. From May 2006 to December 2009 and from January 2009 to April 2016, the 5yOS was 48.1%±12.1% and 73.3%±7.9%, respectively (P=0.042), and the 5y-EFS was 35.9%±11.7% and 69.8%±8.7%, respectively (P=0.064). The univariate analysis showed that the pathological subtypes, postoperative clinical group (P=0.025), treatment patterns, and risk groups were significantly associated with overall survival (OS)(P<0.05). Multivariate analysis revealed that the postoperative clinical group and treatment patterns were independent prognostic factors (P<0.05). Conclusions On diagnosis, most of the children with RMS are already in the middle and late stages with low OS. MDT could effectively improve the prognosis and reduce the rate of relapse/progression in children with RMS, and it has remarkable efficacy for local control of disease relapse/progression. -
Key words:
- rhabdomyosarcoma (RMS) /
- multidisciplinary treatment (MDT) /
- prognosis /
- children
-
表 1 71例RMS患儿临床特征及生存分析

表 2 影响RMS患儿预后的多因素分析

-
[1] Dasgupta R, Fuchs J, Rodeberg D. Rhabdomyosarcoma[J]. Semin Pediatr Surg, 2016, 25(5):276-283. doi: 10.1053/j.sempedsurg.2016.09.011 [2] Shern JF, Yohe ME, Khan J. Pediatric rhabdomyosarcoma[J]. Crit Rev Oncog, 2015, 20(3-4):227-243. doi: 10.1615/CritRevOncog.2015013800 [3] Ognjanovic S, Linabery AM, Charbonneau B, et al. Trends in childhood rhabdomyosarcoma incidence and survival in the United States, 1975-2005[J]. Cancer, 2009, 115(18):4218-4226. doi: 10.1002/cncr.24465 [4] Smith MA, Altekruse SF, Adamson PC, et al. Declining childhood and adolescent cancer mortality[J]. Cancer, 2014, 120(16):2497-2506. doi: 10.1002/cncr.28748 [5] 汤静燕, 潘慈, 徐敏, 等.儿童横纹肌肉瘤"上海儿童医学中心Rs-99方案"临床报告[J].中国实用儿科杂志, 2003, 18(4):208-211. doi: 10.3969/j.issn.1005-2224.2003.04.009 [6] 中国抗癌协会小儿肿瘤专业委员会, 中华医学会儿科学分会血液学组, 中华医学会小儿外科学分会肿瘤组: 中国儿童及青少年横纹肌肉瘤诊疗建议(CCCG-RMS-2016)[J].中华儿科杂志, 2017, 55(10): 724-728. [7] Jo VY, Fletcher CD. WHO classification of soft tissue tumours:an update based on the 2013(4th) edition[J]. Pathology, 2014, 46(2):95-104. doi: 10.1097/PAT.0000000000000050 [8] Lawrence W, Anderson JR, Gehan EA, et al. Pretreatment TNM staging of childhood rhabdomyosarcoma:a report of the Intergroup Rhabdomyosarcoma Study Group. Children's Cancer Study Group. Pediatric Oncology Group[J]. Cancer, 1997, 80(6):1165-1170. doi: 10.1002/(SICI)1097-0142(19970915)80:6<1165::AID-CNCR21>3.0.CO;2-5 [9] Maurer HM, Gehan EA, Beltangady M, et al. The Intergroup Rhabdomyosarcoma Study-Ⅱ[J]. Cancer, 1993, 71(5):1904-1922. doi: 10.1002/1097-0142(19930301)71:5<1904::AID-CNCR2820710530>3.0.CO;2-X [10] Sangkhathat S. Current management of pediatric soft tissue sarcomas[J]. World J Clin Pediatr, 2015, 4(4):94-105. doi: 10.5409/wjcp.v4.i4.94 [11] Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours:revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2):228-247. doi: 10.1016/j.ejca.2008.10.026 [12] Crist W, Gehan EA, Ragab AH, et al. The third intergroup rhabdomyosarcoma study[J]. J Clin Oncol, 1995, 13(3):610-630. doi: 10.1200/JCO.1995.13.3.610 [13] Panda SP, Chinnaswamy G, Vora T, et al. Diagnosis and management of rhabdomyosarcoma in children and adolescents:ICMR consensus document[J]. Indian J Pediatr, 2017, 849(5):393-402. [14] Skapek SX, Ferrari A, Gupta AA, et al. Rhabdomyosarcoma[J]. Nat Rev Dis Primers, 2019, 5(1):1. [15] Fischer TD, Gaitonde SG, Bandera BC, et al. Pediatric-protocol of multimodal therapy is associated with improved survival in AYAs and adults with rhabdomyosarcoma[J]. Surgery, 2018, 163(2):324-329. doi: 10.1016/j.surg.2017.10.027 [16] Hendricks M, Parkes J, Pillay K, et al. Outcomes of children with rhabdomyosarcoma treated with intensive chemotherapy, surgery, and radiotherapy through a period of protocol revision at a South African center, 1990-2010[J]. Pediatr Blood Cancer, 2017, 64(3):10. [17] Rudzinski ER, Anderson JR, Chi YY, et al. Histology, fusion status, and outcome in metastatic rhabdomyosarcoma:a report from the Children's Oncology Group[J]. Pediatr Blood Cancer, 2017, 64(12):10. [18] Hibbitts E, Chi YY, Hawkins DS, et al. Refinement of risk stratification for childhood rhabdomyosarcoma using FOXO1 fusion status in addition to established clinical outcome predictors:A report from the Children's Oncology Group[J]. Cancer Med, 2019, 8(14):6437-6448. doi: 10.1002/cam4.2504 [19] Kazanowska B, Reich A, Stegmaier S, et al. Pax3-fkhr and pax7-fkhr fusion genes impact outcome of alveolar rhabdomyosarcoma in children[J]. Fetal Pediatr Pathol, 2007, 26(1):17-31. doi: 10.1080/15513810701394702 [20] Gallego S, Zanetti I, Orbach D, et al. Fusion status in patients with lymph node-positive (N1) alveolar rhabdomyosarcoma is a powerful predictor of prognosis:Experience of the European Paediatric Soft Tissue Sarcoma Study Group (EpSSG)[J]. Cancer, 2018, 124(15):3201-3209. doi: 10.1002/cncr.31553 [21] Yohe ME, Heske CM, Stewart E, et al. Insights into pediatric rhabdomyosarcoma research:challenges and goals[J]. Pediatr Blood Cancer, 2019, 66(10):e27869. [22] Borinstein SC, Steppan D, Hayashi M, et al. Consensus and controversies regarding the treatment of rhabdomyosarcoma[J]. Pediatr Blood Cancer, 2018, 65(2):10. [23] Antillon F, Castellanos M, Valverde P, et al. Treating pediatric soft tissue sarcomas in a country with limited resources:the experience of the Unidad Nacional de Oncologia Pediatrica in Guatemala[J]. Pediatr Blood Cancer, 2008, 51(6):760-764. doi: 10.1002/pbc.21699 [24] Bansal D, Das A, Trehan A, et al. Pediatric rhabdomyosarcoma in india:a single-center experience[J]. Indian Pediatr, 2017, 54(9):735-738. doi: 10.1007/s13312-017-1164-5 [25] 李艳华, 杨静薇, 廖雪莲, 等.儿童横纹肌肉瘤分层化疗疗效及生存因素分析[J].同济大学学报(医学版), 2020, 41(3):373-377. [26] 苗真, 王娴静, 孙静, 等.儿童横纹肌肉瘤17例病例分析[J].中国小儿血液与肿瘤杂志, 2017, 22(3):159-163. doi: 10.3969/j.issn.1673-5323.2017.03.009 [27] 徐娜, 段超, 金眉, 等.单中心多学科联合诊治儿童横纹肌肉瘤的临床及预后分析[J].中华儿科杂志, 2019, 57(10):767-773. doi: 10.3760/cma.j.issn.0578-1310.2019.10.008 [28] Doyen J, Jazmati D, Geismar D, et al. Outcome and patterns of relapse in childhood parameningeal rhabdomyosarcoma treated with proton beam therapy[J]. Int J Radiat Oncol Biol Phys, 2019, 105(5):1043-1054. doi: 10.1016/j.ijrobp.2019.08.005 [29] Mohan AC, Venkatramani R, Okcu MF, et al. Local therapy to distant metastatic sites in stage Ⅳ rhabdomyosarcoma[J]. Pediatr Blood Cancer, 2018, 65(2):10. [30] Mohan AC, Venkatramani R, Okcu MF, et al. Local therapy to distant metastatic sites in stage Ⅳ rhabdomyosarcoma[J]. Pediatr Blood Cancer, 2018, 65(2):10. -




 下载:
下载: