Differences in brain cortical structure between non-small cell lung cancer patients and healthy controls: a retrospective magnetic resonance imaging study
-
摘要:
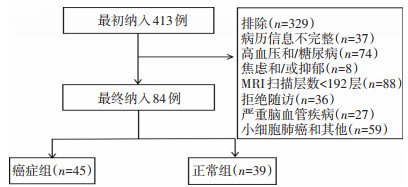
目的 分析非小细胞肺癌(non-small cell lung cancer,NSCLC)患者颅脑核磁共振影像学(magnetic resonance imaging,MRI)改变,并探讨其相关的影响因素,为患者的脑健康预防及保护提供依据。 方法 选取2018年7月至2019年3月四川省肿瘤医院45例初诊为NSCLC(癌症组)及39例健康对照组(正常组)颅脑MRI数据,利用Freesurfer软件对MRI数据进行基于大脑皮层表面形态学测量(surface-based morphometry,SBM)分析,统计对比癌症组与正常组的脑区变化,利用偏相关分析方法将血常规、血脂、肿瘤标志物与变化脑区行相关性分析。 结果 与正常组相比,癌症组右脑中央后回及顶叶上回脑皮质体积明显萎缩(P < 0.05),但其厚度、表面积、曲率、脑沟深度未见明显变化(P>0.05)。此外,癌症组神经元特异性烯醇化酶(neuron-specific enolase,NSE)水平明显高于正常组[(27.02±33.16)ng/mL vs.(7.8±3.85)ng/mL,P < 0.05],相关性分析结果显示中央后回体积萎缩与NSE呈负相关(r=-0.268,P=0.039),血常规、血脂、癌胚抗原(carcinoembryonic antigen,CEA)与中央后回体积萎缩无明显相关性。 结论 NSCLC可导致右脑中央后回及顶叶上回体积缩小,且中央后回体积缩小与NSE呈显著负相关,提示NSE可作为预测NSCLC相关脑损伤的潜在预测分子。 -
关键词:
- 肺癌 /
- 非小细胞肺癌 /
- 大脑皮层 /
- 癌症相关认知功能障碍
Abstract:Objective To analyze changes on brain magnetic resonance imaging (MRI) in patients with non-small cell lung cancer (NSCLC) and to determine the influencing factors to provide evidence for the prevention and maintenance of brain health in patients with cancer. Methods Forty-five newly diagnosed patients with NSCLC (cancer group) and 39 healthy controls (control group) were enrolled at Sichuan Cancer Hospital & Institute from July 2018 to March 2019. MRI data were analyzed based on surface-based morphometry (SBM) of the cerebral cortex. Changes in brain regions were statistically compared between the two groups. Routine blood markers, blood lipid profiles, tumor markers, and changes in brain regions were analyzed using partial correlation analysis. Results Regarding changes in cerebral cortex volume, the right posterior central gyrus and right superior parietal gyrus were significantly atrophied in the cancer group compared to that in the control group (P < 0.05); however, the thickness, surface area, curvature, and sulcus depth were not significantly different (P>0.05). In addition, the level of neuron-specific enolase (NSE) in the cancer group was significantly higher than that in the control group [(27.02± 33.16) ng/mL vs. (7.8±3.85) ng/mL, P < 0.05]. Partial correlation analysis showed that the atrophy volume of the central posterior gyrus was negatively correlated with NSE levels (r=-0.268, P=0.039); However, the atrophy volume of the central posterior gyrus was not significantly correlated with routine blood markers, blood lipid profiles, and carcinoembryonic antigen (CEA) levels. Conclusions Volumes of the posterior central gyrus and superior parietal gyrus of the right brain are atrophied in NSCLC patients, and volume reduction in the posterior central gyrus is significantly negatively correlated with NSE levels. These findings suggest that NSE can be used as a potential predictor of NSCLCrelated brain damage. -
表 1 癌症组与正常组的临床资料

表 2 正常组与癌症组皮层体积变化脑区

-
[1] Oxman TE, Silberfarb PM. Serial cognitive testing in cancer patients receiving chemotherapy[J]. Am J Psychiatry, 1980, 137(10): 1263-1265. doi: 10.1176/ajp.137.10.1263 [2] Ahles TA, Saykin AJ. Candidate mechanisms for chemotherapy-induced cognitive changes[J]. Nat Rev Cancer, 2007, 7(3): 192-201. doi: 10.1038/nrc2073 [3] Simó M, Rifà- Ros X, Rodriguez- Fornells A, et al. Chemobrain: A systematic review of structural and functional neuroimaging studies[J]. Neurosci Biobehav Rev, 2013, 37(8): 1311-1321. doi: 10.1016/j.neubiorev.2013.04.015 [4] Asher A, Myers JS. The effect of cancer treatment on cognitive function [J]. Clin Adv Hematol Oncol, 2015, 13(7): 441-450. http://www.ncbi.nlm.nih.gov/pubmed/26353040 [5] Chao HH, Hu S, Ide JS, et al. Effects of androgen deprivation on cerebral morphometry in prostate cancer patients-an exploratory study[J]. PLoS One, 2013, 8(8): e72032. doi: 10.1371/journal.pone.0072032 [6] Zec RF, Trivedi MA. The effects of estrogen replacement therapy on neuropsychological functioning in postmenopausal women with and without dementia: a critical and theoretical review[J]. Neuropsychol Rev, 2002, 12(2): 65-109. doi: 10.1023/A:1016880127635 [7] Horowitz TS, Suls J, Treviño M. A call for a neuroscience approach to cancer-related cognitive impairment[J]. Trends Neurosci, 2018, 41(8): 1-3. http://europepmc.org/abstract/MED/29907436 [8] Liu S, Li X, Ma R, et al. Cancer-associated changes of emotional brain network in non-nervous system metastatic non-small cell lung cancer patients: a structural connectomic diffusion tensor imaging study[J]. Transl Lung Cancer Res, 2020, 9(4): 1101-1111. doi: 10.21037/tlcr-20-273 [9] Kesler SR, Adams M, Packer M, et al. Disrupted brain network functional dynamics and hyper- correlation of structural and functional connectome topology in patients with breast cancer prior to treatment [J]. Brain Behav, 2017, 7(3): e00643. doi: 10.1002/brb3.643 [10] Menning S, de Ruiter MB, Veltman DJ, et al. Multimodal MRI and cognitive function in patients with breast cancer prior to adjuvant treatment—the role of fatigue[J]. NeuroImage Clin, 2015, 7: 547-554. doi: 10.1016/j.nicl.2015.02.005 [11] Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424. doi: 10.3322/caac.21492 [12] Kauffmann-Guerrero D, Kahnert K, Huber RM. Treatment sequencing for anaplastic lymphoma kinase-rearranged non-small-cell lung cancer [J]. Drugs, 2021, 81(1): 87-100. doi: 10.1007/s40265-020-01445-2 [13] Groeneveld O, Reijmer Y, Heinen R, et al. Brain imaging correlates of mild cognitive impairment and early dementia in patients with type 2 diabetes mellitus[J]. Nutr Metab Cardiovasc Dis, 2018, 28(12): 1-8. http://www.ncbi.nlm.nih.gov/pubmed/30355471 [14] Rom S, Zuluaga-Ramirez V, Gajghate S. Hyperglycemia-driven neuroinflammation compromises BBB leading to memory loss in both diabetes mellitus (DM) type 1 and type 2 mouse models[J]. Mol Neurobiol, 2019, 56(3): 1883-1896. doi: 10.1007/s12035-018-1195-5 [15] Mossello E, Simoni D. High blood pressure in older subjects with cognitive impairment[J]. Monaldi Arch Chest Dis, 2016, 84(1-2): 32-36. http://www.ncbi.nlm.nih.gov/pubmed/27374044 [16] Amidi A, Wu LM. Structural brain alterations following adult non-CNS cancers: a systematic review of the neuroimaging literature[J]. Acta Oncol, 2019, 58(5): 522-536. doi: 10.1080/0284186X.2018.1563716 [17] Madhyastha S, Somayaji SN, Rao MS, et al. Hippocampal brain amines in methotrexate- induced learning and memory deficit[J]. Can J Physiol Pharmacol, 2002, 80(11): 1076-1084. doi: 10.1139/y02-135 [18] Bartzokis G, Beckson M, Lu PH, et al. Age-related changes in frontal and temporal lobe volumes in men: a magnetic resonance imaging study [J]. Arch Gen Psychiatry, 2001, 58(5): 461- 465. doi: 10.1001/archpsyc.58.5.461 [19] Raz N, Lindenberger U, Rodrigue KM, et al. Regional brain changes in aging healthy adults: general trends, individual differences and modifiers[J]. Cereb Cortex, 2005, 15(11): 1676-1689. doi: 10.1093/cercor/bhi044 [20] Resnick SM, Pham DL, Kraut MA, et al. Longitudinal magnetic resonance imaging studies of older adults: a shrinking brain[J]. J Neurosci, 2003, 23(8): 3295-3301. doi: 10.1523/JNEUROSCI.23-08-03295.2003 [21] Sowell ER, Thompson PM, Toga AW. Mapping changes in the human cortex throughout the span of life[J]. Neuroscientist, 2004, 10(4): 372- 392. doi: 10.1177/1073858404263960 [22] Debette S, Seshadri S, Beiser A, et al. Midlife vascular risk factor exposure accelerates structural brain aging and cognitive decline[J]. Neurology, 2011, 77(5): 461-468. doi: 10.1212/WNL.0b013e318227b227 [23] Li WL, Fu C, Xuan A, et al. Preliminary study of brain glucose metabolism changes in patients with lung cancer of different histological types[J]. Chin Med J (Engl), 2015, 128(3): 301-304. doi: 10.4103/0366-6999.150089 [24] Simo M, Root JC, Vaquero L, et al. Cognitive and brain structure changes in a lung cancer population[J]. J Thorac Oncol, 2015, 10(1): 38-45. doi: 10.1097/JTO.0000000000000345 [25] Velásquez C, Goméz E, Martino J. Mapping visuospatial and self-motion perception functions in the left parietal lobe[J]. Neurosurg Focus, 2018, 45(Suppl2): V8. http://www.ncbi.nlm.nih.gov/pubmed/30269556 [26] Allavena P, Mantovani A. Immunology in the clinic review series; focus on cancer: Tumour associated macrophages: undisputed stars of the inflammatory tumour microenvironment[J]. Clin Exp Immunol, 2012, 167(2): 195-205. doi: 10.1111/j.1365-2249.2011.04515.x [27] Zipp F, Aktas O. The brain as a target of inflammation: common pathways link inflammatory and neurodegenerative diseases[J]. Trends Neurosci, 2006, 29(9): 518-527. doi: 10.1016/j.tins.2006.07.006 [28] Koh YQ, Tan CJ, Toh YL, et al. Role of exosomes in cancer-related cognitive impairment[J]. Int J Mol Sci, 2020, 21(8): 2755-2770. doi: 10.3390/ijms21082755 [29] Hinzman CP, Baulch JE, Mehta KY, et al. Plasma-derived extracellular vesicles yield predictive markers of cranial irradiation exposure in mice [J]. Sci Rep, 2019, 9(1): 9460-9468. doi: 10.1038/s41598-019-45970-x [30] Schmechel D, Marango P, Brightman M. Neurone-specific enolase is a molecular marker for peripheral and central neuroendocrine cells[J]. Nature, 1978, 276(5690): 834-836. doi: 10.1038/276834a0 [31] Tapia FJ, Polak J, Barbosa AJ, et al. Neuron-specific enolase is produced by neuroendocrine tumours[J]. Lancet, 1981, 1(8224): 808-811. http://europepmc.org/abstract/MED/6111674 [32] Lamers KJ, Vos P, Verbeek MM, et al. Protein S-100B, neuron-specific enolase (NSE), myelin basic protein (MBP) and glial fibrillary acidic protein (GFAP) in cerebrospinal fluid (CSF) and blood of neurological patients[J]. Brain Res Bull, 2003, 61(3): 261-264. doi: 10.1016/S0361-9230(03)00089-3 [33] Huang Z, Xu D, Zhang F, et al. Pro-gastrin-releasing peptide and neuronspecific enolase: useful predictors of response to chemotherapy and survival in patients with small cell lung cancer[J]. Clin Transl Oncol, 2016, 18(10): 1019-1025. doi: 10.1007/s12094-015-1479-4 [34] Zhou M, Wang Z, Yao Y, et al. Neuron-specific enolase and response to initial therapy are important prognostic factors in patients with small cell lung cancer[J]. Clin Transl Oncol, 2017, 19(7): 865-873. doi: 10.1007/s12094-017-1617-2 [35] Liu X, Zhang W, Yin W, et al. The prognostic value of the serum neuron specific enolase and lactate dehydrogenase in small cell lung cancer patients receiving first-line platinum-based chemotherapy[J]. Medicine (Baltimore), 2017, 96(46): e8258. doi: 10.1097/MD.0000000000008258 [36] Dong Y, Zheng X, Yang Z, et al. Serum carcinoembryonic antigen, neuronspecific enolase as biomarkers for diagnosis of non-small cell lung cancer[J]. J Cancer Res Ther, 2016, 12(Suppl): 34-36. http://www.ncbi.nlm.nih.gov/pubmed/27721249 [37] Zhou Y, Chen WZ, Peng A, et al. Neuron-specific enolase, histopathological types, and age as risk factors for bone metastases in lung cancer[J]. Tumour Biol, 2017, 39(7): 1-5. http://www.ncbi.nlm.nih.gov/pubmed/28671048 [38] 陈燕, 彭伟, 黄艳芳, 等. 治疗前血清神经元特异性烯醇化酶水平在预测晚期非小细胞肺癌脑转移及预后中的意义[J]. 中华肿瘤杂志, 2015, 37(7): 508-511. doi: 10.3760/cma.j.issn.0253-3766.2015.07.006 [39] Tiseo M, Ardizzoni A, Cafferata MA, et al. Predictive and prognostic significance of neuron- specific enolase (NSE) in non-small cell lung cancer[J]. Anticancer Res, 2008, 28(1B): 507-513. http://www.ncbi.nlm.nih.gov/pubmed/18383893 [40] Ganti L, Serrano E, Toklu HZ. Can neuron specific enolase be a diagnostic biomarker for neuronal injury in COVID-19[J]? Cureus, 2020, 12 (10): e11033. http://www.researchgate.net/publication/346279323_Can_Neuron_Specific_Enolase_Be_a_Diagnostic_Biomarker_for_Neuronal_Injury_in_COVID-19 -




 下载:
下载: